Abstract
The fishery of inshore hagfish (Eptatretus burgeri) is particularly important from the perspective of the eel-skin leather industry in the northwest Pacific. In order to reveal the genetic diversity and population structure of E. burgeri in the northwest Pacific, we analyzed partial nucleotide sequences of three mitochondrial DNA regions (523 bp in COI, 712 bp in ND4 and 617 bp in Cytb) based on specimens collected from six locations in Korea and Japan. The genetic diversities of E. burgeri were higher in Korean locations compared to Japanese ones. AMOVA showed that E. burgeri was completely separated into two groups (group A: southern coast of Korea and western coast of Japan vs. group B: eastern coast of Japan). Furthermore, groups A and B were divided into each two lineages (lineage I: west southern coast of Korea, lineage II: east southern coast of Korea and western coast of Japan, lineage III and IV: eastern coast of Japan). Our molecular results suggest that these two groups and lineages of E. burgeri may be different evolutionary significant unit and management unit, respectively.
Introduction
Hagfish has a high commercial value and significant economic importance from the perspective of the fisheries industry (eel-skin leather and food) in the northwest Pacific (Kase et al. Citation2018). Hagfish catches have greatly declined over the last decades in Korean and Japanese waters (McMillan and Wisner Citation2004; Knapp et al. Citation2011). Eptatretus burgeri, which is major commercial species, is found in the restricted patch range around the northwest Pacific such as Korea, Japan, and Taiwan (Mok and Chen Citation2001; Froese and Pauly Citation2019). Most in Korea is incidentally caught by fish traps for Conger myriaster and directly targeted by using longlines, and its production is approximately within 93 tons during a five-year period (MOF Citation2020). There are no regulations governing the harvesting of E. burgeri. Mitochondrial DNA (mtDNA) has proven to be useful for population genetic studies (Avise et al. Citation1987; Schultheis et al. Citation2002). To manage a sustainable E. burgeri fishery, we investigated the genetic diversity and population structure of Korean and Japanese E. burgeri using three Mitochondrial DNA (mtDNA) sequence markers.
Materials and methods
Sampling
A total of 180 specimens of E. burgeri were collected from Tongyeong (TY; n = 38; 34°39′1.21″N; 128°26′6.15″E), Wando Island (WD; n = 30; 33°59′56.5″N 126°53′57.0″E), Jeju Island (JJ; n = 33; 33°37′51.5″N 126°53′11.4″E) in Korean waters, and Kyoto (KT; n = 31; 35°40′29.1″N 135°27′03.4″E), Tsushima Island (TS; n = 30; 34°20′21.5″N 129°28′23.9″E), Shikoku Island (SK; n = 18; 32°47′19.2″N 132°32′55.6″E) in Japanese waters between May 2014 and Sep 2017, caught by hook and line, and fishing trap (Figure S1). The specimens have been deposited in the Marine Fish Resource Bank of Korea (MFRBK) at Pukyong National University (PKU), Korea. We loaned the Kyoto specimens and received the Shikoku Island specimens from Kyoto University (FAKU).
DNA extraction, PCR and sequencing, and data analysis
Genomic DNA was extracted from the muscle tissues using DNA Extraction Kit (Bioneer Trade Co. Ltd, Korea). We analyzed three mitochondrial DNA markers: Cytochrome c oxidase subunit I (COI), NADH dehydrogenase subunit four (ND4), and cytochrome b (Cytb). PCR was then performed using an MJ Mini Thermal Cycler PTC-1148 (Bio-Rad) in mixtures consisting 1 μL of genomic DNA (ca. 100 ∼ 200 ng), 2 μL of 10× PCR buffer, 1.6 μL of 2.5 mM dNTPs, 1 μL of each primer (10 pmol), 0.1 μL of TaKaRa EX-Taq polymerase (TaKaRa Bio Inc., Kyoto, Japan), and distilled water to bring the final volume to 20 μL. PCR products were amplified using universal and species-specific designed primers (Table S1). The PCR profile for the mtDNA COI consisted of initial denaturation at 94 °C for 4 min, followed by 34 cycles of denaturation at 94 °C for 30 s, annealing at 52 °C for 30 s, extension at 72 °C for 30 s, and a final extension at 72 °C for 8 min; for the mtDNA ND4 gene consisted of initial denaturation at 94 °C for 3 min, followed by 34 cycles of denaturation at 94 °C for 30 s, annealing at 60 °C for 1 min, extension at 72 °C for 5 min, and a final extension at 72 °C for 10 min; and for the mtDNA Cytb consisted of initial denaturation at 94 °C for 5 min, followed by 35 cycles of denaturation at 94 °C for 30 s, annealing at 58–60 °C for 1 min, extension at 72 °C for 1 min, and a final extension at 72 °C for 7 min. The PCR products were purified using a Davinch™ PCR Purification Kit (Davinch-K Co. Ltd, Seoul, Korea). The purified products of mitochondrial DNA were sequenced based on the Sanger sequencing method with an Applied Biosystems ABI 3730XL analyzer (Applied Biosystems, Foster City, CA) using an ABI PRISM BigDyeTM Terminator Cycle Sequencing Ready Reaction Kit v3.1 (Applied Biosystems). The sequences were aligned using ClustalW (Thompson et al. Citation1994) in BioEdit version 7 (Hall 1999).
Genetic diversities were evaluated using Arlequin 3.5.1.2 (Excoffier et al. Citation2005). To quantify the genetic differences among the six locations, pairwise ΦST values were calculated with 10,000 permutations. Phylogenetic trees were constructed: Neighbor-joining (NJ) and Bayesian inference (BI). The genetic distances were calculated using the Kimura 2-parameter (K2P) model (Kimura Citation1980) with Mega 6 (Tamura et al. Citation2013), which showed that the intraspecific genetic distance, and confidence was assessed with 1000 bootstrap replications. The best-fit model of sequence evolution in BI was selected using MrModeltest 2.3 (Nylander Citation2004). BI was constructed using BEAST 2.5.1 (Drummond and Bouckaert Citation2015), and importing data and specifying the evolutionary models were done using BEAUti (Drummond et al. Citation2012). The Markov Chain Monte Carlo (MCMC) analysis of region was run for 10 million generations. The consensus tree along with posterior probabilities was visualized using FigTree Ver. 1.4.3. (Rambaut Citation2016). The numbers on the nodes indicated posterior probability in the BI. We also performed an analysis of molecular variance, hierarchical AMOVA (analysis of molecular variation) using Arlequin 3.5.1.2. (Excoffier et al. Citation1992). It was performed to test the best hierarchical groupings based on three alternative hypotheses in geographic structure. The significance of the observed variances for each hierarchical comparison was tested by 10,000 permutations. The haplotype sequences of each region have been deposited with GenBank (MN921151-MN921161, COI; MN965669-MN965691, ND4; MN965641-MN965668, Cytb).
Results and discussion
Sequence variation and genetic diversity
All populations displayed various numbers of mitochondrial haplotypes, with a total 11 haplotypes detected among 124 individuals for COI, 23 haplotypes detected among 180 individuals for ND4, and 28 haplotypes detected among 152 individuals for Cytb (Table S2). Analyses of 1852 bp of the combined mtDNA (COI, ND4, and Cytb) identified 43 haplotypes detected among 112 individuals from six locations (Table S2). In the combined dataset, HP1 and HP3 were the most abundant haplotypes, appearing 19 and 11 times, respectively. Ten haplotypes (HP34 to HP43) were shared by only Shikoku Island. Compared molecular diversities, the Korean E. burgeri generally showed higher haplotype diversity (0.8696–1.0000) and nucleotide diversity (0.001–0.003) than the Japanese one (0.6209–0.9381, 0.000–0.003, respectively) based on combined mtDNA (). In case of the brown hagfish, Eptatretus atami, they were genetically different in the eastern and the western coast of Japan and their genetic diversity indicated that genetic diversity of E. atami in the eastern coast of Japan was higher than those of E. atami in the western coast of Japan (Kase et al. Citation2018). When compared between lampreys and hagfishes which are cyclostomata, Mateus et al. (Citation2013) reported that Lampetra fluviatilis (lamprey) showed high haplotype diversity and low nucleotide diversity, and revealed that the existence of four highly divergent allopatric groups within the Iberian Peninsula. The genetic diversity of E. burgeri in Kyoto was the lowest, but those of other locations showed higher molecular diversities. Density stratification in the water column occurred in the western coast of Japan during the LGM, and have resulted in serious anaerobic water columns at the sea bottom (Itaki et al. Citation2004). Thus, E. burgeri in Kyoto might have experienced extinction and recolonization in anoxic bottom condition during glacial and interglacial cycle. Some deep-sea species might have experienced a rapid decrease in anaerobic or hypoxia environment after the western coast of Japan was separated from Pacific Ocean during the LGM, which might have influenced their low genetic diversity (Adachi et al. Citation2009; Sakuma et al. Citation2014; Habib et al. Citation2015).
Table 1. Summary of genetic variability in the four lineages and two groups of Eptatretus burgeri based on the combined mtDNA (COI + ND4 + Cytb).
Phylogenetic analysis and population structure
Eptatretus burgeri were geographically structured in two distinct groups (Groups A and B) in the northwest Pacific (). The genetic distances between Group A and B were 1.1–1.6%, while the within-groups distances were 0.3–0.4% based on combined DNA (COI + ND4 + Cytb). Group A included E. burgeri collected from southern coast of Korea and western coast of Japan, whereas Group B included E. burgeri collected from the eastern coast of Japan. Group A is geographically divided into two lineages; lineage I which is mainly detected in west-southern coast of Korea, and lineage II which is mainly detected in east-southern coast of Korea and western coast of Japan. Group B also consists of two lineages, lineage III, and lineage IV ().
Figure 1. The phylogenetic tree based on the neighbor-joining and Bayesian Inferences for the 43 combined mtDNA region (COI + ND4 + Cytb) haplotypes of Eptatretus burgeri. Eptatretus atami was chosen as an outgroup. The numbers on the node is bootstrap value (%) by 1000 replicates in Neighbor-joining methods and posterior probability by Bayesian inferences.
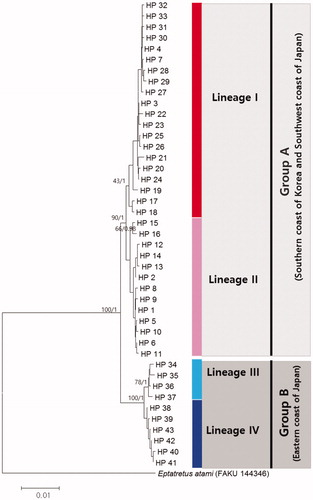
Most ΦST population pairwise comparisons were significant (p < .01) except for the ΦST value between Tongyeong and Tsushima Island, and between Jeju Island and Wando Island (Table S3). The pairwise ΦST values between Shikoku Island and the other five locations were especially high than the values among the others (Table S3). The highest ΦCT value, which indicates the highest proportion of genetic variance able to be attributed to groups given the present data, was observed as 0.684 (p < .000) when the locations were grouped into following two groups: (TY&JJ&WD&TS&KT) and SK (). Shikoku Island (SK) showed significant genetic differentiation and no shared haplotypes between other locations, indicating no gene flow between two groups. Generally, hagfishes tend to live and breed locally (Walvig Citation1963) as well as a unique reproductive cycle (e.g. no pelagic larvae) and a lack of migration (Patzner Citation1978). Therefore, their biological trait and no gene flow might have facilitated allopatric speciation of group A and B of hagfish in the quite different marine environment. In case of benthic fish, the restricted gene flow between the western and eastern coast of Japan of Okamejei kenojei have been derived from constrains of low dispersal because of large benthic egg capsules and the absence of a pelagic larval stage (Misawa et al. Citation2018). Wando Island and Jeju Island (west) are effectively separated from Tongyeong and Tsushima Island (east) by the Seomjin River. Wando Island was only included in lineage I for the COI and the combined mtDNA, and Kyoto was only included in lineage II for all locations except for the Cytb (Figure S2). The different proportion of lineage I to lineage II between the west and the east might have been caused by this low-salinity boundary. In the past, the incised-channel system of the paleo-Seomjin River has significantly affected the sedimentary processes of the southern sea of Korea (Kim et al. Citation2019). Actually, the two lineages of Ammodytes japonicus may be influenced from physical barriers of halocline caused by the Nakdong River in Korea (Kim et al. Citation2015; Kim et al. Citation2017). Therefore, the presence of a strong physical barrier and low salinity water column may act as a biogeographical boundary between West and East.
Table 2. AMOVA for E. burgeri based on the combined mtDNA (COI + ND4 + Cytb) haplotypes from six local populations allocated into two, three or four groups.
The distribution pattern of hagfish could reflect the habitat preferences (TNC Citation2019). Because hagfishes tend to prefer a habitat of mud or muddy sand bottom (Fernholm Citation1974), environmental change including topographic change might affect the distribution and genetic structure of hagfishes. The temperature in the southern coast of Korea have lower than the western coast of Japan, and the bottom substrate of the southern coast of Korea is made of a higher proportion of sand and muddy than those of the western coast of Japan (Yoo et al. Citation2014). Therefore, high adaptation to habitat might have resulted in higher genetic diversities in the Korean E. burgeri than the Japanese one. In demersal and deep-sea fish similar to the habitat trait of hagfish, another reason to affect gene flow may consider the biogeographical barrier. Lycodes matsubarai indicated the high genetic population structure because they have been restricted post-glacial migration between western coast of Japan and Sea of Okhotsk by the shallow strait (13–55 m depth) (Sakuma et al. Citation2014).
Some studies have revealed that genetic groups or lineages with high divergence could be considered a complex of incipient or cryptic resident species, which allows the definition of evolutionary significant unit (ESU) and management unit (MU) (Ryder Citation1986; Mateus et al. Citation2013; Guo et al. Citation2014). Considering the apparent genetic divergence and different genetic diversity between the two groups, we proposed that the E. burgeri in the southern coast of Korea and the western coast of Japan should be recognized as distinct ESU differing from the E. burgeri in the eastern coast of Japan. In addition, the E. burgeri in southern coast of Korea should be recognized as distinct MU differing from the E. burgeri in the western coast of Japan.
Supplemental Material
Download MS Word (361 KB)Acknowledgements
We sincerely thank Dr. Yoshiaki Kai (Kyoto University) and Dr. Naohide Nakayama (Kyoto University Museum) for his kind sampling of the Japanese hagfish. We also thank two anonymous reviewers for their valuable comments that improved the quality of this article.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Data availability statement
The data that support the findings of this study are openly available in [NCBI] at [https://www.ncbi.nlm.nih.gov/], reference number [MN921151-MN921161; MN965669-MN965691; MN965641-MN965668]. The other data generated or analyzed during this study are included in this published article and its supplementary information files.
Additional information
Funding
References
- Adachi T, Hagihara S, Itoh M, Shinohara G. 2009. Genetic population structure and morphological characters of Japanese psychrolutids of genus Malacocottus (Scorpaeniformes: Psychrolutidae. Ichthyol Res. 56(4):323–329.
- Avise JC, Arnold J, Ball RM, Bermingham E, Lamb T, Neigel JE, Reeb CA, Saunders NC. 1987. Intraspecific phylogeography: the mitochondrial DNA bridge between population genetics and systematics. Annu Rev Ecol Syst. 18(1):489–522.
- Drummond AJ, Bouckaert RR. 2015. Bayesian evolutionary analysis with BEAST. Cambridge: Cambridge University Press; pp. 249.
- Drummond AJ, Suchard MA, Xie D, Rambaut A. 2012. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol. 29(8):1969–1973.
- Excoffier L, Laval G, Schneider S. 2005. Arlequin ver. 3.0: an integrated software package for population genetics data analysis. Evol Bionform Online. 1:47–50.
- Excoffier L, Smouse P, Quattro J. 1992. Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics. 131(2):479–491.
- Fernholm B. 1974. Diurnal variations in the behaviour of the Hagfish Eptatretus burgeri. Mar Biol. 27(4):351–356.
- Froese R, Pauly D, editors. 2019. FishBase. World Wide Web electronic publication. version (12/2019). www.fishbase.org
- Guo SS, Zhang GR, Guo XZ, Wei KJ, Yang RB, Wei QW. 2014. Genetic diversity and population structure of Schizopygopsis younghusbandi Regan in the Yarlung Tsangpo River inferred from mitochondrial DNA sequences analysis. Biochem Syst Ecol. 57:141–151.
- Habib KA, Jeong D, Myoung JG, Lee YH. 2015. Population panmixia and the pleistocene demographic expansion of spotty belly greenling Hexagrammos agrammus in the East Sea and Northwest Pacific. Ocean Sci J. 50(2):143–154.
- Itaki T, Ikehara K, Motoyama I, Hasegawa S. 2004. Abrupt ventilation changes in the Japan Sea over the last 30 ky: evidence from deep-dwelling radiolarians. Palaeogeogr Palaeoclimatol Palaeoecol. 208(3-4):263–278.
- Kase M, Shimizu T, Kamino K, Umetsu K, Sugiyama H, Kitano T. 2018. Brown hagfish from the northwest and east coasts of Honshu, Japan are genetically different. Genes Genet Syst. 92(4):197–203.
- Kim JK, Bae SE, Lee SJ, Yoo MG. 2017. New insight into hybridization and unidirectional introgression between Ammodytes japonicus and Ammodytes heian (Trachiniformes, Ammodytidae). PLOS One. 12(6):e0178001.
- Kim JC, Cheong D, Shin S, Park YH, Hong SS. 2015. OSL chronology and accumulation rate of the Nakdong deltaic sediments, southeastern Korean Peninsula. Quat Geochronol. 30:245–250.
- Kim GY, Park JK, Lee GS, Yoo DG, Kong GS. 2019. Physical property characterization of quaternary sediments in the vicinity of the paleo-Seomjin River of the continental shelf of the South Sea, Korea. Quatern Int. 503:153–162.
- Kimura M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol. 16(2):111–120.
- Knapp L, Mincarone MM, Harwell H, Polidoro B, Sanciangco J, Carpenter K. 2011. Conservation status of the world’s hagfish species and the loss of phylogenetic diversity and ecosystem function. Aquatic Conserv Mar Freshw Ecosyst. 21(5):401–411.
- Mateus CS, Stange M, Berner D, Roesti M, Quintella BR, Alves BJ, Almeida PR, Salzburger W. 2013. Strong genome-wide divergence between sympatric European river and brook lampreys. Curr Biol. 23:649–650.
- McMillan CB, Wisner RL. 2004. Review of the Hagfishes (Myxinidae: Myxiniformes) of the Northwestern Pacific Ocean, with Descriptions of Three New Species, Eptatretus fernholmi, Paramyxine moki. P walker. Zool Stud. 43:51–73.
- Misawa R, Narimatsu Y, Endo H, Kai Y. 2018. Population structure of the ocellate spot skate (Okamejei kenojei) inferred from variations in mitochondrial DNA (mtDNA) sequences and from morphological characters of regional populations. FB. 117(1):24–36.
- MOF (Ministry of Oceans and Fisheries). 2020. Fisheries Information Service, http://www.fips.go.kr/
- Mok H, Chen YW. 2001. Distribution of hagfish (Myxinidae: Myxiniformes) in Taiwan. Zool Stud. 40:233–239.
- Nylander JAA. 2004. MrModeltest v2. Program distributed by the author. Uppsala: Evolutionary Biology Centre, Uppsala University.
- Patzner RA. 1978. Experimental studies on the light sense in the hagfish, Eptatretus burgeri and Paramyxine atami (Cyclostomata). Helgoliinder wiss. Meeresunters. 31:180–190.
- Rambaut A. 2016. FigTree, version 1.4.3. [accessed 2017 Feb 22]. http://tree.bio.ed.ac.uk/software/figtree/.
- Ryder OA. 1986. Species conservation and systematics: the dilemma of the subspecies. Trends Ecol Evol. 1(1):9–10.
- Sakuma K, Ueda Y, Hamatsu T, Kojima S. 2014. Contrasting population histories of the deep-sea demersal fish, Lycodes matsubarai, in the sea of Japan and the sea of Okhotsk. Zool Sci. 31(6):375–382.
- Schultheis AS, Weigt LA, Hendricks AC. 2002. Gene flow, dispersal, and nested clade analysis among populations of the stonefly Peltoperla tarteri in the southern Appalachians. Mol Ecol. 11(3):317–327.
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. 2013. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 30(12):2725–2729.
- Thompson JD, Higgins DG, Gibson TJ. 1994. Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22:4673–4680.
- TNC (Tha Nature Conservancy). 2019. Resource and market analysis of Oregon’s hagfish fishery. In preparation for a future fishery management plan. Oregon. pp. 49.
- Walvig F. 1963. Gonads and the formation of sexual cells. In: Brodal A, Fange F, editors. The biology of Myxine. Oslo: Universitetforlaget; p. 530–580.
- Yoo DG, Kim SP, Lee CW, Chang TS, Kang NK, Lee GS. 2014. Late Quaternary transgressive deposits in a low-gradient environmental setting: Korea Strait shelf, SE Korea. Quatern Int. 344:143–155.
