Abstract
In this paper, we present the results of a reanalysis of the data of two large randomized, double-blind, parallel group studies with a similar design, comparing the efficacy of an angiotensin-receptor blocker (olmesartan medoxomil) with that of an angiotensin-converting enzyme inhibitor (ramipril), by applying two different blood pressure targets recently recommended by hypertension guidelines for all patients, irrespective of the presence of diabetes (<140/90 mmHg), and for elderly hypertensive patients (<150/90 mmHg). The efficacy of olmesartan was not negatively affected by age, sex, hypertension type, diabetes status or other concomitant clinical conditions, or cardiovascular risk factors. In most cases, olmesartan provided better blood pressure control than ramipril. Olmesartan was significantly more effective than ramipril in male patients, in younger patients (aged 65–69 years), in those with metabolic syndrome, obesity, dyslipidemia, preserved renal function, diastolic ± systolic hypertension, and, in general, in patients with a high or very high cardiovascular risk. Interestingly, patients previously untreated or treated with two or more antihypertensive drugs showed a significantly larger response with olmesartan than with ramipril. Thus, our results confirm the good efficacy of olmesartan in elderly hypertensives even when new blood pressure targets for antihypertensive treatment are considered. Such results may be relevant for the clinical practice, providing some hint on the possible different response of elderly hypertensive patients to two different drugs acting on the renin–angiotensin system, when patients are targeted according to the blood pressure levels recommended by recent hypertension guidelines.
Introduction
Until recently, major guidelines recommended two distinct blood pressure targets for treated hypertensives, namely <140/90 mmHg in low-moderate risk individuals and <130/80 mmHg in high-risk ones.Citation1,Citation2 According to these guidelines, the blood pressure goal in treated older patients had to be the same as in younger patients, namely <140/90 mmHg or below, if tolerated.Citation1,Citation2 However, such recommendations were not supported by incontrovertible trial evidence. As a matter of fact, in all the large randomized trials of antihypertensive treatment in the elderly, showing a reduction in cardiovascular events through lowering blood pressure, the average systolic and diastolic blood pressure levels attained with treatment were never <140/90 mmHg.Citation3,Citation4 Other trials of more vs less intensive blood pressure lowering were unable to demonstrate benefits, in either aged individuals or high-risk hypertensive patients, by lowering systolic blood pressure <140 mmHg.Citation5–Citation11 Additionally, the results of extensive reviews of randomized controlled trials showed that recommendation to lower blood pressure <130/80 mmHg in patients with diabetes or a history of cardiovascular or renal disease was not supported by any evidence.Citation3,Citation12–Citation14
Taken together, results of all these studies suggested that evidence-based recommendations could be a most appropriate and modern approach to hypertension treatment management. Accordingly, most recent guidelines now recommend that patients with arterial hypertension associated with diabetes or chronic kidney disease must be treated to attain the goal of systolic blood pressure <140 mmHg and diastolic blood pressure <90 mmHg. They also suggest that in older persons it may be sufficient to treat high blood pressure to a target of 150/90 mmHg or lower.Citation1,Citation2,Citation14–Citation17
Given these premises, the question arises as whether the current available antihypertensive armamentarium, and particularly monotherapies, may be suitable to achieve modern blood pressure targets in older individuals, regardless of the presence of associated clinical conditions or additional cardiovascular risk factors. The availability of a large database of elderly hypertensive patients enrolled in two randomized, double-blind, parallel group studies with a similar design, comparing the efficacy of an angiotensin-receptor blocker (ARB), olmesartan medoxomil, with that of the angiotensin-converting enzyme (ACE)-inhibitor, ramipril, gave us the possibility to explore such a scenario.Citation18,Citation19 The original studies were devised at the time when old recommendations were still valid, and thus blood pressure targets differed between nondiabetics (<140/90 mmHg) and diabetics (<130/80 mmHg). Therefore, in the present paper we reanalyzed the data and compared the results by applying two different blood pressure targets indicated by the new hypertension guidelines: <140/90 mmHg, irrespective of the presence of diabetes, and <150/90 mmHg, as recommended for older hypertensives.
Methodology
The details on the study design and population can be found in previous publications.Citation18–Citation20 Briefly, the two original studiesCitation18,Citation19 had a multicenter, randomized, double-blind, parallel group design, consisting of a 2-week washout with placebo, followed by 12 weeks of treatment with olmesartan medoxomil or ramipril at initial doses of 10 or 2.5 mg once daily, respectively. The initial drug dose could be doubled after the 2nd or 6th week of treatment in case of lack of normalization (systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg for nondiabetic, systolic blood pressure ≥130 mmHg or diastolic blood pressure ≥80 mmHg for diabetic patients). Elderly patients aged between 65 and 89 years, of either sex, with grade 1 or 2 essential hypertension (systolic blood pressure between 140 and 179 mmHg and diastolic blood pressure between 90 and 109 mmHg) were studied.
All patients gave their written informed consent before being enrolled in the study. The study was approved by the Ethics Committees of each study center. As in the original studies, analysis was performed on patients who were eligible for intention-to-treat, defined as all randomized patients receiving at least one dose of active treatment drug and having at least one office blood pressure measurement after randomization, using the last-observation-carried-forward method for patients prematurely leaving the study.
Response to antihypertensive treatment was evaluated by using a blood pressure target of <140/90 mmHg in all patients, irrespective of the presence of diabetes, and <150/90 mmHg, as recommended for older hypertensives. Analysis of variance was used to assess the differences between groups for continuous variables, whereas comparison of normalized patients was performed by the chi-squared test. Subgroup analyses for sex, age groups, level of cardiovascular risk, type of hypertension, metabolic status, renal functions status, number and type of previous antihypertensive drugs, and according to drug tolerability were also made. The level of statistical significance was kept at 0.05 throughout the whole study. Data are shown as means ± standard deviation (SD) or as numbers and percentages.
Results
Treatment efficacy according to new blood pressure targets in the whole study group
The pooled dataset from the two studies consisted of 1,426 patients (intention-to-treat population) of which 712 were treated with olmesartan at an average dose of 27.2±12.6 mg (47.1% of patients taking the full drug dosage) and 714 treated with ramipril at an average dose of 7.3±3.1 mg (55.3% of patients taking the full drug dosage, P=0.008 vs olmesartan).
As shown in , no statistically significant differences existed between the two treatment groups for the main demographic and clinical characteristics at baseline.
Table 1 Demographic and clinical characteristics of the 1,426 patients of the intention-to-treat population of the two studies pooled together
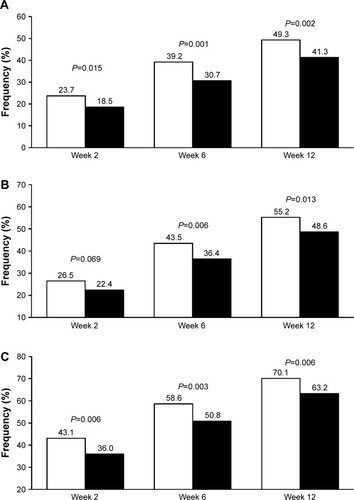
As expected, using the new cutoffs, the rate of normalization at 12 weeks (study end) increased as compared to the original reports, being still significantly larger under olmesartan than under ramipril (). When the blood pressure target was set at <140/90 mmHg for all patients, 55.2% of olmesartan-treated patients attained blood pressure normalization vs 48.6% of ramipril-treated patients (P=0.013). The use of a less rigid target (<150/90 mmHg), as indicated for the elderly, resulted in 70.1% rate of normalization under olmesartan and 63.2% under ramipril (P=0.006) at the end of the study. The superiority of olmesartan was observed at each study time point and the difference vs ramipril was statistically significant, except in one case.
Figure 1 Percentage of normalized patients according to different thresholds.
A summary of other studies assessing the efficacy of olmesartan monotherapy in elderly hypertensive patients is reported in . Although the studies performed so far are not entirely homogenous for design, inclusion criteria, study duration, and endpoints, some similarities with our results may be observed, at least for some studies. For instance, the application of new blood pressure targets to our data gave results superimposable to those observed in a similar study by Kereiakes et al which made use of olmesartan at dosages of 20 or 40 mg with blood pressure targets evaluated at <140/90 mmHg ().Citation21 Our responder rate was also not dissimilar from that observed in a study by Saito et al employing olmesartan monotherapy at dosages of 5–40 mg, with the addition of other antihypertensive drugs.Citation22 In the other three studies, the percentages were higher than in our studies, but one study enrolled patients with entry blood pressure levels higher than those of our population and evaluated only diastolic blood pressure response.Citation23 Another study was characterized by a very long follow-up.Citation24 A last study included only patients with systolic hypertension and the proportion of responders, larger than in our study, was estimated taking into account the systolic blood pressure only.Citation25
Table 2 Blood pressure response to olmesartan monotherapy in elderly patients with systolic and/or diastolic hypertension or isolated systolic hypertension in different open-label or double-blind randomized studies
Interestingly, in the present study, the proportion of patients achieving a blood pressure <140/90 mmHg was only marginally lower than that observed in a large cohort of 5,141 individuals aged 65–85 years receiving olmesartan 5–40 mg combined with a calcium channel blocker (amlodipine 2.5 or 5 mg, azelnidipine 8 or 16 mg) or a low-dose diuretic.Citation26 As a matter of fact, at the end of the 3.3 years of median follow-up, 68.0% of patients achieved the target blood pressure levels of <140/90 mmHg.
Blood pressure response according to sex, age, and absolute level of cardiovascular risk
In the original study report, blood pressure response was significantly better with olmesartan than with ramipril, in both men and women, as well as in younger individuals (65–69 years).Citation20 When the new therapeutic targets were applied, in the present analysis, the difference observed in favor of olmesartan was no more statistically significant for women (). Such differences as respect to the main study may be explained by the retrospective nature of the analysis, but we cannot exclude sex differences in response to olmesartan or ramipril, as postulated by recent studies in humans and animals.Citation27,Citation28 Concerning age, olmesartan was still significantly more effective than ramipril in patients younger than 70 years, whereas no superiority was observed in the other age categories, as in the original study ().
Table 3 Percentage of normalized and normalized or responder patients after 12 weeks of treatment with olmesartan medoxomil 10–40 mg (n=712) or ramipril 2.5–10 mg (n=714), according to sex, age and 10-year cardiovascular risk category (low-moderate: <5% and high-very high: ≥5%)
When patients were classified according to the 10-year absolute risk of fatal cardiovascular disease, according to the SCORE (Systematic COronary Risk Evaluation) algorithm,Citation29 a larger proportion of patients in the low-moderate (≤5%) risk category responded to either treatment, with a statistically significant superiority of olmesartan over ramipril in the high- or very high-risk category (>5%) (). Although application of the SCORE algorithm to elderly population may be not completely appropriate, given the fact that this population is at high risk for itself, such results may support the use of ARBs as an alternative to ACE inhibitors for the achievement of adequate blood pressure control with less intensive treatment in older hypertensive patients at higher risk of cardiovascular events. This is particularly relevant because there is consistent evidence that olmesartan may reduce cardiovascular risk by simultaneously normalizing blood pressure and reversing the proatherogenic effects of angiotensin II, an effect which is particularly desirable in the elderly.Citation30,Citation31
Blood pressure response according to type of hypertension
Olmesartan medoxomil also proved to be effective in controlling blood pressure, regardless of the type of hypertension. Most of the studied patients (75.5%) were affected by diastolic ± systolic hypertension. In these patients, the chance of attaining blood pressure normalization was significantly larger under olmesartan than under ramipril for both the thresholds considered (<140/90 mmHg: 54.4% vs 46.7%, P=0.012; <150/90 mmHg: 68.4% vs 60.3%, P=0.006), confirming the results of the original analysis. In three previous studies, which enrolled a total of 1,235 hypertensive patients aged ≥65 years, the proportion of patients with a blood pressure <140/90 mmHg at the end of the study who were given olmesartan at doses 5–40 mg was 56.0%, thus very close to our finding.Citation21,Citation22,Citation24
In our population, isolated systolic hypertension was less common than diastolic ± systolic hypertension (24.5%), but the chance of achieving blood pressure normalization with treatment was similar for the two hypertension subtypes. As in the original publication, no statistically significant differences were observed between the treatment groups in terms of blood pressure normalization either considering the 140/90 mmHg (olmesartan 58.0% vs ramipril 54.0%, P=0.451) or the 150/90 mmHg cutoff (75.9% vs 71.1%, P=0.311). Such results indicate that also in a relatively small subgroup of high-risk patients such as those with isolated systolic hypertension, olmesartan is capable of adequately controlling blood pressure.
Blood pressure response in patients with metabolic disorders
The metabolic syndrome is characterized by the association of different cardiovascular risk factors such as abdominal obesity, atherogenic dyslipidemia, insulin resistance or glucose intolerance, and blood pressure elevation.Citation32 Patients suffering from this condition have a higher risk of cardiovascular fatal and nonfatal events than healthy people, particularly in the presence of diabetes mellitus.Citation33,Citation34 Treatment with a drug acting on the renin–angiotensin system (RAS) has been shown to be particularly effective for controlling blood pressure and reducing major cardiovascular events, in the presence of metabolic abnormalities, such as metabolic syndrome or diabetes.Citation35–Citation37
In the pooled analysis of our studies, we analyzed the antihypertensive effect of olmesartan and ramipril in patients with metabolic syndromes, defined according to the International Diabetes Federation criteria and observed a significantly higher proportion of normalized patients under olmesartan, irrespective of the blood pressure target considered ().Citation38,Citation39 In addition to this finding, we observed a superior efficacy of olmesartan vs ramipril in patients with central or peripheral obesity (waist circumference >102 cm in men and >88 cm in women, or body mass index ≥30 kg/m2), as well as in those with dyslipidemia (total cholesterol >190 mg/dL, or low-density lipoprotein cholesterol >115 mg/dL, or high-density lipoprotein cholesterol <40 mg/dL in men and <46 mg/dL in women, or triglycerides >150 mg/dL, or under specific treatment with a lipid lowering drug).Citation1 The superiority of olmesartan for controlling blood pressure in patients with metabolic disorders may be explained by an overexpression of vascular angiotensin II type 1 (AT1)-receptors and overactivation of the RAS, which are the targets for the drug, and by a more specific inhibiting action of the ARB on the systemic and adipose tissue RAS.Citation40,Citation41
Figure 2 Percentage of normalized patients.
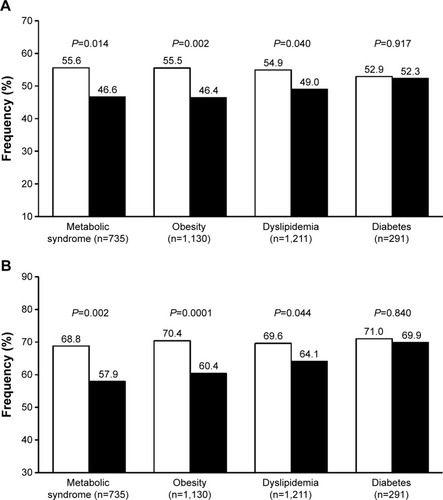
Despite a high rate of blood pressure normalization in the subgroup of diabetics, no statistically significant differences were observed between the two treatment groups (). The fact that olmesartan is as effective as ramipril in diabetic patients is in line with the results of a recent meta-analysis of 23 randomized controlled studies comparing ARBs and ACE inhibitors: no significant difference was found in the proportion of patients who achieved successful blood pressure control on a single antihypertensive agent of the ARB or ACE-inhibitor class.Citation36,Citation42
Blood pressure goal attainment according to renal function status
An impaired renal function is a frequent finding in hypertensive patients and constitutes a very potent predictor of future cardiovascular events.Citation43 Current evidence supports the use of ARBs or ACE inhibitors as the therapy of choice for hypertension in patients with chronic kidney disease, due to specific renoprotective effects of these drugs, which are beyond their antihypertensive effect.Citation44–Citation46
A post hoc analysis of the two pooled studies has previously shown that olmesartan medoxomil is efficacious in controlling blood pressure in the elderly patients of the study, independently of their renal function status, assessed by estimated glomerular filtration rate (eGFR), using the Cockroft–Gault equation.Citation47 The efficacy of olmesartan proved to be generally superior to that of ramipril, in terms of blood pressure normalization, particularly in patients with normal or increased eGFR (≥90 mL/min/1.73 m2) and in those with slightly reduced eGFR (60–90 mL/min/1.73 m2). Data reanalysis based on the currently recommended blood pressure targets (<140/90 or <150/90 mmHg) confirmed a statistically significant superiority of olmesartan vs ramipril in the normal or increased eGFR subgroup, with a comparable efficacy of the two drugs in the other two categories ().
Figure 3 Percentage of normalized patients.
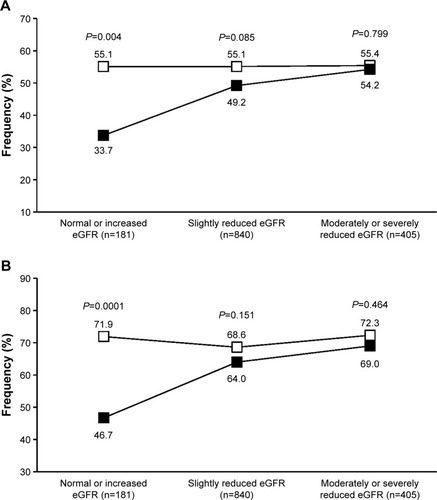
Thus, it seems that RAS inhibition is effective in controlling blood pressure in older hypertensives, regardless of the renal status of the patients, even when less tight blood pressure control is required. Also in this case, as in the case of a tighter blood pressure control, olmesartan may help achieving a better blood pressure control in the subgroup of patients with a preserved renal function or at an early stage of the kidney disease.
Patients at target according to number and types of previous antihypertensive treatment
Patients with hypertension at highest risk of cardiovascular complications, such as older persons, have a greater chance of being resistant to particular classes of drugs or may not adequately respond to monotherapy.Citation1 In order to gain further insight into the mechanisms behind the efficacy of olmesartan and ramipril in our pooled datasets of aged hypertensives, we evaluated response rate in subgroups of patients according to the number of drugs and type or RAS antagonist (ARB or ACE inhibitor) used at the time of entering the studies.
In never-treated patients (24.8% of the whole study population), olmesartan ability to achieve the blood pressure target, whether it was based on the 140/90 or the 150/90 mmHg threshold, was better than that of ramipril (with statistically significant differences for the <140/90 mmHg target), suggesting that the subgroup of patients of our population previously receiving no antihypertensive drug was more prone to respond to the ARB than to the ACE inhibitor (). Additionally, olmesartan appeared to be significantly superior to ramipril in the subgroup of patients that were previously treated with a combination therapy, suggesting that an olmesartan-based monotherapy may be a possible choice for patients that are less susceptible to an adequate blood pressure response to previous multiple antihypertensive treatment.
Figure 4 Percentage of normalized patients.
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin-receptor blocker.
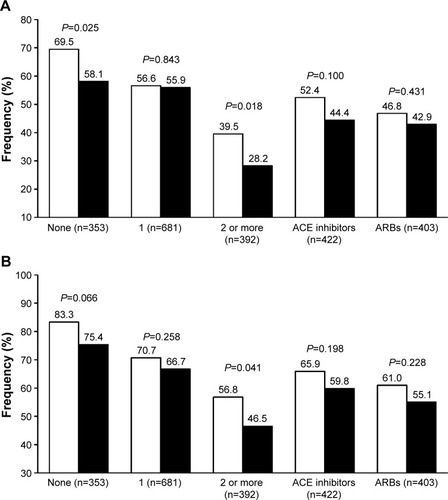
Blood pressure response to a RAS inhibitor may be reduced or event blunted in patients previously not responding to a drug from the same class. In our population, the rate of blood pressure control was similar with olmesartan and ramipril, independently of the kind of previous antihypertensive treatment, whether it was based on an ACE inhibitor or an ARB ().
Drug safety according to blood pressure targets
As previously shown, 44 patients (3.1%) reported 67 adverse events attributed to study treatment: 21 patients received olmesartan (33 adverse events) and 23 ramipril (34 adverse events) (P=0.767 between treatments).Citation20 The rate of patients with drug-related adverse events was the same in the group normalized at a target of <140/90 mmHg (23 of 740 patients, 3.1%) and of <150/90 mmHg (20 of 950 patients, 3.1%, P=0.948). In these two target groups, the rate of patients reporting adverse events attributed to study drug never differed between treatments (<140/90 mmHg: olmesartan 2.0% vs ramipril 4.3%, P=0.074; <150/90 mmHg: olmesartan 2.2% vs ramipril 4.0%, P=0.110). Thus, in our study, the risk of reporting an adverse drug reaction was not related to the blood pressure level achieved during treatment.
Discussion and overall conclusion
All current hypertension guidelines have raised the target blood pressure goals in older hypertensive patients, while eliminating the tighter control recommendations in patients with diabetes and renal disease.Citation1,Citation2,Citation15–Citation17 Notwithstanding such recommendations, strong debate exists among scientists on whether, in older or high-risk populations, blood pressure cutoffs should remain more conservative, namely kept higher, or rather a more aggressive approach should be followed, as it was in the past.Citation7,Citation48–Citation50 The disagreement between studies and the difficulty in weighting the available evidence in the absence of definite data are reflected in the guidelines: some of them recommend a blood pressure target of <150/90 mmHg for persons older than 60 years,Citation2 whereas others recommend a goal of <140/90 mmHg, in persons aged 80 years or younger and <150/90 mmHg only in frail persons aged 80 years or more.Citation1,Citation15–Citation17
We attempted to provide a better insight into this controversy, by reanalyzing the results of two large randomized studies at the light of the new blood pressure targets recommended by present hypertension guidelines. As in the original study, the efficacy of olmesartan was not negatively affected by age, sex, hypertension type, diabetes status, or other concomitant clinical conditions or cardiovascular risk factors. In most cases, olmesartan provided better blood pressure control than ramipril. Olmesartan was significantly more effective than ramipril in male patients, in younger patients (aged 65–59 years), in patients with a normal eGFR, and in those with diastolic ± systolic hypertension. Olmesartan showed better results than ramipril also in specific categories of high-risk patients, such as those with metabolic syndrome, obesity, dyslipidemia, and in general, in patients with a high or very high cardiovascular risk. Interestingly, patients previously untreated or treated with two or more antihypertensive drugs showed a significantly larger response with olmesartan than with ramipril.
Having said so, we must acknowledge some limitations of our post hoc analysis. First, although we pooled together data from two adequately powered, randomized, double-blind, parallel group studies with an identical design, the fact of raising the target of adequate blood pressure control and applying less stringent criteria increased per se the rate of responders in both study treatments. This is because in the original study drug, uptitration and treatment tailoring were based on targets lower than those used in this reanalysis (<140/90 mmHg in nondiabetic patients and <130/80 mmHg in diabetic patients). Second, we should acknowledge as a potential source of difference among treatment groups the fact that the antihypertensive effect of the maximum dose of ramipril employed in our study (10 mg) might not correspond in terms of efficacy to that of olmesartan (40 mg). The use of higher doses of ramipril could have allowed achieving better responses, but in the original study, comparisons were limited to the maximum doses currently recommended for the two drugs. Third, one meta-analysis documented that while the blood pressure dependent effects of ACE inhibitors and ARBs on the risk of stroke, coronary heart disease, and heart failure are similar, ACE inhibitors but not ARBs have blood pressure independent effect on the risk of major coronary disease events.Citation51 Very recently, a systematic review showed that ACE inhibitors are more effective in preventing coronary heart disease and less in preventing stroke, whereas ARBs are inferior in preventing coronary heart disease.Citation52 However, the blood pressure lowering effect of the two classes of drugs in hypertensive patients seems to be quite superimposable.Citation36,Citation42 Thus, in spite of some superiority in terms of antihypertensive effect of a given ARB over a given ACE inhibitor, as in our study, we must admit that there is no evidence in medical literature to recommend ARB over ACE inhibitor therapy. Differences found among active principles may instead suggest specific choices in specific conditions, or preferable combinations of drugs and doses. Fourth, in our study we showed a better blood pressure response with olmesartan, but we could not demonstrate any superiority in terms of prevention of cardiovascular outcomes because these endpoints were not assessed in the study. Thus, we cannot conclude that olmesartan is superior to ramipril in terms of cardiovascular protection in the elderly hypertensive patient.
Notwithstanding these limitations, our results may be relevant for the clinical practice, providing some indication on the possible different response of elderly hypertensive patients to two different RAS inhibitors, when patients are targeted according to the blood pressure levels recommended by recent hypertension guidelines.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper, and agree to be accountable for all aspects of the work.
Acknowledgments
This work was financially supported by Menarini International Operations Luxembourg through an unconditional and unrestricted grant. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Disclosure
SO, EM, JMM, and MV have occasionally received grants for lectures by the manufacturers of olmesartan or ramipril. MV has been consultant in scientific advisory board of Daiichi Sankyo, manufacturer of olmesartan.
References
- ESH/ESC Task Force for the Management of Arterial Hypertension2013 Practice guidelines for the management of arterial hypertension of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC): ESH/ESC Task Force for the Management of Arterial HypertensionJ Hypertens201331101925193824107724
- JamesPAOparilSCarterBL2014 Evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8)JAMA2014311550752024352797
- ZanchettiAGrassiGManciaGWhen should antihypertensive drug treatment be initiated and to what levels should systolic blood pressure be lowered? A critical reappraisalJ Hypertens200927592393419381107
- BeckettNSPetersRFletcherAETreatment of hypertension in patients 80 years of age or olderN Engl J Med2008358181887189818378519
- JATOS Study GroupPrincipal results of the Japanese trial to assess optimal systolic blood pressure in elderly hypertensive patients (JATOS)Hypertens Res200831122115212719139601
- OgiharaTSarutaTRakugiHTarget blood pressure for treatment of isolated systolic hypertension in the elderly: valsartan in elderly isolated systolic hypertension studyHypertension201056219620220530299
- ZhangYZhangXLiuLZanchettiAIs a systolic blood pressure target <140 mmHg indicated in all hypertensives? Subgroup analyses of findings from the randomized FEVER trialEur Heart J201132121500150821345850
- YusufSDienerHCSaccoRLTelmisartan to prevent recurrent stroke and cardiovascular eventsN Engl J Med2008359121225123718753639
- PittBByingtonRPFurbergCDEffect of amlodipine on the progression of atherosclerosis and the occurrence of clinical events. PREVENT InvestigatorsCirculation2000102131503151011004140
- Poole-WilsonPALubsenJKirwanBAEffect of long-acting nifedipine on mortality and cardiovascular morbidity in patients with stable angina requiring treatment (ACTION trial): randomised controlled trialLancet2004364943784985715351192
- BraunwaldEDomanskiMJFowlerSEAngiotensin-converting-enzyme inhibition in stable coronary artery diseaseN Engl J Med2004351202058206815531767
- ArguedasJALeivaVWrightJMBlood pressure targets for hypertension in people with diabetes mellitusCochrane Database Syst Rev201310CD00827724170669
- ArguedasJAPerezMIWrightJMTreatment blood pressure targets for hypertensionCochrane Database Syst Rev20093CD00434919588353
- UpadhyayAEarleyAHaynesSMUhligKSystematic review: blood pressure target in chronic kidney disease and proteinuria as an effect modifierAnn Intern Med2011154854154821403055
- National Clinical Guideline Centre (UK)Hypertension: The Clinical Management of Primary Hypertension in Adults: Update of Clinical Guidelines 18 and 34 InternetLondonRoyal College of Physicians (UK)2011 Available from: http://www.ncbi.nlm.nih.gov/books/NBK83274/Accessed July 31, 2015
- AronowWSFlegJLPepineCJACCF/AHA 2011 expert consensus document on hypertension in the elderly: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents developed in collaboration with the American Academy of Neurology, American Geriatrics Society, American Society for Preventive Cardiology, American Society of Hypertension, American Society of Nephrology, Association of Black Cardiologists, and European Society of HypertensionJ Am Soc Hypertens20115425935221771565
- WeberMASchiffrinELWhiteWBClinical practice guidelines for the management of hypertension in the community: a statement by the American Society of Hypertension and the International Society of HypertensionJ Clin Hypertens (Greenwich)2014161142624341872
- MalaccoEOmboniSVolpeMAuteriAZanchettiAESPORT Study GroupAntihypertensive efficacy and safety of olmesartan medoxomil and ramipril in elderly patients with mild to moderate essential hypertension: the ESPORT studyJ Hypertens201028112342235020829713
- MallionJMOmboniSBartonJAntihypertensive efficacy and safety of olmesartan and ramipril in elderly patients with mild to moderate systolic and diastolic essential hypertensionBlood Press Suppl2011131121091270
- OmboniSMalaccoEMallionJMFabrizziPVolpeMOlmesartan vs ramipril in elderly hypertensive patients: review of data from two published randomized, double-blind studiesHigh Blood Press Cardiovasc Prev201421111924435506
- KereiakesDJNeutelJStoakesKAThe effects of an olmesartan medoxomil-based treatment algorithm on 24-hour blood pressure levels in elderly patients aged 65 and olderJ Clin Hypertens (Greenwich)200911841142119695028
- SaitoIKushiroTHirataKThe use of olmesartan medoxomil as monotherapy or in combination with other antihypertensive agents in elderly hypertensive patients in JapanJ Clin Hypertens (Greenwich)200810427227918401224
- HeagertyAMMallionJMOlmesartan medoxomil in elderly patients with essential or isolated systolic hypertension: efficacy and safety data from clinical trialsDrugs Aging2009261617619102515
- OgawaHKim-MitsuyamaSMatsuiKAngiotensin II receptor blocker-based therapy in Japanese elderly, high-risk, hypertensive patientsAm J Med20121251098199022503610
- MallionJMHeagertyALaeisPSystolic blood pressure reduction with olmesartan medoxomil versus nitrendipine in elderly patients with isolated systolic hypertensionJ Hypertens200725102168217717885562
- OgiharaTSarutaTRakugiHCombinations of olmesartan and a calcium channel blocker or a diuretic in elderly hypertensive patients: a randomized, controlled trialJ Hypertens201432102054206324999799
- MatsuiKKim-MitsuyamaSOgawaHSex differences in response to angiotensin II receptor blocker-based therapy in elderly, high-risk, hypertensive Japanese patients: a subanalysis of the OSCAR studyHypertens Res201437652653224599010
- SeelandURegitz-ZagrosekVSex and gender differences in cardiovascular drug therapyHandb Exp Pharmacol201221421123623027453
- ConroyRMPyöräläKFitzgeraldAPEstimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE projectEur Heart J20032411987100312788299
- RoseiEAReduction of cardiovascular risk through angiotensin II type 1 receptor antagonism: focus on olmesartan medoxomilHigh Blood Press Cardiovasc Prev200815423124323355126
- VolpeMTocciGOlmesartan in the treatment of hypertension in elderly patients: a review of the primary evidenceDrugs Aging2013301298799824170236
- CornierMADabeleaDHernandezTLThe metabolic syndromeEndocr Rev200829777782218971485
- GamiASWittBJHowardDEMetabolic syndrome and risk of incident cardiovascular events and death: a systematic review and meta-analysis of longitudinal studiesJ Am Coll Cardiol200749440341417258085
- WuSHLiuZHoSCMetabolic syndrome and all-cause mortality: a meta-analysis of prospective cohort studiesEur J Epidemiol201025637538420425137
- NakaoYMTeramukaiSTanakaSEffects of renin-angiotensin system blockades on cardiovascular outcomes in patients with diabetes mellitus: a systematic review and meta-analysisDiabetes Res Clin Pract2012961687522197527
- PowersBGreeneLBalfeLMUpdates on the treatment of essential hypertension: a summary of AHRQ’s comparative effectiveness review of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and direct renin inhibitorsJ Manag Care Pharm2011178 SupplS1S1422088101
- MatcharDBMcCroryDCOrlandoLASystematic review: comparative effectiveness of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers for treating essential hypertensionAnn Intern Med20081481162917984484
- AlbertiKGZimmetPShawJMetabolic syndrome – a new worldwide definition. A Consensus Statement from the International Diabetes FederationDiabet Med200623546948016681555
- OmboniSMalaccoEMallionJMVolpeMAntihypertensive efficacy and safety of olmesartan medoxomil and ramipril in elderly mild to moderate essential hypertensive patients with or without metabolic syndrome: a pooled post hoc analysis of two comparative trialsDrugs Aging2012291298199223179896
- SharmaAMEngeliSThe role of renin-angiotensin system blockade in the management of hypertension associated with the cardiometabolic syndromeJ Cardiometab Syndr200611293517675902
- UngerTThe role of the renin-angiotensin system in the development of cardiovascular diseaseAm J Cardiol2002892A3A10A
- PowersBJCoeytauxRRDolorRJUpdated report on comparative effectiveness of ACE inhibitors, ARBs, and direct renin inhibitors for patients with essential hypertension: much more data, little new informationJ Gen Intern Med201227671672922147122
- MahmoodiBKMatsushitaKWoodwardMAssociations of kidney disease measures with mortality and end-stage renal disease in individuals with and without hypertension: a meta-analysisLancet201238098541649166123013600
- CasasJPChuaWLoukogeorgakisSEffect of inhibitors of the renin-angiotensin system and other antihypertensive drugs on renal outcomes: systematic review and meta-analysisLancet200536695022026203316338452
- KunzRFriedrichCWolbersMMannJFMeta-analysis: effect of monotherapy and combination therapy with inhibitors of the renin angiotensin system on proteinuria in renal diseaseAnn Intern Med20081481304817984482
- VejakamaPThakkinstianALertrattananonDIngsathitANgarmukosCAttiaJReno-protective effects of renin-angiotensin system blockade in type 2 diabetic patients: a systematic review and network meta-analysisDiabetologia201255356657822189484
- MalaccoEOmboniSMallionJMVolpeMAntihypertensive efficacy of olmesartan medoxomil and ramipril in elderly patients with mild to moderate hypertension grouped according to renal function status: a retrospective analysisHigh Blood Press Cardiovasc Prev201219421322223430666
- LiuLZhangYLiuGThe Felodipine Event Reduction (FEVER) Study: a randomized long-term placebo-controlled trial in Chinese hypertensive patientsJ Hypertens200523122157217216269957
- WrightJTJrFineLJLacklandDTOgedegbeGDennison HimmelfarbCREvidence supporting a systolic blood pressure goal of less than 150 mmHg in patients aged 60 years or older: the minority viewAnn Intern Med2014160749950324424788
- XuWGoldbergSIShubinaMTurchinAOptimal systolic blood pressure target, time to intensification, and time to follow-up in treatment of hypertension: population based retrospective cohort studyBMJ2015350h15825655523
- Blood Pressure Lowering Treatment Trialists’ CollaborationTurnbullFNealBBlood pressure-dependent and independent effects of agents that inhibit the renin-angiotensin systemJ Hypertens20072595195817414657
- ThomopoulosCParatiGZanchettiAEffects of blood pressure-lowering on outcome incidence in hypertension: 5. Head-to-head comparisons of various classes of antihypertensive drugs – overview and meta-analysesJ Hypertens2015331321134126039526
