Abstract
Background
The epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) is an effective treatment for advanced lung cancer harboring EGFR gene mutations, and has improved progression-free survival in several clinical trials.
Methods
We investigated 30 stage I non-small-cell lung cancer patients harboring EGFR gene mutations with postoperative intrapulmonary recurrence. Progression-free survival and response rate were analyzed.
Results
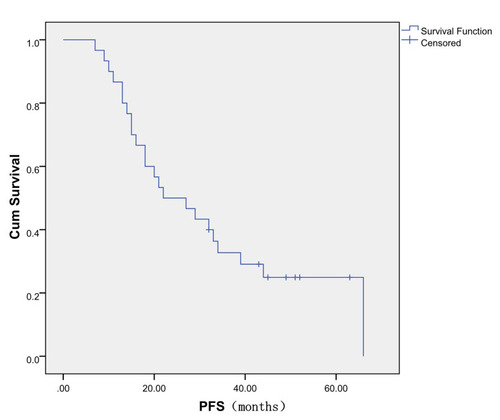
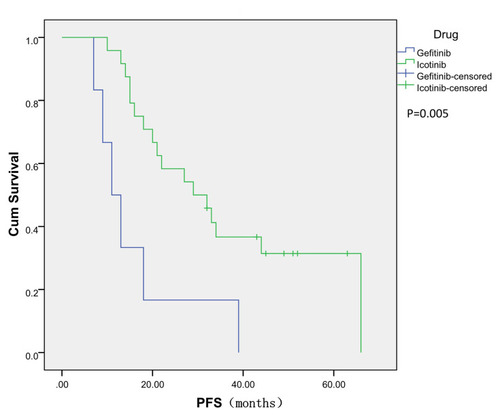
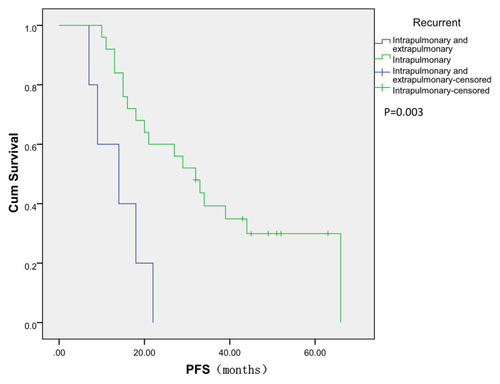
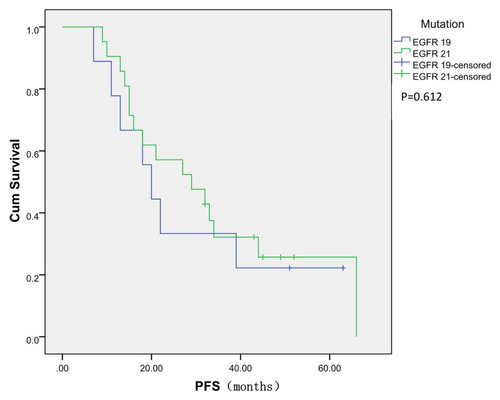
Partial response was achieved in 23 patients and stable disease was found in 7 patients. The objective response rate was 76.7% and disease control rate was 100%. The median progression-free survival (PFS) time was 24.5 months. The median PFS in patients with only intrapulmonary recurrence was significantly superior to patients with both intrapulmonary recurrence and metastasis (32.0 months vs 14.0 months, P = 0.003). The median PFS observed in patients who underwent icotinib treatment was significantly longer than in patients who underwent gefitinib treatment (30.5 months vs 12.0 months, p = 0.005). There were no statistical differences in median PFS between patients with tumors harboring exon 21 mutation and exon 19 deletion, age <65 and ≥65, male and female, smoker and non-smoker.
Conclusion
Our result reveals that first-line EGFR-TKIs treatment for stage I non-small-cell lung cancer patients harboring EGFR gene mutations with postoperative intrapulmonary recurrence is effective and could be a useful option in practical setting.
Introduction
Cancer is one of the major causes of deaths in the world, and over 20% of these dead people are related to lung cancer.Citation1 Lung cancer can be divided into small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), and the most common type is NSCLC. For early stage NSCLC patients with resectable lesions, the best effective treatment is surgery which can extend the long-life expectancy of patients.Citation2 However, tumor recurrence is always the leading cause of postoperative death.Citation3 About 50–60% NSCLC patients in I to IIIA stage suffer from tumor recurrence after surgery.Citation4,Citation5 For extending the survival time of the patients with postoperative recurrence, finding the way to prevent the recurrence of lung cancer and the effective treatment becomes an important research direction.
In the past decade, epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) treatment had become an important therapy for the treatment of advanced and recurrent NSCLC, precisely because of its ability to facilitate the long-term survival of these patients. Specifically, EGFR-TKIs (such as gefitinib, erlotinib, and icotinib) are generally recommended for advanced stage NSCLC patients or postoperatively recurrent NSCLC patients with EGFR mutations and approximately 90% of the mutations are exon 19 deletions or exon 21 L858R point mutations. Several clinical trials have shown that EGFR-TKIs provided better progression-free survival than standard chemotherapy.Citation6–Citation14 However, for the stage I NSCLC patients with postoperative intrapulmonary recurrence whose tumor burden are smaller, there has not been sufficient evidence revealing the clinical curative effect of EGFR-TKIs.
The aim of our study was to evaluate the efficacy of EGFR-TKIs in the stage I NSCLC patients harboring EGFR gene mutations with postoperative intrapulmonary recurrence.
Methods
Patients
The study group included 30 patients with NSCLC. All patients were diagnosed as stage I lung adenocarcinoma harboring activating EGFR mutations and had undergone complete resection at the Department of Thoracic Surgery, the Second Affiliated Hospital, School of Medicine, Zhejiang University between January 2009 and March 2017. NSCLC histology was classified according to the World Health Organization classification and pathological staging was classified according to the new criteria of the Union for International Cancer Control, version 8. All the patients were found to have recurrences in the lungs during routine postoperative examinations and at least one recurrence in the lungs was used to evaluate the therapeutic effects. All the patients were treated with gefitinib or icotinib as the first-line targeted therapy after intrapulmonary recurrence were found. After the start of EGFR-TKIs treatment, chest computed tomography (CT) scan, blood examination and B ultrasonic examination (liver, supraclavicular lymph nodes and adrenal gland) were performed at bimonthly intervals. And magnetic resonance imaging (MRI) of the brain was performed based on the doctor’s decision when clinical symptoms indicating brain involvement were present.
Evaluation
Response measurements to EGFR-TKIs were assessed by the Response Evaluation Criteria in Solid Tumors (RECIST, version 1.1) criteria for each CT evaluation during targeted therapy. The progression-free survival (PFS) was defined as the time between the day of EGFR-TKIs treatment initiation and the day when progressive disease (PD) was observed. The disease control rate (DCR) was defined as the percentage of subjects with complete response (CR), partial response (PR) or stable disease (SD) relative to all randomly assigned patients. The objective response rate (ORR) was defined as the percentage of subjects with CR or PR relative to all randomly assigned patients. Toxicity was evaluated in accordance with the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.0 (CTC-AE).
Statistical Analysis
The survival curves were plotted using the Kaplan-Meier method and differences in PFS was analyzed by Log rank test. Variables include gender, age, smoking status, type of EGFR mutation, type of EGFR-TKIs and recurrence of lesions when starting EGFR-TKIs treatment. Analyses were performed using the SPSS software program, version 24.0 (SPSS Inc., Chicago, IL, USA), for Windows. And P < 0.05 was considered to indicate a statistically significant difference.
Data Availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Ethics
The study was approved by the Institutional Ethics Committee of the Second Affiliated Hospital of Zhejiang University School of Medicine. All patients provided informed consent, and this study was conducted in accordance with the Declaration of Helsinki.
Results
Patient Characteristics
The median age of the patients was 68 years (range, 54–89 years); there were 12 (40%) men and 18 (60%) women; there were 9 patients (30%) patients with an exon 19 deletion and 21 patients (70%) with an exon 21 mutation. Of the 30 patients, 8 (26.7%) were current smokers and 22 (73.3%) were not. All the patients had adenocarcinoma. Of the 30 patients, 6 (20.0%) patients received gefitinib as the first-line treatment and 24 (80.0%) patients received icotinib (). All the patients were found postoperative intrapulmonary recurrence and 5 patients were also found distant metastases like liver metastases, bone metastases and brain metastases.
Table 1 Analysis of PFS for 30 Patients with Intrapulmonary Recurrent NSCLC Treated with EGFR-TKIs
Treatment Efficacy
Until March 2020, PR (according to RECIST) was observed in 23 patients and SD was observed in 7 patients. No one had CR. The DCR was 100.0% and the ORR was 76.7%. The median PFS time for all patients was 24.5 months (, 7.0–66.0 months). The median PFS was 12.0 months in patients who received gefitinib as the first-line treatment and 30.5 months in those who received icotinib as the first-line treatment (), which corresponded to a significant difference between these sets of patients (P = 0.005, Log rank test). The median PFS was 32.0 months in patients with only intrapulmonary recurrence and 14.0 months in patients with both intrapulmonary recurrence and metastasis (), which corresponded to a significant difference between these sets of patients (P = 0.003, Log rank test). The median PFS was 20.0 months in patients with EGFR gene mutations within exon 19 and 29.0 months in patients with mutations within EGFR exon 21 (). There was no significant difference in median PFS among these sets of patients with different EGFR gene mutations (P = 0.612, Log rank test).
Toxicity
Adverse events have been observed in 15 (50.0%) patients, and no one stopped the targeted therapy due to uncontrollable toxicity. The most common adverse events were rash, diarrhea and liver function abnormalities.
Discussion
In this study of first-line EGFR-TKIs treatment in stage I NSCLC patients harboring EGFR gene mutations with postoperative intrapulmonary recurrence, the DCR was 100.0% and the ORR was 76.7%, the median PFS time was 24.5 months and the toxicities experienced were acceptable These results suggest that first-line EGFR-TKIs treatment is a promising therapeutic approach for these patients.
Gefitinib is an oral, small-molecule EGFR-TKI for NSCLC. Previously, a Phase III study of the IPASS (Iressa Pan-Asia Study) trial revealed that, for patients with EGFR mutations, PFS and ORR were significantly longer among those who received gefitinib than among those who received carboplatin–paclitaxel (9.8 months vs 6.4 months, P < 0.001). What’s more, gefitinib caused less adverse events.Citation6 Icotinib is also an oral, small-molecule EGFR-TKI for NSCLC and is approved in China. A phase III study of the ICOGEN trial compared icotinib with gefitinib in unselected Chinese patients with advanced NSCLC. And the results demonstrated that icotinib was non-inferior to gefitinib in terms of PFS at 4.6 months vs 3.4 months, but the difference was not statistically significant. What’s more, the icotinib had less incidence of adverse reactions than gefitinib (60.5% vs 70.4%, P = 0.04).Citation15–Citation18 In our study, the median PFS observed in patients who underwent icotinib treatment was longer than in patients who underwent gefitinib treatment. (30.5 months vs 12.0 months, P = 0.005). This result may be affected by the small sample size.
In addition, in clinical trials of EGFR-TKIs treatment for recurrent or advanced NSCLC with EGFR gene mutations, the median PFS was 10.8 months in the NEJ002 study, 9.2 months in the WJTOG3405 study.Citation12,Citation13 They were all less than the median PFS in our study (24.5 months). What’s more, the response rates and disease-control rates were 73.7% and 79.5% in the NEJ002 study, 62.1% and 93.1% in the WJTOG3405 study and 71.2% and 91.7% in the IPASS study.Citation6,Citation12,Citation13,Citation19 Compared with these studies, our study’s ORR is 76.7% and DCR is 100%, which was better. We supposed the small tumor burden caused the result. The patients in our study all underwent the radical resection of lung cancer, which meant they probably had smaller tumor burden at the start of EGFR-TKIs treatment than the patients with advanced stage NSCLC in these studies. A small tumor burden could increase the efficacy of EGFR-TKIs, delay the arrival of tolerance moment and help the drug play a long-time anti-cancer efficacy. In our study, the patients with only intrapulmonary recurrence whose tumor burden was thought to be smaller, had a longer PFS than the patients with both intrapulmonary recurrence and metastasis (32.0 months vs 14.0 months, P = 0.003). By the way, in the WJTOG3405 study, the patients with postoperative recurrence had a longer median PFS than the patients with advantage stage NSCLC (13.7 months vs 8.4 months), thus supporting our conjecture.Citation13
In our study, there was no difference in the median PFS between the patients with tumors harboring exon 21 mutation and those tumors harboring exon 19 deletion (29.0 months vs 20.0 months, P = 0.612). In Yamada et al’s study, the patients with tumors harboring exon 21 mutations had a longer PFS than that of patients harboring exon 19 deletion (11.0 months vs 8.0 months) who underwent erlotinib treatment. However, there was no significant difference in PFS with respect to patients with tumors harboring exon 19 and 21 mutations who underwent gefitinib treatment in the NEJ002 study and WJTOG3405 study.Citation12,Citation13 Sequist et al even showed that the patients with tumors harboring exon 19 deletion also had a longer PFS than that of patients harboring exon 21 mutations who underwent afatinib treatment.Citation14 We suspect that the different choices of EGFR-TKIs for patients with tumors harboring different exon mutations may lead to these results. Further investigations are needed to evaluate the speculation.
There are several limitations in the present study. Firstly, this study is a retrospective study that the patient-selection and time-trend biases regarding the treatment for recurrent disease are unavoidable. Secondly, the sample size of the study is small and the follow-up time among the patients is different. Because of the small sample size, multivariate analysis of many factors is very difficult. Therefore, to obtain more credible data, we will further improve the study and enlarge the sample size. At the same time, we hope more relevant studies can appear in the future.
Conclusion
Our result reveals that first-line EGFR-TKIs treatment for stage I NSCLC harboring EGFR gene mutations with postoperative intrapulmonary recurrent is effective and could be a useful option in practical setting. Further studies are warranted.
Abbreviations
EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; NSCLC, non-small cell lung cancer; SCLC, small cell lung cancer; PFS, progression-free survival; PD, progressive disease; DCR, disease control rate; CR, complete response; PR, partial response; SD, stable disease; ORR, objective response rate.
Disclosure
The authors report no conflict of interest in this work.
Acknowledgments
This work was supported by grants from the Science and Technology Department of Zhejiang Province (2013c03044-7, YC), Natural Science Foundation of Zhejiang Province (LY13H160016, YC), and Wu Jieping Medical Foundation (320.6799.15040).
References
- Jemal A, Siegel R, Ward E, et al. Cancer Statistics. CA Cancer J Clin. 2007;57(1):43–66.17237035
- Masuda M, Kuwano H, Okumura M, et al. Committee for scientific affairs, the Japanese Association for Thoracic Surgery. Thoracic and cardiovascular surgery in Japan during 2012: annual report by The Japanese Association for Thoracic Surgery. Gen Thorac Cardiovasc Surg. 2014;62(12):734–764. doi:10.1007/s11748-014-0464-025355583
- Sonobe M, Yamada T, Sato M, et al. Identification of subsets of patients with favorable prognosis after recurrence in completely resected non-small cell lung cancer. Ann Surg Oncol. 2014;21(8):2546–2554. doi:10.1245/s10434-014-3630-924633668
- Arriagada R, Bergman B, Dunant A, Le Chevalier T, Pignon JP, Vansteenkiste J. International Adjuvant Lung Cancer Trial Collaborative Group: cisplatin-based adjuvant chemotherapy in patients with completely resected non-small-cell lung cancer. N Engl J Med. 2004;350:351–360.14736927
- Paez JG, Jänne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–1500. doi:10.1126/science.109931415118125
- Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–957. doi:10.1056/NEJMoa081069919692680
- Lee CK, Brown C, Gralla RJ, et al. Impact of EGFR inhibitor in non-small cell lung cancer on progression-free and overall survival: a meta-analysis. J Natl Cancer Inst. 2013;105:595–605. doi:10.1093/jnci/djt07223594426
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350:2129–2139. doi:10.1056/NEJMoa04093815118073
- Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised phase III trial. Lancet. 2008;372(9652):1809–1818. doi:10.1016/S0140-6736(08)61758-419027483
- Thatcher N, Chang A, Parikh P, et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa survival evaluation in lung cancer). Lancet. 2005;366(9496):1527–1537. doi:10.1016/S0140-6736(05)67625-816257339
- Mitsudomi T, Kosaka T, Yatabe Y. Biological and clinical implications of EGFR mutations in lung cancer. Int J Clin Oncol. 2006;11(3):190–198. doi:10.1007/s10147-006-0583-416850125
- Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non–small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362(25):2380‑2388. doi:10.1056/NEJMoa0909530
- Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised Phase 3 trial. Lancet Oncol. 2010;11(2):121‑128. doi:10.1016/S1470-2045(09)70364-X
- Sequist LV, Joshi VA, Jänne PA, et al. Response to treatment and survival of patients with non‑small cell lung cancer undergoing somatic EGFR mutation testing. Oncologist. 2007;12:90‑98. doi:10.1634/theoncologist.12-1-90
- Shi Y, Zhang L, Liu X, et al. Icotinib versus gefitinib in previously treated advanced non-small-cell lung cancer (ICOGEN): a randomised, double-blind phase 3 non-inferiority trial. Lancet Oncol. 2013;14(10):953–961. doi:10.1016/S1470-2045(13)70355-323948351
- Liang JL, Ren XC, Lin Q. Treating advanced non-small-cell lung cancer in Chinese patients: focus on icotinib. Onco Targets Ther. 2014;7:761–770. doi:10.2147/OTT.S4923324876785
- Zhao Q, Shentu J, Xu N, et al. Phase I study of icotinib hydrochloride (BPI-2009H), an oral EGFR tyrosine kinase inhibitor, in patients with advanced NSCLC and other solid tumors. Lung Cancer. 2011;73:195–202. doi:10.1016/j.lungcan.2010.11.00721144613
- Sun Y, Shi Y, Zhang L, et al. A randomized, double-blind phase III study of icotinib versus gefitinib in patients with advanced non-small cell lung cancer (NSCLC) previously treated with chemotherapy (ICOGEN). J Clin Oncol. 2011;29(15_suppl):abstr 7522. doi:10.1200/jco.2011.29.15_suppl.7522
- Fukuoka M, Wu YL, Thongprasert S, et al. Biomarker analyses and final overall survival results from a Phase III, randomized, open-label, first-line study of gefitinib versus carboplatin/paclitaxel in clinically selected patients with advanced non-small-cell lung cancer in Asia (IPASS). J Clin Oncol. 2011;29:2866–2874. doi:10.1200/JCO.2010.33.423521670455