Abstract
Objective
The aim of this study was to evaluate the efficacy, cardiotoxicity profile and long-term benefits of neoadjuvant therapy in human epidermal growth factor receptor 2-positive operable breast cancer patients.
Patients and methods
A total of 142 patients diagnosed from 2005 to 2016 were included in the study. The treatment consisted of a sequential regimen of taxanes and anthracyclines plus trastuzumab. The clinical and pathological responses were evaluated and correlated with clinical and biological factors. The cardiotoxicity profile and long-term benefits were analyzed.
Results
The median age was 49 years, and 4%, 69% and 27% of patients had stage I, II and III breast cancer, respectively, while 10% had inflammatory breast cancer at diagnosis. Hormone receptor (HR) status was negative in 43%, and 62% had grade III breast cancer. The clinical complete response rate was 49% and 63% as assessed using ultrasound and magnetic resonance imaging, respectively, and this allowed a high rate of conservative surgery (66%). The pathological complete response (pCR) rate was 52%, and it was higher in HR-negative (64%) patients than in HR-positive (41%) patients and in grade III breast cancer (53%) patients than in grade I–II breast cancer (45%) patients. Patients who achieved pCR had longer disease-free survival and a trend toward improved overall survival. A total of 2% of patients showed a 10% decrease in left ventricular ejection fraction to <50% during treatment. All patients except one recovered after discontinuation of trastuzumab.
Conclusion
A sequential regimen of taxanes and anthracyclines plus trastuzumab was effective, with high pCR rates and long-term benefit, and had a very good cardiotoxicity profile.
Introduction
Neoadjuvant therapy (NT) is the standard of care for locally advanced and inflammatory breast cancer.Citation1–Citation4
It is now known that it offers similar benefits like adjuvant therapy in terms of disease-free survival (DFS) and overall survival (OS),Citation5,Citation6 and so NT is currently implemented for operable disease.
Currently, NT is generally used to improve surgical options, to determine the response to therapy, and it is expected to produce long-term benefits.
Aggressive breast cancer subtypes benefit the most from primary systemic therapy: these include human epidermal growth factor receptor 2 (HER2)-positive and triple-negative tumors.Citation7
In HER2-positive tumors, a combination of taxanes and anthracyclines plus trastuzumab has been the standard treatment.Citation8–Citation13
Trastuzumab interacts with the extracellular domain of HER2 and inhibits signaling from HER2 homodimers more effectively than from HER2 heterodimers with EGF receptor 1 (HER1) or EGF receptor 3 (HER3). This results in the downregulation of the phosphatidylinositol-3 kinase (PI3K) pathway and cell apoptosis. Trastuzumab can also induce antibody-dependent cellular cytotoxicity.Citation14–Citation16
Despite the advance in the knowledge of HER2-positive tumors and improvements in treatments, a small percentage of patients still suffer from recurrence and die.
Many mechanisms underlying resistance to anti-HER2 therapies have been studied. They include three major groups. The first group involves the concept of “redundancy”. Examples include the inhibition of the incomplete receptor family by a truncated form of the HER2 receptor (p95-HER2) and the lack of exon 16 in the extracellular domain of the HER2 receptor (D16 isoform).
The second major group involves “reactivation”. Examples include the deregulation of the PI3K pathway because of mutations in PI3K catalytic subunit or reduced levels of phosphatidylinositol-3,4,5-triphosphate 3-phosphatase. The third group involves the concept of “scape”. This involves the use of other pathways that can preexist or be acquired at the time of resistance. An example is the cross talk between the estrogen receptor and the HER2 pathways.
Other mechanisms of resistance to anti-HER2 therapies are being studied and include regulators of cell cycle and apoptosis, the tumor microenvironment and immune system, mucins and various receptors and tyrosine kinases.Citation17,Citation18
However, combinations of targeted agents represent a way to reduce resistance to trastuzumab.
In recent years, we have observed that dual blockade of the HER2 pathway produces better results with an increase in pathological complete response (pCR) rates. In the Neosphere trial, pertuzumab plus trastuzumab and chemotherapy demonstrated higher pCR rates compared with the trastuzumab or pertuzumab arms, and these pCR rates were associated with better DFS.Citation19–Citation24
Lapatinib was studied in this context in combination with trastuzumab and chemotherapy in several clinical trials. Although some trials of the combination showed higher pCR rates, they did not demonstrate statistically significant long-term benefits. In addition, lapatinib was associated with a worse toxicity profile.Citation25–Citation29
It is known that the response to treatment depends on clinical factors such as lower stage at diagnosis (small tumors and few or no metastatic axillary nodes) and biological factors such as negative hormone receptor (HR) status, high ki-67 percentage and grade II–III tumors.Citation30–Citation32
The association between clinical and pathological response has been studied in the neoadjuvant setting. The best imaging methods, such as ultrasound or magnetic resonance imaging (MRI), to monitor tumor shrinkage during treatmentCitation33–Citation38 and their correlation with pathological response have also been analyzed.Citation39
pCR has been associated with long-term benefits, DFS and OS, in several clinical trials and meta-analyses,Citation7,Citation40 but controversies exist on this point. Some authors consider pCR a surrogate end point that has yet to demonstrate a real long-term benefit.Citation41,Citation42
The sequential administration of a taxane and an anthracycline plus trastuzumab provides a significant benefit to patients with a high pCR rate.Citation43 The cardiotoxicity associated with this anthracycline plus trastuzumab combination is a drawback,Citation44–Citation46 but formulations such as epirubicin or liposomal anthracyclines have reduced this negative impact on heart function.Citation47,Citation48 Although it is still important to monitor left ventricular ejection fraction (LVEF) during treatment, improved knowledge of long-term cardiotoxicity years after the end of treatment is needed.Citation21
We analyzed the efficacy data, cardiotoxicity and long-term follow-up in 142 patients with HER2-positive breast cancer tumors diagnosed from 2005 to 2016 at a single institution who were treated homogenously through sequential administration of a taxane and an anthracycline plus trastuzumab.
Patients and methods
This is a retrospective observational study that included 142 patients with stage I–III HER2-positive breast cancer tumors diagnosed from 2005 to 2016 at a single institution.
The study, as well as all procedures performed in the study, was approved by the ethics committee of the Galician region (Comité Autonómico de Ética de Investigación de Galicia, Spain) in 2015 with the code: SAN-TRA-2015-01, and all the patients who were alive at the time the retrospective study was started provided written informed consent for participation.
The study procedures were carried out in accordance with the 1975 Declaration of Helsinki, as revised in 2000, and Good Clinical Practice guidelines.
Patients
All patients included were ≥18 years of age and had Eastern Cooperative Oncology Group Performance Status of ≤1.
Histological type, tumor grade, ki-67 index, estrogen and progesterone receptor and HER2 status were determined locally using pretreatment core biopsies.
HER2-positive breast cancer was considered if the tumors exhibited threefold overexpression of the HER2 receptor as assessed using immunohistochemical techniques (Herceptest™ Dako until 2013 and Roche until 2016) or a threefold overexpression of the HER2 gene as assessed using fluorescent (until 2013) or silver (2013–2016) in situ hybridization (in the same laboratory) in accordance with 2007 and 2013 American Society of Clinical Oncology/College of American Pathologist guidelines.Citation49,Citation50
An ultrasound-guided fine-needle puncture aspiration was also performed on suspected malignant axillary lymph nodes at diagnosis.
The tumor site was marked using a stainless steel marker placed using ultrasound guidance in the majority of patients. Multicentric or inflammatory tumors were not marked because of the indication for mastectomy despite the response to treatment.
From 2005 to 2012, a sentinel lymph node biopsy (SLNB) was performed prior to treatment in patients with clinically negative axilla, and from 2012 to 2016 this procedure was performed after treatment.Citation51,Citation52
At diagnosis, patients were free from cardiovascular disease and demonstrated adequate cardiac function with an LVEF >50%, as measured using echocardiography.
This echocardiography was repeated after three to four cycles, at the end of chemotherapy and during the follow-up period, at least 6 months after the end of adjuvant trastuzumab.
Patients also had adequate hematological, renal and hepatic function.
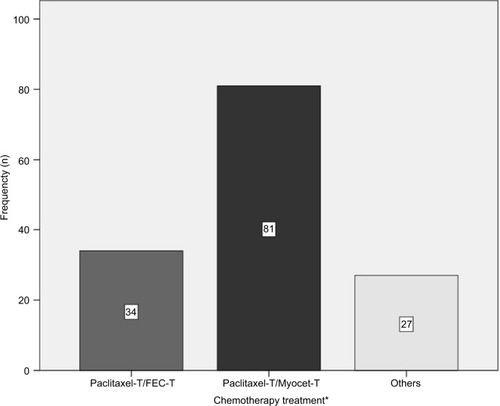
The different treatment regimens administered are shown in . The vast majority of patients received a total of eight cycles consisting of a sequence of anthracyclines and taxanes with trastuzumab.
Figure 1 Types of chemotherapy.
Abbreviations: FEC, 5-fluorouracil–epirubicin–cyclophosphamide; IV, intravenous; T, trastuzumab.
Physical examinations were performed every 3 weeks during chemotherapy treatment. Mammograms, ultrasound and MRI were performed before and after neoadjuvant treatment.
The clinical response (CR) was measured through physical examination and MRI in accordance with the response evaluation criteria in solid tumors.Citation53,Citation54
Patients underwent surgery between 3 and 5 weeks from the end of chemotherapy. There was an increase in conservative breast surgery in patients that were candidates for mastectomy at diagnosis.
pCR was defined as the total absence of invasive tumor in both breast and axillary nodes (ypT0/is ypN0).
The pathological response was measured in accordance with the Miller and Payne system in most patients.Citation55
We used the revised American Joint Committee on Cancer TNM system in patients included in the first 2 years of the study.Citation56
This pathological response was correlated with clinical and biological factors (tumor size, axillary nodes, HR status and ki-67 index).
All patients treated using conservative breast surgery received whole-breast irradiation at a standard dose. Some patients with T3 tumors at diagnosis were also irradiated after mastectomy. Regional nodal irradiation of the supraclavicular fossa-axillary apex was used in patients with clinical stage III disease, in patients with four or more positive lymph nodes and in selected patients with one to three positive lymph nodes.Citation57,Citation58
After completion of systemic and local therapy, patients with HR-positive tumors received tamoxifen or an aromatase inhibitor if the patient was menopausal.
We report DFS (defined as the time from surgery to the first documented disease progression) and OS (time from surgery to death).
Statistical analyses
We performed a descriptive analysis for all variables. Continuous variables were reported using the median as the central value and the SD. For dichotomous or categorical variables, absolute numbers and percentages were computed. The chi-squared two-tailed test was used for comparative analyses between categorical variables.
We estimated DFS and OS rates for each group using the Kaplan–Meier method. A comparison of survival curves was performed using the log-rank test. Differences in survival between groups were compared using the Cox regression test. All statistical tests were two sided, and a significance level of 0.05 was applied.
Results
A total of 142 patients with stage I–III HER2-positive breast cancer tumors, who were candidates for primary systemic therapy and were diagnosed from 2005 to 2016 at our institution, were included in the study. The pretreatment characteristics of the patients are listed in .
Table 1 Patient characteristics
The median patient age was 49 (30–79) years, with stage I, II or III breast cancer at diagnosis in 4%, 69% and 27%, respectively. A total of 10% of patients had inflammatory breast cancer, 43% of tumors were HR negative and 62% were grade III, while 90% of tumors were confirmed to be HER2 positive through immunohistochemistry (threefold increase in protein expression) and the rest through fluorescent in situ hybridization or silver in situ hybridization. A total of 84% of patients had an indication for mastectomy at diagnosis.
A total of 98% of patients completed the planned cycles of chemotherapy. The remaining 2% (three patients) could not complete chemotherapy because of hepatotoxicity in one patient, sustained grade III asthenia and grade II neutropenia in an elderly patient and a massive pulmonary thromboembolism in the third patient after four cycles of chemotherapy.
Clinical response
The CR was assessed through mammography, ultrasound imaging and MRI before and after systemic therapy and immediately before surgery in most patients.
In the first years of the study, breast MRI was not a routine imaging technique, so it was not performed in the first 18 patients.
The CR data are listed in .
Table 2 Complete response
A total of 49% and 63% of patients demonstrated a clinical complete response, as measured through ultrasound and MRI, respectively.
Pathological response
The pCR rate, defined as the total absence of invasive tumor in both breast and axillary nodes (ypT0/is ypN0), was 52%. The pCR rates in breast, breast and axilla and in different subgroups are summarized in and .
Table 3 pCR in breast and breast/axilla
Table 4 pCR by subgroups
The pCR rate was higher in ductal (53%) than in lobular (14%) cancer, in HR-negative (64%) than in HR-positive (41%) cancer, in grade III (53%) than in grade I–II (45%) cancer and in tumors with a ki-67 value >20 (85%) than in those with a ki-67 value <20 (15%).
A total of 61% and 68% of patients with CR, as assessed using ultrasound and MRI, respectively, also demonstrated pCR.
pCR was higher in patients treated using paclitaxel/trastuzumab (PT) followed by Myocet/cyclophosphamide/trastuzumab (54%) than those treated using PT followed by fluorouracil/epirubicin/cyclophosphamide/trastuzumab (38%), but the numbers of patients assigned to each group were too low to result in statistically significant differences.
Surgery
At diagnosis, 84% of patients were indicated for mastectomy. After treatment, tumorectomy was performed in 66%.
Axillary dissection, SLNB and no axillary procedure were performed in 69%, 22% and 9% of patients, respectively. Those patients with a negative SLNB before treatment did not undergo an axillary procedure.
Long-term efficacy data
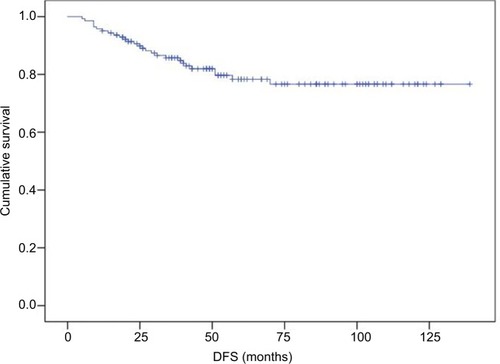
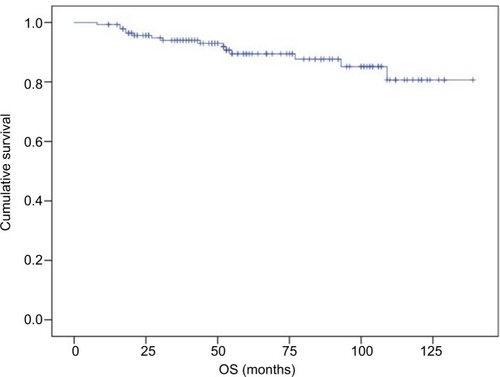
At the time of the analysis in March 2018, and with a median follow-up of 55 months, 27 patients suffered from breast cancer recurrence and 15 patients died as a result of any cause.
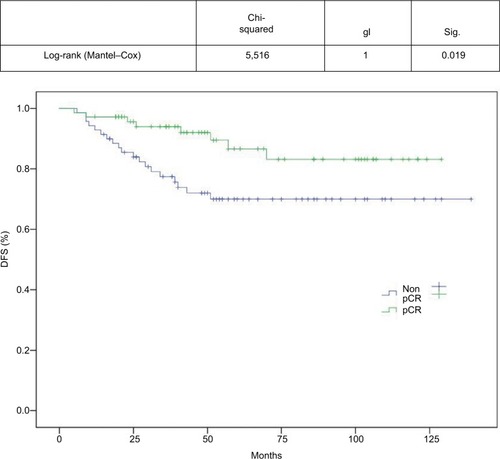
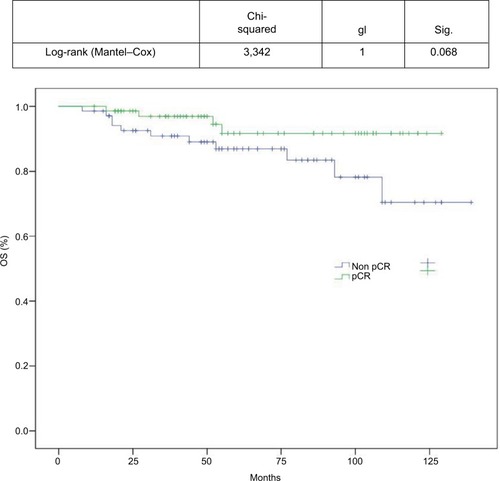
DFS and OS curves are shown in and , respectively. We were able to demonstrate an association between pCR and better DFS with statistical significance (P=0.19) and a trend toward improved OS (P=0.068; and ).
Cardiotoxicity
Because of the potential cardiotoxicity associated with anthracyclines and trastuzumab, knowledge of cardiovascular risk factors and patient monitoring were very important during the study.
Age at diagnosis, body mass index (BMI), arterial hypertension, dyslipidemia, preexisting cardiovascular disease and previous radiotherapy are recognized as risk factors for heart disease in breast cancer patients treated using anthracyclines and trastuzumab.Citation59–Citation65
The risk factors for developing a cardiac event in the current cohort of patients are listed in .
Table 5 Risk factors for cardiac events
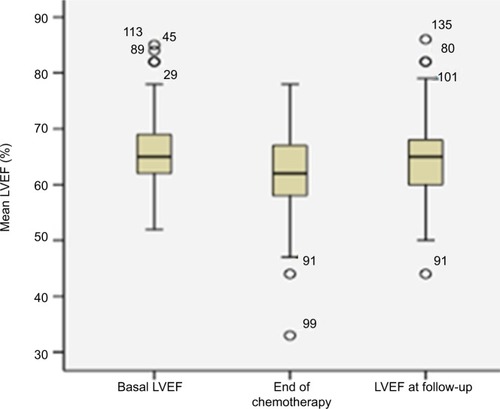
At diagnosis, all patients had adequate cardiac function with an LVEF of >50%, as measured using echocardiography.
The echocardiography was repeated after chemotherapy treatment and during follow-up at least 6–12 months after the end of adjuvant trastuzumab in 70% of patients.
At the end of chemotherapy treatment, five patients suffered a decline in LVEF <50% (3.5% of patients). The cardiovascular risk factors for these patients are listed in .
Table 6 Cardiovascular risk factors in patients who suffered a decline in LVEF <50%
Of these patients, four did not experience cardiac symptoms and demonstrated a rapid recovery after temporarily stopping trastuzumab. One did not receive adjuvant trastuzumab. Three received left breast radiotherapy and two had a history of high blood pressure.
The fifth patient who suffered a decline in LVEF below 50% did not receive adjuvant trastuzumab and developed severe ventricular dysfunction over subsequent years.
This patient had a high BMI and received left breast radiotherapy, which are risk factors for heart disease.
LVEF at baseline, end of chemotherapy and beyond 6 months from the end of trastuzumab treatment is shown in .
Discussion
Although clear clinical and preclinical evidence for dual blockade of the HER2 pathway in neoadjuvant treatment of HER2-positive breast cancer has been reported and this has become a standard approach in combination with chemotherapy, some issues remain controversial. Do all patients benefit from the addition of the second biological inhibitor to their chemotherapy regimen, and what is the role of anthracyclines and cardiotoxicity?
Considering that we retrospectively analyzed a homogeneous population treated using a taxane and anthracycline concomitantly with trastuzumab and all treatment was administered before surgery, the median pCR (total absence of invasive tumor in both breast and axillary nodes) achieved in our cohort was high (52%) and is comparable to other treatment series employing a similar sequence. In a previous study, Buzdar et alCitation11 used sequential 5-fluorouracil–epirubicin–cyclophosphamide (FEC) and paclitaxel with trastuzumab and achieved a higher pCR (56%), but only evaluated pCR in the breast.
The median pCR achieved in studies employing dual blockade of the HER2 pathway ranges from 45% to 62%. In the most important of these studies (Neosphere and Try-phaena), only pCR in the breast was evaluated.
It is known that the combination of anthracyclines and trastuzumab is very active, but it can increase cardiotoxicity. There are anthracyclines such as epirubicin or liposomal formulations that can reduce this toxicity.
A Phase III trial reported that adding trastuzumab to epirubicin does not improve efficacy in terms of pCR in a sequence with taxane and trastuzumab vs epirubicin alone using the same sequence. However, long-term efficacy and cardiotoxicity data are needed to draw robust conclusions.Citation10
In the current study, 37 patients were treated with epirubicin and 85 with liposomal doxorubicin (Myocet®; Teva B.V, Haarlem, the Netherlands), achieving a higher pCR rate in the Myocet group (38% and 51%, respectively). The number of patients assigned to each group was too low to produce statistically significant differences.
These rates are comparable to those in other published series using FEC or classic anthracyclines. There is little evidence to support the use of liposomal anthracyclines in the neoadjuvant setting. Most published series uses concomitant administration of chemotherapeutics instead of sequential administration and a higher number of cycles.Citation48,Citation66–Citation68
We identified three biological factors associated with a better response to therapy: HR-negative status, ki-67 >20% and grade III breast cancer.
The high pCR achieved in the HR-negative patients vs the HR-positive patients was also seen in previous clinical trials in the HER2 neoadjuvant setting,Citation19,Citation26,Citation69 and this could be explained by the cross talk between the HER2 and endocrine pathway conferring resistance to agents targeting either pathway.
Many studies have demonstrated the prognostic and predictive value of ki-67, whereby it is used to identify patients who will benefit most from systemic therapy. These studies used different methods for scoring this proliferation antigen and included a heterogeneous population with different biological subtypes.Citation70,Citation71
A 20% cutoff point for the ki-67 index appears to be a statistically significant prognostic factor in luminal B HER2-negative tumors. The median value of ki-67 is usually higher in triple-negative and HER2-positive tumors. Median ki-67 values of 35%–40% in HER2-positive and 50% in triple-negative breast cancer have been previously reported.Citation72
In the current study, we examined three different cutoff points for the ki-67 index and their relationship with pCR, DFS and OS.
Tumors with ki-67 >20% (especially those >35%) achieved higher pCR rates, but no group was correlated with long-term benefits.
The correlation between histological grade and prognosis has been confirmed in multiple reports and currently helps clinicians to make treatment decisions.Citation73–Citation78 Its value as an independent factor to predict the response to NT is unclear.Citation30,Citation31 In the current series, grade III tumors achieved a higher pCR rate compared with grade I–II tumors.
There was a high rate of indication for mastectomy at diagnosis in the current series. This could be explained by the incidence of T3 and T4 tumors and the relationship between breast/tumor size in T2 tumors.
After systemic treatment, the rates of breast conservation were also high and greater than in other previously reported series.Citation79–Citation81 We attributed this to the high pCR achieved in this selected HER2 population.
The potential for an increase in local recurrence after NT followed by breast conservation was analyzed in several studies and meta-analyses.Citation5,Citation82 The MD Anderson Prognostic Index concludes that the local recurrence rate could depend on other biological factors such as clinical N2 or N3 disease, lymphovascular space invasion, a multifocal pattern of residual disease and a residual pathological primary tumor >2 cm, rather than the timing of chemotherapy delivery.Citation83,Citation84
Our locoregional recurrence rate of 3.9% is low at the time of data cutoff.
A high percentage (61%–68%) of tumors that achieved CR, assessed using ultrasound and MRI, also demonstrated pCR. MRI has been reported as superior to ultrasound and mammography in the assessment of tumor extent and is highly sensitive in identifying residual disease following neoadjuvant treatment.Citation85,Citation86 We perceived a slight superiority of MRI in the current series.
pCR in HER2-positive breast cancer is associated with a substantially longer time to recurrence and death, as reported in a meta-analysis published by Broglio et al,Citation40 which included a total of 5,500 HER2-positive breast cancer patients.
We demonstrated an association between pCR and improved DFS (P=0.19) in the current patients and a trend toward improved OS (P=0.068). This absence of a statistically significant benefit in OS could be explained by the low number of deaths at the time of analyses and to improvements in treatments for metastatic disease. None of the patients in the current series received pertuzumab in the neoadjuvant setting.
Concomitant administration of trastuzumab and classic anthracyclines is cardiotoxic and not permitted in clinical use.
In recent years, the incidence of congestive heart failure associated with trastuzumab and anthracyclines has become very low, since close monitoring of cardiac function and liposomal and less cardiotoxic anthracyclines have been used.Citation11,Citation43,Citation48
We used two-dimensional echocardiography to monitor trastuzumab-related cardiotoxicity following the current clinical guidelines. This is a readily available cardiac imaging modality with low cost and is radiation free. However, the current knowledge indicates that this technique does not accurately predict the development of cardiotoxicity. New imaging modalities can provide more information in the early stages and can quantify myocardial deformation as a marker of contractility. These new echocardiographic techniques, such as global longitudinal strain, can provide information that is predictive of cardiac dysfunction.Citation87,Citation88
Cardiac biomarkers, such as troponins and amino-terminal fragment of brain natriuretic peptide, are also a useful tool to monitor cardiotoxicity. They have been studied as early predictors of cardiac damage in patients receiving anthracyclines and anti-HER2 therapy with disparate results.Citation89,Citation90
After a long follow-up period, our data showed a low incidence of cardiac events: 2.95% of patients demonstrated an asymptomatic decrease in LVEF and 0.64% demonstrated symptomatic ventricular dysfunction. Both cardiac events were related to other cardiovascular risk factors such as high BMI or high blood pressure and left breast radiotherapy.
This low incidence of cardiac events did not allow us to achieve statistically significant differences between the cardiotoxicity profiles of the chemotherapy treatments. The five patients who suffered a decline in LVEF <50% at the end of chemotherapy treatment had received concomitant anthracyclines and trastuzumab instead of the sequential treatments.
Although the incidence of cardiotoxicity was low, it is desirable to avoid it through better selection of patients and the correction of cardiovascular risk factors (high BMI, arterial hypertension, dyslipidemia) from the beginning, not only through diet and exercise but also using medication. The early administration of angiotensin–convertin–enzyme inhibitors (enalapril type) or beta-blockers (carvedilol or bisoprolol type) or a combination of both has produced improvements in cardiac function in breast cancer patients treated using anthracyclines. This highlights the need for collaboration between cardiologists and oncologists from the beginning of treatment.Citation91–Citation93
Conclusion
The sequence of a taxane and a less cardiotoxic anthracycline concomitant to trastuzumab was effective in the HER2 neoadjuvant setting with high pCR and conservative breast surgery rates. Grade III and HR-negative tumors demonstrated the greatest benefit.
We observed a good correlation between the clinical complete response, as measured using ultrasound and MRI, and pCR.
We were able to associate pCR with improved DFS. Although there was a trend toward improved OS, it was not statistically significant.
The treatment was safe with an excellent long-term cardiotoxicity profile.
Acknowledgments
We would like to thank all the patients who participated in the study and the following people: Javier Prato, Aurea Molina, Joaquin Mosquera, Cristina Reboredo, Begoña Graña, María Quindós (Medical Oncology Department, A Coruña University Hospital), Manuel Juaneda (Surgery Department, Breast Unit, A Coruña University Hospital), Alberto Bouzón, Carmen Conde (Gynecology Department, Breast Unit, A Coruña University Hospital), Carlota Diaz (Gynecology Department, Breast Unit, A Coruña University Hospital), Carmen Cereijo (oncology nurse, Breast Unit, A Coruña University Hospital). This study did not receive any specific grants from funding agencies in the public, commercial or nonprofit sectors.
Disclosure
The authors report no conflicts of interest in this work.
References
- HortobagyiGNBlumenscheinGRSpanosWMultimodal treatment of locoregionally advanced breast cancerCancer19835157637686687377
- HortobagyiGNAmesFCBuzdarAUManagement of stage III primary breast cancer with primary chemotherapy, surgery, and radiation therapyCancer19886212250725163056604
- BearHDIndications for neoadjuvant chemotherapy for breast cancerSemin Oncol1998252 Suppl 3312
- RastogiPAndersonSJBearHDPreoperative chemotherapy: updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27J Clin Oncol200826577878518258986
- MauriDPavlidisNIoannidisJPNeoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysisJ Natl Cancer Inst200597318819415687361
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG)Long-term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: meta-analysis of individual patient data from ten randomised trialsLancet Oncol2018191273929242041
- CortazarPZhangLUntchMPathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysisLancet2014384993816417224529560
- BurrisHYardleyDJonesSPhase II trial of trastuzumab followed by weekly paclitaxel/carboplatin as first-line treatment for patients with metastatic breast cancerJ Clin Oncol20042291621162915117984
- BuzdarAUIbrahimNKFrancisDSignificantly higher pathologic complete remission rate after neoadjuvant therapy with trastuzumab, paclitaxel, and epirubicin chemotherapy: results of a randomized trial in human epidermal growth factor receptor 2-positive operable breast cancerJ Clin Oncol200523163676368515738535
- BuzdarAUValeroVIbrahimNKNeoadjuvant therapy with paclitaxel followed by 5-fluorouracil, epirubicin, and cyclophosphamide chemotherapy and concurrent trastuzumab in human epidermal growth factor receptor 2-positive operable breast cancer: an update of the initial randomized study population and data of additional patients treated with the same regimenClin Cancer Res200713122823317200359
- BuzdarAUSumanVJMeric-BernstamFAmerican College of Surgeons Oncology Group investigatorsFluorouracil, epirubicin, and cyclophosphamide (FEC-75) followed by paclitaxel plus trastuzumab versus paclitaxel plus trastuzumab followed by FEC-75 plus trastu-zumab as neoadjuvant treatment for patients with HER2-positive breast cancer (Z1041): a randomised, controlled, phase 3 trialLancet Oncol201314131317132524239210
- UntchMRezaiMLoiblSNeoadjuvant treatment with trastu-zumab in HER2-positive breast cancer: results from the GeparQuattro studyJ Clin Oncol201028122024203120308670
- UntchMFaschingPAKonecnyGEPathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: results from the TECHNO trial of the AGO and GBG study groupsJ Clin Oncol201129253351335721788566
- YardenYBiology of HER2 and its importance in breast cancerOncology200161Suppl 2113
- GhoshRNarasannaAWangSETrastuzumab has preferential activity against breast cancers driven by HER2 homodimersCancer Res20117151871188221324925
- RimawiMFSchiffROsborneCKTargeting HER2 for the treatment of breast cancerAnnu Rev Med20156611112825587647
- RexerBNArteagaCLIntrinsic and acquired resistance to HER2-targeted therapies in HER2 gene-amplified breast cancer: mechanisms and clinical implicationsCrit Rev Oncog201217111622471661
- RimawiMFde AngelisCSchiffRResistance to anti-HER2 therapies in breast cancerAm Soc Clin Oncol Educ Book201535e157e164
- GianniLPienkowskiTImYHEfficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trialLancet Oncol2012131253222153890
- GianniLPienkowskiTImYH5-Year analysis of neoadjuvant pertuzumab and trastuzumab in patients with locally advanced, inflammatory, or early-stage HER2-positive breast cancer (NeoSphere): a multicentre, open-label, phase 2 randomised trialLancet Oncol201617679180027179402
- SchneeweissAChiaSHickishTPertuzumab plus trastuzumab in combination with standard neoadjuvant anthracycline-containing and anthracycline-free chemotherapy regimens in patients with HER2-positive early breast cancer: a randomized phase II cardiac safety study (TRYPHAENA)Ann Oncol20132492278228423704196
- UntchMJackischCSchneeweissAGerman Breast Group (GBG); Arbeitsgemeinschaft Gynäkologische Onkologie—Breast (AGO-B) InvestigatorsNab-paclitaxel versus solvent-based paclitaxel in neoadjuvant chemotherapy for early breast cancer (GeparSepto-GBG 69): a randomised, phase 3 trialLancet Oncol201617334535626869049
- HurvitzSASymmansWFJungKHHuangC-SThompsonAMPathologic complete response (pCR) rates after neoadjuvant trastuzumab emtansine (T-DM1 [K]) + pertuzumab (P) vs docetaxel + carboplatin + trastuzumab + P (TCHP) treatment in patients with HER2-positive (HER2+) early breast cancer (EBC) (KRISTINE)J Clin Oncol2016500500
- Ulrike NitzOGChristgenMGrischkeE-MAugustinDKümmelSFinal analysis of WSG-ADAPT HER2+/HR− trial: efficacy, safety, and predictive markers for 12-weeks of neoadjuvant dual blockade with trastuzumab + pertuzumab ± weekly paclitaxel in HER2+/HR− early breast cancer (EBC)J Clin Oncol2016518518
- BaselgaJBradburyIEidtmannHLapatinib with trastuzumab for HER2-positive early breast cancer (NeoALTTO): a randomised, open-label, multicentre, phase 3 trialLancet2012379981663364022257673
- de AzambujaEHolmesAPPiccart-GebhartMLapatinib with trastuzumab for HER2-positive early breast cancer (NeoALTTO): survival outcomes of a randomised, open-label, multicentre, phase 3 trial and their association with pathological complete responseLancet Oncol201415101137114625130998
- GuarneriVFrassoldatiABottiniAPreoperative chemotherapy plus trastuzumab, lapatinib, or both in human epidermal growth factor receptor 2-positive operable breast cancer: results of the randomized phase II CHER-LOB studyJ Clin Oncol201230161989199522493419
- UntchMLoiblSBischoffJGerman Breast Group (GBG); Arbeitsgemeinschaft Gynäkologische Onkologie-Breast (AGO-B) Study GroupLapatinib versus trastuzumab in combination with neoadjuvant anthracycline-taxane-based chemotherapy (GeparQuinto, GBG 44): a randomised phase 3 trialLancet Oncol201213213514422257523
- CareyLAClinical and translational results of CALGB 40601: A neo-adjuvant phase III trial of weekly paclitaxel and trastuzumab with or without lapatinib for HER2-positive breast cancerJ Clin Oncol2013500500
- EllisPSmithIAshleySClinical prognostic and predictive factors for primary chemotherapy in operable breast cancerJ Clin Oncol19981611071149440730
- ChangJPowlesTJAllredDCBiologic markers as predictors of clinical outcome from systemic therapy for primary operable breast cancerJ Clin Oncol199917103058306310506600
- PusztaiLGianniLTechnology insight: emerging techniques to predict response to preoperative chemotherapy in breast cancerNat Clin Pract Oncol200411445016264799
- VinnicombeSJMacVicarADGuyRLPrimary breast cancer: mammographic changes after neoadjuvant chemotherapy, with pathologic correlationRadiology199619823333408596827
- HerradaJIyerRBAtkinsonENSneigeNBuzdarAUHortobagyiGNRelative value of physical examination, mammography, and breast sonography in evaluating the size of the primary tumor and regional lymph node metastases in women receiving neoadjuvant chemotherapy for locally advanced breast carcinomaClin Cancer Res199739156515699815844
- MartincichLMontemurroFde RosaGMonitoring response to primary chemotherapy in breast cancer using dynamic contrast-enhanced magnetic resonance imagingBreast Cancer Res Treat2004831677614997056
- ChenJHBahriSMehtaRSBreast cancer: evaluation of response to neoadjuvant chemotherapy with 3.0-T MR imagingRadiology2011261373574321878615
- ChenJHSuMYClinical application of magnetic resonance imaging in management of breast cancer patients receiving neoadjuvant chemotherapyBiomed Res Int20132013234816723862143
- WoolfDKPadhaniARMakrisAMagnetic resonance imaging, digital mammography, and sonography: tumor characteristics and tumor biology in primary settingJ Natl Cancer Inst Monogr2015201551152026063879
- ChenJHFeigBAgrawalGMRI evaluation of pathologically complete response and residual tumors in breast cancer after neoadjuvant chemotherapyCancer20081121172618000804
- BroglioKRQuintanaMFosterMAssociation of pathologic complete response to neoadjuvant therapy in HER2-positive breast cancer with long-term outcomes: a meta-analysisJAMA Oncol201626E1E10
- von MinckwitzGFontanellaCComprehensive review on the surrogate endpoints of efficacy proposed or hypothesized in the scientific community todayJ Natl Cancer Inst Monogr2015201551293126063882
- Wang-LopezQChalabiNAbrialCCan pathologic complete response (pCR) be used as a surrogate marker of survival after neoadjuvant therapy for breast cancer?Crit Rev Oncol Hematol20159518810425900915
- GianniLEiermannWSemiglazovVNeoadjuvant chemotherapy with trastuzumab followed by adjuvant trastuzumab versus neoadjuvant chemotherapy alone, in patients with HER2-positive locally advanced breast cancer (the NOAH trial): a randomised controlled superiority trial with a parallel HER2-negative cohortLancet2010375971237738420113825
- SlamonDJLeyland-JonesBShakSUse of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2N Engl J Med20013441178379211248153
- Tan-ChiuEYothersGRomondEAssessment of cardiac dysfunction in a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel, with or without trastuzumab as adjuvant therapy in node-positive, human epidermal growth factor receptor 2-overexpressing breast cancer: NSABP B-31J Clin Oncol200523317811781916258083
- PerezEASumanVJDavidsonNECardiac safety analysis of doxorubicin and cyclophosphamide followed by paclitaxel with or without trastuzumab in the North Central Cancer Treatment Group N9831 adjuvant breast cancer trialJ Clin Oncol20082681231123818250349
- BatistGBartonJChaikinPSwensonCWellesLMyocet (liposome-encapsulated doxorubicin citrate): a new approach in breast cancer therapyExpert Opin Pharmacother20023121739175112472371
- GaviláJGuerreroÁClimentMÁEfficacy and safety of neoadjuvant chemotherapy with concurrent liposomal-encapsulated doxorubicin, paclitaxel and trastuzumab for human epidermal growth factor receptor 2-positive breast cancer in clinical practiceInt J Clin Oncol201520348048925011497
- WolffACHammondMESchwartzJNAmerican Society of Clinical Oncology; College of American PathologistsAmerican Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancerJ Clin Oncol200725111814517159189
- WolffACHammondMEHicksDGAmerican Society of Clinical Oncology; College of American PathologistsRecommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline updateArch Pathol Lab Med2014138224125624099077
- KuehnTBauerfeindIFehmTSentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort studyLancet Oncol201314760961823683750
- BougheyJCSumanVJMittendorfEAAlliance for Clinical Trials in OncologySentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the American College of Surgeons Oncology Group (ACOSOG) Z1071 clinical trialJAMA2013310141455146124101169
- TherassePArbuckSGEisenhauerEAGwyther new guidelines to evaluate the response to treatment in solid tumorsJ Natl Cancer Inst200092320521610655437
- EisenhauerEATherassePBogaertsJNew response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1)Eur J Cancer200945222824719097774
- OgstonKNMillerIDPayneSA new histological grading system to assess response of breast cancers to primary chemotherapy: prognostic significance and survivalBreast200312532032714659147
- CareyLAMetzgerRDeesECAmerican joint committee on cancer tumor-node-metastasis stage after neoadjuvant chemotherapy and breast cancer outcomeJ Natl Cancer Inst200597151137114216077072
- GargAKStromEAMcNeeseMDT3 disease at presentation or pathologic involvement of four or more lymph nodes predict for locoregional recurrence in stage II breast cancer treated with neoadjuvant chemotherapy and mastectomy without radiotherapyInt J Radiat Oncol Biol Phys200459113814515093909
- HuangEHTuckerSLStromEAPostmastectomy radiation improves local-regional control and survival for selected patients with locally advanced breast cancer treated with neoadjuvant chemotherapy and mastectomyJ Clin Oncol200422234691469915570071
- SwainSMWhaleyFSEwerMSCongestive heart failure in patients treated with doxorubicin: a retrospective analysis of three trialsCancer200397112869287912767102
- TsaiHTIsaacsCFuAZRisk of cardiovascular adverse events from trastuzumab (Herceptin(®)) in elderly persons with breast cancer: a population-based studyBreast Cancer Res Treat2014144116317024469642
- DranitsarisGRaysonDVincentMThe development of a predictive model to estimate cardiotoxic risk for patients with metastatic breast cancer receiving anthracyclinesBreast Cancer Res Treat2008107344345017978870
- FumoleauPRochéHKerbratPFrench Adjuvant Study GroupLong-term cardiac toxicity after adjuvant epirubicin-based chemotherapy in early breast cancer: French Adjuvant Study Group resultsAnn Oncol2006171859216251204
- SciarrettaSPalanoFTocciGBaldiniRVolpeMAntihypertensive treatment and development of heart failure in hypertension: a Bayesian network meta-analysis of studies in patients with hypertension and high cardiovascular riskArch Intern Med2011171538439421059964
- CarverJRShapiroCLNgAASCO Cancer Survivorship Expert PanelAmerican Society of Clinical Oncology clinical evidence review on the ongoing care of adult cancer survivors: cardiac and pulmonary late effectsJ Clin Oncol200725253991400817577017
- GroarkeJDNguyenPLNohriaAFerrariRChengSMoslehiJCardiovascular complications of radiation therapy for thoracic malignancies: the role for non-invasive imaging for detection of cardiovascular diseaseEur Heart J2014351061262323666251
- AntónARuizAPlazaolaAPhase II clinical trial of liposomal-encapsulated doxorubicin citrate and docetaxel, associated with trastuzumab, as neoadjuvant treatment in stages II and IIIA HER2-overexpressing breast cancer patients. GEICAM 2003-03 studyAnn Oncol2011221747920603435
- TuxenMKColdSTangeUBBalslevENielsenDLPhase II study of neoadjuvant pegylated liposomal doxorubicin and cyclophosphamide ± trastuzumab followed by docetaxel in locally advanced breast cancerActa Oncol201453101440144524991893
- Uriarte-PintoMEscolano-PueyoÁGimeno-BallesterVPascual-MartínezOAbad-SazatornilMRAgustín-FerrándezMJTrastuzumab, non-pegylated liposomal-encapsulated doxorubicin and paclitaxel in the neoadjuvant setting of HER-2 positive breast cancerInt J Clin Pharm2016362446453
- CareyLABerryDACirrincioneCTMolecular heterogeneity and response to neoadjuvant human epidermal growth factor receptor 2 targeting in CALGB 40601, a randomized phase III trial of paclitaxel plus trastuzumab with or without lapatinibJ Clin Oncol201634654254926527775
- YerushalmiRWoodsRRavdinPMHayesMMGelmonKAKi67 in breast cancer: prognostic and predictive potentialLancet Oncol201011217418320152769
- DowsettMNielsenTOA’HernRInternational Ki-67 in Breast Cancer Working GroupAssessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working groupJ Natl Cancer Inst2011103221656166421960707
- TashimaRNishimuraROsakoTEvaluation of an optimal cutoff point for the Ki-67 index as a prognostic factor in primary breast cancer: a retrospective studyPLoS One2015157e0119565
- WolffBHistological grading in carcinoma of the breastBr J Cancer196620136405937523
- FisherERSassRFisherBPathologic findings from the National Surgical Adjuvant Project for Breast Cancers (protocol no. 4). X. Discriminants for tenth year treatment failureCancer1984533 Suppl7127236692274
- ContessoGMouriesseHFriedmanSGeninJSarrazinDRouesseJThe importance of histologic grade in long-term prognosis of breast cancer: a study of 1,010 patients, uniformly treated at the Institut Gustave-RoussyJ Clin Oncol198759137813863625256
- ElstonCWEllisIOPathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-upHistopathology19911954034101757079
- GaleaMHBlameyRWElstonCEEllisIOThe Nottingham Prognostic Index in primary breast cancerBreast Cancer Res Treat19922232072191391987
- DabbsDJDuctal carcinoma of breast: nuclear grade as a predictor of S-phase fractionHum Pathol19932466526568389317
- FisherBBrownAMamounasEEffect of preoperative chemotherapy on local-regional disease in women with operable breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-18J Clin Oncol1997157248324939215816
- WolmarkNWangJMamounasEBryantJFisherBPreoperative chemotherapy in patients with operable breast cancer: nine-year results from National Surgical Adjuvant Breast and Bowel Project B-18J Natl Cancer Inst Monogr200120013096102
- van der HageJAvan de VeldeCJJulienJPTubiana-HulinMVanderveldenCDuchateauLPreoperative chemotherapy in primary operable breast cancer: results from the European Organization for Research and Treatment of Cancer trial 10902J Clin Oncol200119224224423711709566
- MieogJSvan der HageJAvan de VeldeCJNeoadjuvant chemotherapy for operable breast cancerBr J Surg200794101189120017701939
- ChenAMMeric-BernstamFHuntKKBreast conservation after neoadjuvant chemotherapy: the MD Anderson cancer center experienceJ Clin Oncol200422122303231215197191
- ChenAMMeric-BernstamFHuntKKBreast conservation after neoadjuvant chemotherapyCancer2005103468969515641036
- LobbesMBIPrevosRSmidtMThe role of magnetic resonance imaging in assessing residual disease and pathologic complete response in breast cancer patients receiving neoadjuvant chemotherapy: a systematic reviewInsights Imaging20134216317523359240
- MarinovichMLHoussamiNMacaskillPMeta-analysis of magnetic resonance imaging in detecting residual breast cancer after neoadjuvant therapyJ Natl Cancer Inst2013105532133323297042
- ThavendiranathanPPoulinFLimKDPlanaJCWooAMarwickTHUse of myocardial strain imaging by echocardiography for the early detection of cardiotoxicity in patients during and after cancer chemotherapy: a systematic reviewJ Am Coll Cardiol20146325 Pt A2751276824703918
- MehtaLSWatsonKEBaracAAmerican Heart Association Cardiovascular Disease in Women and Special Populations Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Quality of Care and Outcomes ResearchCardiovascular disease and breast cancer: where these entities intersect: a scientific statement from the american heart associationCirculation20181378e30e6629437116
- CardinaleDColomboATorrisiRTrastuzumab-induced cardiotoxicity: clinical and prognostic implications of troponin I evaluationJ Clin Oncol201028253910391620679614
- PondeNBradburyILambertiniMCardiac biomarkers for early detection and prediction of trastuzumab and/or lapatinib-induced cardiotoxicity in patients with HER2-positive early-stage breast cancer: a NeoALTTO sub-study (BIG 1-06)Breast Cancer Res Treat2018168363163829280043
- AmmonMArenjaNLeibundgutGCardiovascular management of cancer patients with chemotherapy-associated left ventricular systolic dysfunction in real-world clinical practiceJ Card Fail201319962963424054339
- OliveiraGHMukerjiSHernandezAVIncidence, predictors, and impact on survival of left ventricular systolic dysfunction and recovery in advanced cancer patientsAm J Cardiol2014113111893189824837270
- CuevaJFAntolínSCalvoLGalician consensus on management of cardiotoxicity in breast cancer: risk factors, prevention an early interventionClin Trasl Oncol201719910671078