Abstract
Diabetes and sarcopenia are emerging as serious public health issues. Sarcopenia, an age-related disorder characterized by loss of skeletal muscle mass and function, is recognized as a new complication in elderly patients with type 2 diabetes mellitus (T2DM). Type 2 diabetes is characterized by insulin resistance, chronic inflammation, accumulation of advanced glycation products and increased oxidative stress, which can negatively affect skeletal muscle mass, strength and function leading to sarcopenia. There is a mutual interrelationship between T2DM and sarcopenia in light of pathophysiology mechanism and long-term outcome. T2DM will accelerate the decline of muscle mass and function, which will in turn lead to glucose metabolism disorders, reduced physical activity and the risk of diabetes. However, the specific mechanism involved has not been thoroughly studied. Therefore, this review aims to explore the pathophysiology and therapeutic strategy related to sarcopenia and diabetes and provide insight for future investigations, which is of great significance for improving the quality of life in the elderly with diabetes and concurrently reducing the incidence of related complications.
Introduction
Type 2 Diabetes mellitus (T2DM) is a metabolic disease characterized by chronic hyperglycemia due to insulin secretion and (or) utilization disorders. Epidemiological evidence shows that about 387 million adults are suffering from DM worldwide,Citation1 and it is estimated that it will increase to 693 million by 2045,Citation2 which has greatly burdened patients and society. Studies have found that both men and women with diabetes have lower bone mass index valuesCitation3 and the risk of sarcopenia in patients with T2DM is 1.5–2 times higher than that of non-diabetic people.Citation4 Currently, sarcopenia has been regarded as a new complication of T2DM,Citation5 which not only leads to poor quality of life but also increases the risk of physical disability and even death.Citation6–8
The Asian Working Group for Sarcopenia (AWGS) 2014 consensus defined sarcopenia as “age-related decline of skeletal muscle plus low muscle strength and/or physical performance”,Citation9 which refers to age-related syndrome of progressive skeletal muscle loss with decrease in muscle strength and/or muscle function. It is a progressive and systemic skeletal muscle disorder that usually occurs as an age-related process in older adultsCitation10 and is associated with serious consequences in the elderly such as frailty, falls, fractures, physical disability and death.Citation11,Citation12 According to the latest diagnostic criteria of the Asian Working Group on Sarcopenia (AWGS) in 2019, the prevalence rates of suspected sarcopenia, sarcopenia, and severe sarcopenia in China are 38.5%, 18.6%, and 8.0%, respectively.Citation13 It is estimated that the prevalence of sarcopenia will increase significantly within the next 30 years,Citation14 becoming a major public issue. Sarcopenia and T2DM have a bidirectional relationship,Citation15 increase the risk of each otherCitation16 and lead to functional decline and disability.Citation17 Recent studies have shown that patients with T2DM and sarcopenia have a higher mortality rate than those without sarcopenia.Citation18 However, little attention has been paid to elderly T2DM patients with sarcopenia. Hence, it is important to study the pathogenesis of both for a proper management of this clinical complexity.
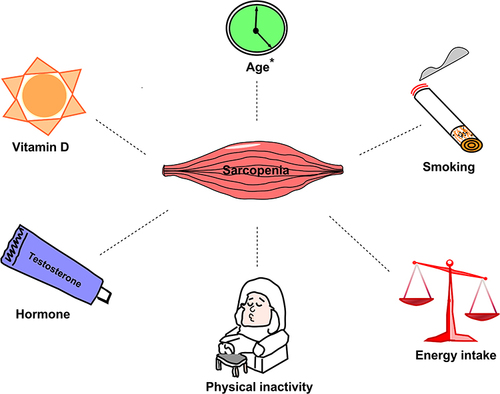
The Converging Risk Factors for Sarcopenia and Diabetes
Aging is one of the most important risks of diabetes and sarcopenia.Citation19 One remarkable physiological sign of the aging process is the gradual loss of skeletal muscle mass and function,Citation20 which latently onset around the age of 30,Citation21 and will accumulate to 30–50% extremely loss in 80 years old.Citation22 The partial mechanism for this phenomenon may lie in the skeletal muscle, the major organ that possesses glucose transporter 4 (GLUT4) responsible for uptake of glucose, accounting for approximately 80% of glucose clearance,Citation23,Citation24 and the utility of glucose also latently declines, increasing susceptibility to T2D.Citation25
Unlike muscle tissue, the transformation of body fat occurs at a certain age.Citation26 The significant re-distribution of the adipose tissue is pivot to this change.Citation12 It is found that persistent localization switches from subcutaneous to visceral may lead to the ectopic accumulation of adipocytes and lipids in, such as bone marrow, liver, and especially skeletal muscle.Citation12,Citation27 Compared with healthy elderly individuals, diabetic patients acquire up to 3-folds of the intramuscular fat stores,Citation28 a trend for the increased risk of sarcopenia and insulin resistance (IR).
In addition, the imbalance between muscle protein anabolic and catabolic pathways paves way for the overall loss of skeletal muscle in aging. Microstructure alterations include the shrinkage and reduction of myofibers, particularly type II myofibers.Citation25 Aging also promotes the satellite cells decrease, as well as the shift from type II to type I myofibers.Citation29 This is more overt in the functional change of muscle fibers, especially type IIb, which release certain proteins and myokines that influence glucose metabolism in a more direct manner. Thus, loss of skeletal muscle mass due to aging or underlying diseases can exacerbate disarrangement of glucose metabolism.Citation30
Due to the decreased protection mechanism or the other agents, aging is also accompanied by an increase in inflammatory markers and oxidative stress, both of which have been implicated as significant pathophysiological changes in DM and sarcopenia.Citation31,Citation32 Specifically, mitochondrial dysfunction and advanced glycation products associated with aging may adversely affect muscle quality and glycemic levels.Citation17 Concurrently, changes in hormone levels also contribute to chronic inflammation.Citation33 For instance, reduced testosterone levels impaired self-renewal of satellite cell in muscle.Citation34 It is noteworthy that decline in serum testosterone levels in males with diabetes may be more significant.Citation35 Therefore, testosterone deficiency is closely associated with sarcopenia in aged diabetic patients.
Besides, both energy requirements and intake decrease with aging. Between the ages of 40 and 70, energy intake decreases by an average of 25%. This reduction inevitably leads to weight and muscle loss, which adversely affects strength and physical performance,Citation36 which in turn has an effect upon glucose metabolism. In addition, a sedentary lifestyle during aging is also a major contributor to the decline of skeletal muscle mass and insulin sensitivity.Citation37
Vitamin D, a dietary precursor of 1,25 (OH)2D3, is often considered an important nutrient for maintaining proper bodily function.Citation38 Vitamin D may affect metabolism and muscle health. Low vitamin D levels are associated with poorer physical function,Citation39 worse glycemic controlCitation40 and an increased risk of developing diabetes in elderly adults.Citation41 Inversely, vitamin D deficiency is common in patients with T2DM.Citation42 Vitamin D plays many critical roles in the function of skeletal muscle, such as maintaining muscle excitability through intracellular calcium, the proliferation and differentiation of skeletal muscle stem cells, and so on.Citation43,Citation44 Multiple studies have also demonstrated that vitamin D supplementation can improve muscle strength and prevent atrophy of type II fibers where vitamin D is deficient.Citation45 These evidences suggest that vitamin D emerges as a critical risk factor mediating the development of IR and sarcopenia, but the conclusion remains to be determined.
According to the 2014 Surgeon General’s Report, active smokers are at higher risk of developing T2D by 30–40% when compared to non-smokers.Citation46 Cigarette smoking impacts body weight and composition, peripheral insulin sensitivity, and pancreatic β cell function.Citation47 Numerous studies have described the vicious effects of smoking on skeletal muscle function and morphology, especially, the thigh muscles.Citation48 The partial consequence is degeneration in muscle fatigue resistanceCitation49 associated with compromised muscle oxidative capacity,Citation50 and a transition from slow twitch to fast twitch fiber type.Citation51 Furthermore, smoking could promote skeletal muscle wasting via smoking-induced inflammation that facilitate protein breakdown and suppress protein synthesisCitation52 ().
The Metabolic Pathophysiology for Sarcopenia in T2DM
Insulin Resistance (IR)
The main mechanism for T2DM is insulin resistance (IR). Skeletal muscle is one of the target organs for insulin action and insulin resistance.Citation53 Impaired insulin action may promote protein degradation and hamper protein synthesis, resulting in both mass and strength drops of muscle.Citation4 IR in skeletal muscle is the most important factor exacerbating sarcopenia in T2DM patients.Citation54
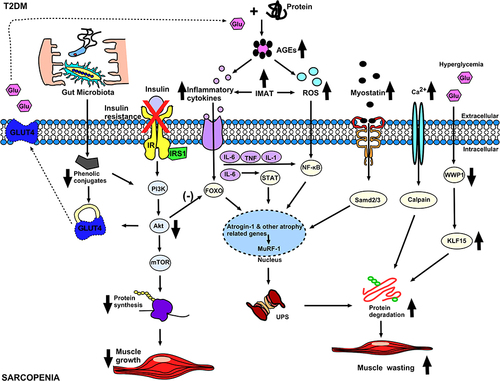
The main proteolytic pathways in muscle involve the ATP-dependent ubiquitin proteasome pathway (UPP), lysosomal autophagy pathway, caspase hydrolysis pathway and calcium-dependent calpain pathway, of which were mainly mediated by IL6/STAT, TNF&IL6/NFκB, myostatin/Smad2/3 and FoxO1/3 signaling pathways.Citation55 Insulin itself reduces proteasome catalytic activity in muscles.Citation56 In the case of IR, high blood glucose levels may cause muscle atrophy via the proteolytic pathway of the ubiquitin-proteasome.Citation57 Metabolic dysregulation due to insulin resistanceCitation58 leads to hyperglycemia and muscle atrophy via the WWP1/KLF15 pathway.Citation59 WWP1 is a member of the ubiquitin ligase protein family. Studies have shown that hyperglycemia upregulates KLF15 protein in skeletal muscle of diabetes models of animals by downregulating the E3 ubiquitin ligase WWP1, followed by inhibition of ubiquitin-dependent degradation of KLF15. KLF15 is a protein associated with muscle wasting. The abundance of the transcription factor KLF15 and the expression of genes associated with muscle atrophy were increased in skeletal muscle of diabetic mice, and mice with muscle-specific KLF15 deficiency were protected from diabetes-induced muscle atrophy.Citation60 This pathway may serve as a therapeutic target for insulin resistance-induced sarcopenia.
Muscle protein synthesis is mainly regulated by two factors: insulin-like growth factor 1 (IGF1) and myostatin.Citation61,Citation62 Protein synthesis inhibition is mediated by inhibition of the IGF1-PI3K-Akt-mTOR and SC-Gαi2 pathways.Citation55 The signaling system of IGF1 and a series of intracellular components plays a crucial role in the regulation of skeletal muscle growth. Akt kinase, also known as protein kinase B (PKB), is a central component of this cascade, controlling protein synthesis through mammalian target of rapamycin (mTOR) and glycogen synthase kinase 3 (GSK3), and through FoxO family of transcription factors for protein degradation.Citation63 IR inhibits mammalian targets of rapamycin pathway,Citation64 while simultaneously stimulating the ubiquitin-proteasome system to upregulate protein catabolism through proteins belonging to FoxO family, and their downstream E3 ubiquitin ligases, resulting in decreased muscle mass.Citation65 As skeletal muscle mass decreases, IR increases lipolysis, releases free fatty acids (FFA) from adipose tissue, and inhibits the growth hormone (GH)-insulin-like growth factor (IGF1) axis that promotes protein synthesis in skeletal muscle.Citation66
Myostatin, a member of the transforming growth factor β (TGF-β) superfamily, downregulates mTORC1 signaling and increases AMPKα2 phosphorylation, both processes could lead to dysregulation of protein synthesis and enhance autophagic proteolysis.Citation67 Myostatin binds to its cognate receptor, the activin II receptor type B (ActRIIB), and thereby exerts multiple effects. After activation of Smad2/Smad3 and dephosphorylation of Akt, muscle protein ubiquitination and proteasomal degradation as well as autophagy will be induced by Atrogin-1 and MuRF1, resulting in process favors for protein degradation.Citation68,Citation69 Additionally, myostatin also inhibits the mammalian target of rapamycin complex 1 (mTORC1).Citation68,Citation69 Besides, caspase, an apoptotic enzyme specific for caspase-3, is overexpressed in T2DM. Myostatin-induced apoptosis was shown to be elicited via activation of the p38-caspase pathway, leading to increased protein degradation.Citation69,Citation70 Hyperinsulinemia caused by IR increases the content of myostatin,Citation71 thereby reducing skeletal muscle through the above-mentioned pathway.
Increased IR increases ectopic fat accumulation and promotes the production of pro-inflammatory factors that inhibit myogenesis and increase muscle catabolism.Citation72 In skeletal muscle, insulin binds to the insulin receptor (GLUT4), allowing glucose uptake by the muscle cells. In the case of IR, its uptake is reduced, leading to hyperglycemia.Citation57
Inflammation
Type 2 Diabetes Mellitus (T2DM) is characterized by chronic low-grade inflammationCitation73 which disrupts glucose and muscle homeostasis.Citation57 This inflammation leads to a downregulation of protein anabolism via the PI3K-Akt pathway, while simultaneously upregulating protein catabolism through the stimulation of the ubiquitin-proteasome system by FoxO family proteins and their downstream E3 ubiquitin ligases.Citation74 The main inflammatory molecules involved in inflammation are TNF-α, IL-6, IL-1 and chemokines, which promote inflammatory cell infiltration and muscle degeneration through NF-κB.Citation75 Inflammatory markers, including interleukin 6 (IL-6), tumor necrosis factor-α (TNF-α), and C-reactive protein (CRP), are often elevated in individuals with T2DMCitation76,Citation77 and associated with IR.Citation78 Increases in pro-inflammatory factors such as IL-6, TNF-α, and CRP have been shown to adversely affect muscle mass and function.Citation79 IL-6 can increase muscle catabolism.Citation80 In an animal experiment, low concentrations of IL-6 were injected into the muscles of mice. The muscles exposed to IL-6 subsequently atrophied due to protein breakdown.Citation81 In addition, clinical studies have also shown that compared with non-diabetic controls, older adults with T2DM have greater loss of leg muscle and strength over 3 years. After adjusting for cytokines including IL-6 and TNF-α, these associations were only partially attenuated.Citation82 IL-6 is associated with increased loss of muscle and strength in T2DM, probably due to direct effects on myocytes, as well as indirect effects on neurons and vasculature. Furthermore, IL-6 in chronically low levels controls signal transducer and activator of transcription 3 (STAT3) phosphorylation through a negative feedback mechanism leading to protein catabolism in muscle via JAK/STATCitation81 and NF-κB-dependent pathways.Citation83 The inflammatory cascade begins with the activation of other interleukins, such as TNF and IL1, which act through NFκB, FOXO4 and other signaling pathways, resulting in muscle atrophy.Citation55 Although IL-10 inhibits mTOR activity, it induces mitophagy, which leads to muscle atrophy.Citation84 The specific molecular mechanisms underlying the adverse effects of CRP on muscle require further investigation.
Oxidative Stress
Oxidative stress, caused by increased reactive oxygen species (ROS) and decreased antioxidant effects,Citation85 is considered an important contributor to aging and diseases. Muscle cells produce ROS (hydroxyl radicals, peroxides, and superoxides) as a by-product of normal metabolism and are more susceptible to oxidative stress.Citation54 While T2DM is associated with increased oxidative stress,Citation57 hyperglycemia in T2DM triggers increased production of ROS.Citation86 ROS activates the ubiquitin-proteasome system and accelerates the degradation of muscle proteins,Citation87 which leads to sarcopenia. It has been shown that increased oxidative stress impairs muscle repair,Citation88 satellite cell differentiation and DNA in diabetic rat.Citation89 Furthermore, oxidative stress inhibits the Akt/mTOR pathway and its downstream targets, subsequently inhibiting protein synthesis and promoting muscle atrophy.Citation90
Mitochondrial dysfunction is another age- and T2DM-related factor associated with oxidative stress leading to deterioration of both metabolic and muscle health.Citation91,Citation92 Mitochondria play an important role in muscle function and metabolism. Compared with younger generation, the oxidative capacity per muscle unit is reduced by 50% in the elderly,Citation93 and accompanied by increased incident of mitochondrial DNA mutations.Citation94 Diabetes-induced mitochondrial dysfunction leads to myocyte apoptosis,Citation95 thereby increasing the risk of sarcopenia. Furthermore, loss of Ca2+ homeostasis can activate non-lysosomal Ca2+-regulated calpains due to increased oxidative stress (another marker of T2DM).Citation96 In diabetic patients, abnormally activated calpain leads to muscle atrophy through its activation of the ubiquitin-proteasome pathway (UPP) and inhibition of the Akt pathway.Citation97
Advanced Glycation End-Products (AGEs)
AGEs, a few of heterogeneous molecules, which are derived from non-enzymatic products of the reaction of glucose or other sugar derivatives with proteins or lipids, are biomarkers of aging, and utilized as assessment of disease status.Citation98 Hyperglycemia in T2DM patients develops with chronic accumulation of AGEs, which are associated with IR and aging.Citation99,Citation100 Accumulation of AGEs has been shown to lead to skeletal muscle atrophy and dysfunction in both basic experimentsCitation101 and human studies.Citation102 The formation of AGEs is irreversible, and accumulates in various neuromusculoskeletal tissues, such as bone, cartilage, muscle, tendons, ligaments, and nerves, where they significantly affect biomechanical properties of those tissues.Citation98 The accumulation of AGEs in diabetes affects skeletal muscle health through pathways such as mitochondrial dysfunction and induction of cell death.Citation17 In addition, AGEs also interfere with muscle contractility by causing charge changes and increasing intramuscular protein cross-linking. By binding to AGEs receptors (RAGE) on skeletal muscle cell membranes, AGEs induce inflammation and activate NADPH oxidase through intracellular signal transduction, which increases the amount of circulating reactive oxygen species (ROS) that promotes oxidative stress.Citation98 Recently, a study shows that higher skin AGEs are associated with a higher prevalence of sarcopenia.Citation103 Whether AGEs measurement can be used to detect pathological changes in skeletal muscle caused by hyperglycemia and aging remains to be further investigated.
Intramuscular Adipose Tissue (IMAT)
IMAT is an ectopic fat deposit, which has been associated with poor outcome of metabolismCitation104 or health of muscle.Citation105 A hallmark pathophysiology of diabetes is obesity and ectopic deposition of fat in many insulin target tissues including skeletal muscle.Citation106 Accumulation of ectopic lipids in skeletal muscle increases the risk of skeletal muscle IR.Citation107 Obese adults with T2DM and peripheral neuropathy had higher calf muscle IMAT content compared with age-matched obese controls.Citation108 Fat accumulation increases adipokine secretion and low-grade inflammation, which leads to both insulin signaling and mitochondrial dysfunction in skeletal muscle, which in turn leads to muscle atrophy.Citation109 Abnormal mitochondrial morphology caused by impaired lipid metabolism leads to membrane stiffness and increased ROSs production in the process of muscle degeneration.Citation110 In addition, IMAT is composed of non-contractile tissue and infiltration of fat into muscle affects the elasticity of skeletal muscle.Citation111 The role of IMAT in sarcopenia and T2DM among the elderly is significant and should be prioritized as a crucial outcome measure in upcoming intervention studies.
Gut Microbiota
Human gut microbiota is able to influence host physiology by regulating multiple processes, including nutrient absorption, inflammation, oxidative stress, immune function, and anabolic balance.Citation112 Dysbiosis of the gut microbiota plays an important role in the pathogenesis of IR and T2D through multiple mechanisms,Citation113 which involve increased intestinal permeability, low-grade endotoxemia, short-chain fatty acids (SCFAs) or changes in the production of branched-chain amino acids (BCAAs), bile acid metabolism, and/or effects on gut hormone secretion.Citation114 Compositional and functional changes in the gut microbiota have been observed in patients with T2DM and prediabetes.Citation115,Citation116 Additionally, fecal microbiota transplantation from healthy donors to patients with metabolic syndrome was associated with increased microbial diversity, improved glycemic control, and insulin sensitivity.Citation117 Recent studies have shown that the gut microbiota also influences the onset and progression of sarcopenia.Citation118,Citation119 An in vitro study showed that phenolic conjugates produced by the microbiota increase glucose uptake in muscle fibers by upregulating GLUT4 and PI3K and induce an anabolic response that increases muscle mass.Citation120 Intake of probiotics and prebiotics modulates skeletal muscle mass and function by modulating gut permeability and the gut-muscle axis.Citation121 Thus, the gut microbiota may be an essential contributor involved in the pathogenesis of sarcopenia and diabetes ().
Figure 2 The possible mechanism of diabetes and sarcopenia. In diabetes, insulin resistance inhibits protein synthesis pathways leading to decreased muscle mass. The increase of inflammatory cytokines, ROS, Myostatin and Calpain related to diabetes will not only inhibit the protein synthesis pathway, but also increase the protein catabolism pathway, thereby causing further decline in muscle mass. Hyperglycemia causes decrease of WWP1, which leads to increase of KLF15, which further aggravates the protein catabolic pathway and leads to muscle atrophy.
The Therapeutic Strategy for Sarcopenia in T2DM
Non-Pharmacological Approaches
Physical Exercise
Exercise is the most cost-effective lifestyle intervention for the prevention and treatment paradigm of diabetes.Citation122 Numerous studies have shown that exercise can also retard muscle loss by attenuating the activation of NF-κB.Citation123,Citation124 The 2018 clinical practice guidelines strongly recommend physical exercise as the primary treatment for sarcopenia.Citation125 Both aerobic and resistance exercise can prevent and treat the decline in muscle mass and strength that occurs with aging.Citation126 Aerobic exercise decreases the level of HbA1c, fasting blood glucose, and IR, as well improves skeletal muscle mass, especially leg strength. Resistance exercise (RE) can effectively enhance muscle strength while ameliorating IR, fat accumulation, and maintaining mitochondrial function in patients with T2DM.Citation127,Citation128 Current clinical experiments have proven short-term moderate-intensity resistance exercise (Each exercise consisted of (i) 5 min of warm-up; (ii) 30 min of RE: biceps (left hand 12 times × 3 groups; right hand 12 times × 3 groups; 1–2 min rest between each group); posterior neck arm extension (12 times × 3 groups, 1–2 min rest between each group); static squat (30s × 3 groups, 1–2 min rest between each group); standing upright against the wall for 10 min; and (iii) 5 min of stretching) is an effective and safe form of exercise. This type of exercise can reduce glycemic levels and its fluctuations, as well as hypoglycemia risks in elderly patients with T2DM and sarcopenia, improving the time in target range (TIR).Citation129 It is noteworthy that for the elderly or (and) patients with neurological diseases, high-intensity exercise is difficult to implement; however, blood flow restriction (BFR) training can be used as an alternative to traditional exercise, and even low strength training also significantly increases muscle strength.Citation130
The Nutrition Supplements
Although the exact mechanism is not yet clear, adequate energy and protein intake can help prevent loss of muscle mass. Animal protein intake is associated with an increased overall risk of T2DM compared with plant protein intake.Citation131 Therefore, protein derived from plant may be the most appropriate source to ensure protein needs in older adults with or at risk of T2DM. Notably, leucine exhibits strong insulinotropic properties that increase amino acid availability for muscle protein synthesis, reduce muscle protein breakdown, and enhance glucose disposal to help maintain glycemic homeostasis.Citation132 A recent study showed that the branched-chain amino acids valine, leucine, and isoleucine longitudinally reduce the odds of muscle mass loss.Citation133 Therefore, supplementation with branched-chain amino acids, leucine, or leucine-rich proteins is one of the most common interventions for the treatment of sarcopenia in older adults.Citation134
In addition, intake of other specific nutrients, including Omega-3 fatty acids, nicotinamide adenine dinucleotide (NAD+) precursor, vitamin D, foods containing anti-inflammatory and antioxidants and dietary fiber can modify insulin sensitivity, oxidative stress and inflammation to maintain muscle mass and improve glycemic level.Citation37 Omega-3s are polyunsaturated fatty acids, mainly derived from marine fish, that increase muscle mass and function in healthy older adults.Citation135 Omega-3 supplementation alone and in combination with exercise can substantially improve metabolic and muscle health. Omega-3 supplementation affects muscle directly by increasing muscle protein synthesisCitation136 and indirectly by reducing systemic inflammation.Citation137 Opacity, type and dose of Omega-3 supplements are most effective in the prevention and treatment of sarcopenia, thus, more prospective cohort studies are needed.
Vitamin D can improve muscle mass and function and is beneficial for patients with sarcopenia.Citation138 Therefore, adequate vitamin D intake is recommended for elderly diabetic patients, especially those with sarcopenia.Citation139 However, the optimal dose and frequency of administration, as well as the duration of treatment, remain unclear.Citation4 In a study conducted on older adults with sarcopenia, it was found that combining exercise with daily intake of whey protein (22 g), essential amino acids (11 g, including 4 g leucine) and vitamin D (100 IU) resulted in a gain of almost 2 kg greater in lean mass compared to exercise alone. Additionally, the group that received the combination treatment showed significant improvements in hand grip strength and a reduction in CRP levels. In addition, it was reported that low intake of vitamins B1 and B12 increased the chance of sarcopenia in elderly patients with T2D.Citation8 Further research is needed on whether vitamin B1 and B12 supplementation improves metabolic and muscle health issues. Inversely, the results of vitamin A and E in different studies are still controversial, where the sample size needs to be further expanded. Interestingly, reduced speed of eating was associated with an increased risk of sarcopenia in aged T2D patients.Citation140 Recent studies have shown potential benefits of low-dose lithium (Lithium) in T2D and sarcopenia,Citation141 while specific mechanisms need further study.
Although sarcopenia and T2DM are prevalent in the modern population, the current evidence for nutritional intervention is not consistent. The complement of evidences in the future research and practice will assist successful management of sarcopenia and diabetes.
Pharmacotherapy
There are currently no approved specific drugs for the therapy of sarcopenia. Possible therapeutic targets include hormonal interventions such as transdermal testosterone gelsCitation142 and selective androgen receptor modulators (SARMs).Citation143 Transdermal testosterone gel increases the blood concentration of androgens, which has a favorable anabolic effect on skeletal muscle.Citation144 However, adverse side effects exist, such as dyslipidemia, benign prostatic hypertrophy and uterine hyper-proliferation.Citation145 Androgens are important for building and maintaining skeletal muscle, and due to their anabolic effects on muscle, they are considered superior in the potential treatment of sarcopenia.Citation146 SARMs have an acceptable safety profile and positive effects on body composition and function in clinical trials.Citation143,Citation146 Hepatotoxicity and off-target effects on genitalia were major treatment-limiting side effects.Citation147 Nonetheless, even if drugs are approved for the treatment of sarcopenia in the future, lifestyle modification will likely remain the mainstay for T2DM and sarcopenia.
Antidiabetic drugs may affect muscle mass; however, the mechanism for most of them on sarcopenia is still unknown. Metformin is a drug widely used in the treatment of T2DM, and different studies have also shown it has positive effects on muscle mass and muscle strength.Citation148,Citation149 However, in the elderly, inconveniences such as digestive intolerance, dysgeusia, hyperoxia, and vitamin b12 deficiency may hamper their prospect widely utilization in these populations.Citation150 Sulfonylureas predispose to muscle atrophy and should be avoided in patients with sarcopenia or in patients with diabetes at risk of developing sarcopenia.Citation151,Citation152 Glinides have a similar mechanism of action to sulfonylureasCitation153 and should also be avoided. Thiazolidinediones act as insulin sensitizers, and some studies have shown that thiazolidinediones are beneficial for muscle performance in diabetics. Pioglitazone improves skeletal muscle energy expenditure by reducing intramuscular lipid content and improving fatty acid metabolism,Citation154 but increases risk of fractures and decompensated heart failure.Citation155 Dipeptidyl peptidase-4 inhibitors (DPP-4) have positive effects on muscle mass and glycemic control,Citation156 which may be related to the ability to enhance the action of GLP-1 or inhibit DPP-4 activity itself, or both.Citation149 DPP-4 inhibitors have few side effects and exhibit minimal risk of hypoglycemia.Citation157 Thus, such drugs may serve as a promising approach for T2DM with sarcopenia. Glucagon-like peptide 1 agonists (GLP-1RA) have anti-inflammatory and antioxidant properties, and the mechanism of their effects on skeletal muscle is still a matter of debate.Citation153 Sodium-Glucose Cotransporter 2 Inhibitors (SGLT-2i) have been shown to be good therapeutic options for the treatment of aged T2D patients. In animal experiments, SGLT2i not only improved hyperglycemia but also improved fatty acid metabolism in muscle, thereby preventing muscle atrophy.Citation158 Regardless of this proposed preclinical studied merit, the effects of SGLT-2i on muscle are still unclear in limited follow-up duration of clinical trials.Citation159 Insulin is actually the most powerful hypoglycemic drug known so far. In limited studies, it claimed that long-term application of insulin therapy may negatively affect muscles encompassing mechanism such as inflammation, oxidative stress and increased ACEs, while its effect on bodyweight may be favored for low BMI patients. Therefore, there is currently no homogenous evidence that insulin therapy is beneficial for sarcopenia in older adultsCitation153 ().
Table 1 Effects of Drug Therapy on Muscle and Adverse Side Effects in Diabetics
Future Directions
Mitochondrial-targeted antioxidants are another potential therapy to reverse oxidative dysfunction, lower ATP levels and restore protein synthesis and muscle mass.Citation160 In addition, myostatin (MYO) plays a powerful role as a negative muscle growth regulator. This is supported by studies showing that MYO gene knockout mice exhibit abnormal muscle hypertrophy and MYO overexpression can lead to severe muscle atrophy.Citation161 In preclinical models, follistatin, a myostatin antagonist, was associated with reduced myostatin expression and increased muscle mass and protein synthesis.Citation162 Endothelin-1 (ET-1) may play a critical role in mediating myopathy and adipose inflammation through the release of IL-6, TNF-α, or adipokines, visfatin, which involve PI3K/Akt/miR-let-7g-5p pathways in elderly individuals with diabetes.Citation163 These findings can promote conceptualization and development of novel approaches or strategies for sarcopenia and with its related diseases in the future.
Conclusion
T2DM and sarcopenia are interrelated and entangled each other. Aging, insulin resistance, inflammation, oxidative stress, accumulation of AGEs, ectopic fat, vitamins and other factors could obviously affect muscle health. Impaired muscle health may also contribute to the development and progression of T2DM. Therefore, specific exercise methods and protein-rich nutrition combination-specific plans should be adopted, and appropriate hypoglycemic drugs selected to be individualized. The evidences backup the most effective and feasible interventions for patients complicated with both diseases are relatively sparse, and important directions in the future needed including intensive exploring underlying comprehensive cellular and molecular pathways and their interrelationships for two diseases, the advancement in developing biomarkers, the accuracy of diagnosis throughout whole life-span. Only by solving these techniques and knowledge limits, could prevention and treatment of sarcopenia and T2DM pierce the chaos of cognitive and practice cave.
Disclosure
The authors declare no conflicts of interest in this work.
Additional information
Funding
References
- Forouhi NG, Wareham NJ. Epidemiology of diabetes. Medicine. 2014;42(12):698–702. doi:10.1016/j.mpmed.2014.09.007
- Cole JB, Florez JC. Genetics of diabetes mellitus and diabetes complications. Nat Rev Nephrol. 2020;16(7):377–390. doi:10.1038/s41581-020-0278-5
- Kim TN, Park MS, Yang SJ, et al. Prevalence and determinant factors of sarcopenia in patients with type 2 diabetes: the Korean Sarcopenic Obesity Study (KSOS). Diabetes Care. 2010;33(7):1497–1499. doi:10.2337/dc09-2310
- Izzo A, Massimino E, Riccardi G, et al. A narrative review on sarcopenia in type 2 diabetes mellitus: prevalence and associated factors. Nutrients. 2021;13(1):183. doi:10.3390/nu13010183
- Liccini A, Malmstrom TK. Frailty and sarcopenia as predictors of adverse health outcomes in persons with diabetes mellitus. J Am Med Dir Assoc. 2016;17(9):846–851. doi:10.1016/j.jamda.2016.07.007
- Kawada T. Mortality risk of sarcopenia in older subjects. J Am Med Dir Assoc. 2021;22(9):1883. doi:10.1016/j.jamda.2021.04.011
- Xu J, Wan CS, Ktoris K, et al. Sarcopenia is associated with mortality in adults: a systematic review and meta-analysis. Gerontology. 2022;68(4):361–376. doi:10.1159/000517099
- Takahashi F, Hashimoto Y, Kaji A, et al. Sarcopenia is associated with a risk of mortality in people with type 2 diabetes mellitus. Front Endocrinol (Lausanne). 2021;12:783363. doi:10.3389/fendo.2021.783363
- Chen LK, Liu LK, Woo J, et al. Sarcopenia in Asia: consensus report of the Asian Working Group for sarcopenia. J Am Med Dir Assoc. 2014;15(2):95–101. doi:10.1016/j.jamda.2013.11.025
- Cruz-Jentoft AJ, Sayer AA. Sarcopenia. Lancet. 2019;393(10191):2636–2646. doi:10.1016/S0140-6736(19)31138-9
- Tsekoura M, Kastrinis A, Katsoulaki M, et al. Sarcopenia and its impact on quality of life. Adv Exp Med Biol. 2017;987:213–218.
- Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(4):601. doi:10.1093/ageing/afz046
- Wu X, Li X, Xu M, et al. Sarcopenia prevalence and associated factors among older Chinese population: findings from the China Health and Retirement Longitudinal Study. PLoS One. 2021;16(3):e0247617. doi:10.1371/journal.pone.0247617
- Ethgen O, Beaudart C, Buckinx F, et al. The Future prevalence of sarcopenia in Europe: a claim for public health action. Calcif Tissue Int. 2017;100(3):229–234. doi:10.1007/s00223-016-0220-9
- Wen CY, Lien AS, Jiang YD. Sarcopenia in elderly diabetes. J Diabetes Investig. 2022;13(6):944–946. doi:10.1111/jdi.13752
- Huang S, Xiang C, Song Y. Identification of the shared gene signatures and pathways between sarcopenia and type 2 diabetes mellitus. PLoS One. 2022;17(3):e0265221. doi:10.1371/journal.pone.0265221
- Mesinovic J, Zengin A, De Courten B, et al. Sarcopenia and type 2 diabetes mellitus: a bidirectional relationship. Diabetes Metab Syndr Obes. 2019;12:1057–1072. doi:10.2147/DMSO.S186600
- Lin JA, Hou JD, Wu SY. Effect of sarcopenia on mortality in type 2 diabetes: a long-term follow-up propensity score-matched diabetes cohort study. J Clin Med. 2022;11(15):4424. doi:10.3390/jcm11154424
- Ghafouri-Fard S, Khoshbakht T, Hussen BM, et al. Emerging role of non-coding RNAs in senescence. Front Cell Dev Biol. 2022;10:869011. doi:10.3389/fcell.2022.869011
- Nair KS. Aging muscle. Am J Clin Nutr. 2005;81(5):953–963. doi:10.1093/ajcn/81.5.953
- Volpi E, Nazemi R, Fujita S. Muscle tissue changes with aging. Curr Opin Clin Nutr Metab Care. 2004;7(4):405–410. doi:10.1097/01.mco.0000134362.76653.b2
- Mccormick R, Vasilaki A. Age-related changes in skeletal muscle: changes to life-style as a therapy. Biogerontology. 2018;19(6):519–536. doi:10.1007/s10522-018-9775-3
- Son JW, Lee SS, Kim SR, et al. Low muscle mass and risk of type 2 diabetes in middle-aged and older adults: findings from the KoGES. Diabetologia. 2017;60(5):865–872. doi:10.1007/s00125-016-4196-9
- Wu H, Liu M, Chi VTQ, et al. Handgrip strength is inversely associated with metabolic syndrome and its separate components in middle aged and older adults: a large-scale population-based study. Metabolism. 2019;93:61–67. doi:10.1016/j.metabol.2019.01.011
- Mitchell WK, Williams J, Atherton P, et al. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front Physiol. 2012;3:260. doi:10.3389/fphys.2012.00260
- Kelly TL, Wilson KE, Heymsfield SB. Dual energy X-ray absorptiometry body composition reference values from NHANES. PLoS One. 2009;4(9):e7038. doi:10.1371/journal.pone.0007038
- Zamboni M, Gattazzo S, Rossi AP. Myosteatosis: a relevant, yet poorly explored element of sarcopenia. Eur Geriatr Med. 2019;10(1):5–6. doi:10.1007/s41999-018-0134-3
- Park SW, Goodpaster BH, Strotmeyer ES, et al. Decreased muscle strength and quality in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes. 2006;55(6):1813–1818. doi:10.2337/db05-1183
- Par A, Hegyi JP, Vancsa S, et al. Sarcopenia - 2021: pathophysiology, diagnosis, therapy. Orv Hetil. 2021;162(1):3–12. doi:10.1556/650.2021.32015
- Walsh K. Adipokines, myokines and cardiovascular disease. Circ J. 2009;73(1):13–18. doi:10.1253/circj.CJ-08-0961
- Beyer I, Mets T, Bautmans I. Chronic low-grade inflammation and age-related sarcopenia. Curr Opin Clin Nutr Metab Care. 2012;15(1):12–22. doi:10.1097/MCO.0b013e32834dd297
- Proctor MJ, Mcmillan DC, Horgan PG, et al. Systemic inflammation predicts all-cause mortality: a Glasgow inflammation outcome study. PLoS One. 2015;10(3):e0116206. doi:10.1371/journal.pone.0116206
- Papadopoulou SK. Sarcopenia: a contemporary health problem among older adult populations. Nutrients. 2020;12(5):1293. doi:10.3390/nu12051293
- Sinha-Hikim I, Cornford M, Gaytan H, et al. Effects of testosterone supplementation on skeletal muscle fiber hypertrophy and satellite cells in community-dwelling older men. J Clin Endocrinol Metab. 2006;91(8):3024–3033. doi:10.1210/jc.2006-0357
- Jang HC. Sarcopenia, frailty, and diabetes in older adults. Diabetes Metab J. 2016;40(3):182–189. doi:10.4093/dmj.2016.40.3.182
- Goodpaster BH, Park SW, Harris TB, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol a Biol Sci Med Sci. 2006;61(10):1059–1064. doi:10.1093/gerona/61.10.1059
- Meier NF, Lee DC. Physical activity and sarcopenia in older adults. Aging Clin Exp Res. 2020;32(9):1675–1687. doi:10.1007/s40520-019-01371-8
- Norman AW, Bouillon R. Vitamin D nutritional policy needs a vision for the future. Exp Biol Med (Maywood). 2010;235(9):1034–1045. doi:10.1258/ebm.2010.010014
- Dang M, Shore-Lorenti C, Mcmillan LB, et al. Associations of serum 25-hydroxyvitamin D with physical performance and bone health in overweight and obese older adults. Int J Environ Res Public Health. 2019;16(3):509. doi:10.3390/ijerph16030509
- Mirhosseini N, Vatanparast H, Mazidi M, et al. Vitamin D supplementation, glycemic control, and insulin resistance in prediabetics: a meta-analysis. J Endocr Soc. 2018;2(7):687–709. doi:10.1210/js.2017-00472
- Lucato P, Solmi M, Maggi S, et al. Low vitamin D levels increase the risk of type 2 diabetes in older adults: a systematic review and meta-analysis. Maturitas. 2017;100:8–15. doi:10.1016/j.maturitas.2017.02.016
- Hirani V, Cumming RG, Naganathan V, et al. Longitudinal associations between vitamin d metabolites and sarcopenia in older Australian men: the concord health and aging in men project. J Gerontol a Biol Sci Med Sci. 2017;73(1):131–138. doi:10.1093/gerona/glx086
- Uchitomi R, Oyabu M, Kamei Y. Vitamin D and sarcopenia: potential of vitamin D supplementation in sarcopenia prevention and treatment. Nutrients. 2020;12(10):3189. doi:10.3390/nu12103189
- Remelli F, Vitali A, Zurlo A, et al. Vitamin D deficiency and sarcopenia in older persons. Nutrients. 2019;11(12):2861. doi:10.3390/nu11122861
- Kalyani RR, Metter EJ, Egan J, et al. Hyperglycemia predicts persistently lower muscle strength with aging. Diabetes Care. 2015;38(1):82–90. doi:10.2337/dc14-1166
- US Department of Health and Human Services. The Health Consequences of Smoking-50 Years of Progress: A Report of the Surgeon General. Atlanta (GA): US Department of Health and Human Services; 2014.
- Maddatu J, Anderson-baucum E, Evans-Molina C. Smoking and the risk of type 2 diabetes. Transl Res. 2017;184:101–107. doi:10.1016/j.trsl.2017.02.004
- Larsson L, Orlander J. Skeletal muscle morphology, metabolism and function in smokers and non-smokers. A study on smoking-discordant monozygous twins. Acta Physiol Scand. 1984;120(3):343–352. doi:10.1111/j.1748-1716.1984.tb07394.x
- Wust RC, Morse CI, De Haan A, et al. Skeletal muscle properties and fatigue resistance in relation to smoking history. Eur J Appl Physiol. 2008;104(1):103–110. doi:10.1007/s00421-008-0792-9
- Degens H, Veerkamp JH. Changes in oxidative capacity and fatigue resistance in skeletal muscle. Int J Biochem. 1994;26(7):871–878. doi:10.1016/0020-711X(94)90079-5
- Orlander J, Kiessling KH, Larsson L. Skeletal muscle metabolism, morphology and function in sedentary smokers and nonsmokers. Acta Physiol Scand. 1979;107(1):39–46. doi:10.1111/j.1748-1716.1979.tb06440.x
- Degens H, Gayan-Ramirez G, Van Hees HW. Smoking-induced skeletal muscle dysfunction: from evidence to mechanisms. Am J Respir Crit Care Med. 2015;191(6):620–625. doi:10.1164/rccm.201410-1830PP
- Yaribeygi H, Farrokhi FR, Butler AE, et al. Insulin resistance: review of the underlying molecular mechanisms. J Cell Physiol. 2019;234(6):8152–8161. doi:10.1002/jcp.27603
- Argyropoulou D, Geladas ND, Nomikos T, et al. Exercise and nutrition strategies for combating sarcopenia and type 2 diabetes mellitus in older adults. J Funct Morphol Kinesiol. 2022;7(2). doi:10.3390/jfmk7020048
- Giha HA, Alamin OAO, Sater MS. Diabetic sarcopenia: metabolic and molecular appraisal. Acta Diabetol. 2022;59(8):989–1000. doi:10.1007/s00592-022-01883-2
- Russell ST, Rajani S, Dhadda RS, et al. Mechanism of induction of muscle protein loss by hyperglycaemia. Exp Cell Res. 2009;315(1):16–25. doi:10.1016/j.yexcr.2008.10.002
- Purnamasari D, Tetrasiwi EN, Kartiko GJ, et al. Sarcopenia and chronic complications of type 2 diabetes mellitus. Rev Diabet Stud. 2022;18(3):157–165. doi:10.1900/RDS.2022.18.157
- Goossens GH, Blaak EE, Theunissen R, et al. Expression of NLRP3 inflammasome and T cell population markers in adipose tissue are associated with insulin resistance and impaired glucose metabolism in humans. Mol Immunol. 2012;50(3):142–149. doi:10.1016/j.molimm.2012.01.005
- Mcbride MJ, Foley KP, D’souza DM, et al. The NLRP3 inflammasome contributes to sarcopenia and lower muscle glycolytic potential in old mice. Am J Physiol Endocrinol Metab. 2017;313(2):E222–E232. doi:10.1152/ajpendo.00060.2017
- Hirata Y, Nomura K, Senga Y, et al. Hyperglycemia induces skeletal muscle atrophy via a WWP1/KLF15 axis. JCI Insight. 2019;4(4). doi:10.1172/jci.insight.124952
- Dasarathy S, Merli M. Sarcopenia from mechanism to diagnosis and treatment in liver disease. J Hepatol. 2016;65(6):1232–1244. doi:10.1016/j.jhep.2016.07.040
- Glass DJ. Skeletal muscle hypertrophy and atrophy signaling pathways. Int J Biochem Cell Biol. 2005;37(10):1974–1984. doi:10.1016/j.biocel.2005.04.018
- Yoshida T, Delafontaine P. Mechanisms of IGF-1-mediated regulation of skeletal muscle hypertrophy and atrophy. Cells. 2020;9(9):1970. doi:10.3390/cells9091970
- Lawrence JC. mTOR-dependent control of skeletal muscle protein synthesis. Int J Sport Nutr Exerc Metab. 2001;11(Suppl):S177–S185. doi:10.1123/ijsnem.11.s1.s177
- Bassil MS, Gougeon R. Muscle protein anabolism in type 2 diabetes. Curr Opin Clin Nutr Metab Care. 2013;16(1):83–88. doi:10.1097/MCO.0b013e32835a88ee
- Kalyani RR, Corriere M, Ferrucci L. Age-related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014;2(10):819–829. doi:10.1016/S2213-8587(14)70034-8
- Qiu J, Thapaliya S, Runkana A, et al. Hyperammonemia in cirrhosis induces transcriptional regulation of myostatin by an NF-kappaB-mediated mechanism. Proc Natl Acad Sci U S A. 2013;110(45):18162–18167. doi:10.1073/pnas.1317049110
- Wang XH, Mitch WE. Mechanisms of muscle wasting in chronic kidney disease. Nat Rev Nephrol. 2014;10(9):504–516. doi:10.1038/nrneph.2014.112
- Bataille S, Chauveau P, Fouque D, et al. Myostatin and muscle atrophy during chronic kidney disease. Nephrol Dial Transplant. 2021;36(11):1986–1993. doi:10.1093/ndt/gfaa129
- Du J, Wang X, Miereles C, et al. Activation of caspase-3 is an initial step triggering accelerated muscle proteolysis in catabolic conditions. J Clin Invest. 2004;113(1):115–123. doi:10.1172/JCI18330
- Baczek J, Silkiewicz M, Wojszel ZB. Myostatin as a biomarker of muscle wasting and other pathologies-state of the art and knowledge gaps. Nutrients. 2020;12(8):2401. doi:10.3390/nu12082401
- Buch A, Carmeli E, Boker LK, et al. Muscle function and fat content in relation to sarcopenia, obesity and frailty of old age--an overview. Exp Gerontol. 2016;76:25–32. doi:10.1016/j.exger.2016.01.008
- Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98–107. doi:10.1038/nri2925
- Perry BD, Caldow MK, Brennan-Speranza TC, et al. Muscle atrophy in patients with type 2 diabetes mellitus: roles of inflammatory pathways, physical activity and exercise. Exerc Immunol Rev. 2016;22:94–109.
- Zhang X, Li H, He M, et al. Immune system and sarcopenia: presented relationship and future perspective. Exp Gerontol. 2022;164:111823. doi:10.1016/j.exger.2022.111823
- Goyal R, Faizy AF, Siddiqui SS, et al. Evaluation of TNF-alpha and IL-6 levels in obese and non-obese diabetics: pre- and postinsulin effects. N Am J Med Sci. 2012;4(4):180–184. doi:10.4103/1947-2714.94944
- King DE, Mainous AG, Buchanan TA, et al. C-reactive protein and glycemic control in adults with diabetes. Diabetes Care. 2003;26(5):1535–1539. doi:10.2337/diacare.26.5.1535
- Vozarova B, Weyer C, Hanson K, et al. Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes Res. 2001;9(7):414–417. doi:10.1038/oby.2001.54
- Visser M, Pahor M, Taaffe DR, et al. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: the Health ABC Study. J Gerontol a Biol Sci Med Sci. 2002;57(5):M326–332. doi:10.1093/gerona/57.5.M326
- Rong YD, Bian AL, Hu HY, et al. Study on relationship between elderly sarcopenia and inflammatory cytokine IL-6, anti-inflammatory cytokine IL-10. BMC Geriatr. 2018;18(1):308. doi:10.1186/s12877-018-1007-9
- Haddad F, Zaldivar F, Cooper DM, et al. IL-6-induced skeletal muscle atrophy. J Appl Physiol. 2005;98(3):911–917. doi:10.1152/japplphysiol.01026.2004
- Park SW, Goodpaster BH, Strotmeyer ES, et al. Accelerated loss of skeletal muscle strength in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes Care. 2007;30(6):1507–1512. doi:10.2337/dc06-2537
- Ma JF, Sanchez BJ, Hall DT, et al. STAT 3 promotes IFN γ/ TNF α-induced muscle wasting in an NF -κB-dependent and IL −6-independent manner. EMBO Mol Med. 2017;9(5):622–637. doi:10.15252/emmm.201607052
- Ip WKE, Hoshi N, Shouval DS, et al. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science. 2017;356(6337):513–519. doi:10.1126/science.aal3535
- Omura T, Araki A. Skeletal muscle as a treatment target for older adults with diabetes mellitus: the importance of a multimodal intervention based on functional category. Geriatr Gerontol Int. 2022;22(2):110–120. doi:10.1111/ggi.14339
- Houstis N, Rosen ED, Lander ES. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature. 2006;440(7086):944–948. doi:10.1038/nature04634
- Bowen TS, Schuler G, Adams V. Skeletal muscle wasting in cachexia and sarcopenia: molecular pathophysiology and impact of exercise training. J Cachexia Sarcopenia Muscle. 2015;6(3):197–207. doi:10.1002/jcsm.12043
- Aragno M, Mastrocola R, Catalano MG, et al. Oxidative stress impairs skeletal muscle repair in diabetic rats. Diabetes. 2004;53(4):1082–1088. doi:10.2337/diabetes.53.4.1082
- Scicchitano BM, Pelosi L, Sica G, et al. The physiopathologic role of oxidative stress in skeletal muscle. Mech Ageing Dev. 2018;170:37–44. doi:10.1016/j.mad.2017.08.009
- Zhang L, Kimball SR, Jefferson LS, et al. Hydrogen peroxide impairs insulin-stimulated assembly of mTORC1. Free Radic Biol Med. 2009;46(11):1500–1509. doi:10.1016/j.freeradbiomed.2009.03.001
- Short KR, Bigelow ML, Kahl J, et al. Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A. 2005;102(15):5618–5623. doi:10.1073/pnas.0501559102
- Abbatecola AM, Paolisso G, Fattoretti P, et al. Discovering pathways of sarcopenia in older adults: a role for insulin resistance on mitochondria dysfunction. J Nutr Health Aging. 2011;15(10):890–895. doi:10.1007/s12603-011-0366-0
- Conley KE, Jubrias SA, Esselman PC. Oxidative capacity and ageing in human muscle. J Physiol. 2000;526(Pt 1):203–210. doi:10.1111/j.1469-7793.2000.t01-1-00203.x
- Bua E, Johnson J, Herbst A, et al. Mitochondrial DNA-deletion mutations accumulate intracellularly to detrimental levels in aged human skeletal muscle fibers. Am J Hum Genet. 2006;79(3):469–480. doi:10.1086/507132
- Chen H, Ma J, Liu A, et al. The association between sarcopenia and fracture in middle-aged and elderly people: a systematic review and meta-analysis of cohort studies. Injury. 2020;51(4):804–811. doi:10.1016/j.injury.2020.02.072
- Hyatt HW, Powers SK. The role of calpains in skeletal muscle remodeling with exercise and inactivity-induced atrophy. Int J Sports Med. 2020;41(14):994–1008. doi:10.1055/a-1199-7662
- Huang J, Zhu X. The molecular mechanisms of calpains action on skeletal muscle atrophy. Physiol Res. 2016;65(4):547–560. doi:10.33549/physiolres.933087
- Suzuki A, Yabu A, Nakamura H. Advanced glycation end products in musculoskeletal system and disorders. Methods. 2022;203:179–186. doi:10.1016/j.ymeth.2020.09.012
- Haus JM, Carrithers JA, Trappe SW, et al. Collagen, cross-linking, and advanced glycation end products in aging human skeletal muscle. J Appl Physiol. 2007;103(6):2068–2076. doi:10.1152/japplphysiol.00670.2007
- Forbes JM, Sourris KC, De Courten MP, et al. Advanced glycation end products (AGEs) are cross-sectionally associated with insulin secretion in healthy subjects. Amino Acids. 2014;46(2):321–326. doi:10.1007/s00726-013-1542-9
- Moon SS. Low skeletal muscle mass is associated with insulin resistance, diabetes, and metabolic syndrome in the Korean population: the Korea National Health and Nutrition Examination Survey (KNHANES) 2009–2010. Endocr J. 2014;61(1):61–70. doi:10.1507/endocrj.EJ13-0244
- Cleasby ME, Jamieson PM, Atherton PJ. Insulin resistance and sarcopenia: mechanistic links between common co-morbidities. J Endocrinol. 2016;229(2):R67–81. doi:10.1530/JOE-15-0533
- Waqas K, Chen J, Trajanoska K, et al. Skin autofluorescence, a noninvasive biomarker for advanced glycation end-products, is associated with sarcopenia. J Clin Endocrinol Metab. 2022;107(2):e793–e803. doi:10.1210/clinem/dgab632
- Boettcher M, Machann J, Stefan N, et al. Intermuscular adipose tissue (IMAT): association with other adipose tissue compartments and insulin sensitivity. J Magn Reson Imaging. 2009;29(6):1340–1345. doi:10.1002/jmri.21754
- Scott D, Shore-Lorenti C, Mcmillan LB, et al. Calf muscle density is independently associated with physical function in overweight and obese older adults. J Musculoskelet Neuronal Interact. 2018;18(1):9–17.
- Kahn SE, Hull RL, Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444(7121):840–846. doi:10.1038/nature05482
- Pellegrinelli V, Rouault C, Rodriguez-Cuenca S, et al. Human Adipocytes Induce Inflammation and Atrophy in Muscle Cells During Obesity. Diabetes. 2015;64(9):3121–3134. doi:10.2337/db14-0796
- Bittel DC, Bittel AJ, Tuttle LJ, et al. Adipose tissue content, muscle performance and physical function in obese adults with type 2 diabetes mellitus and peripheral neuropathy. J Diabetes Complications. 2015;29(2):250–257. doi:10.1016/j.jdiacomp.2014.11.003
- Meex RCR, Blaak EE, Van Loon LJC. Lipotoxicity plays a key role in the development of both insulin resistance and muscle atrophy in patients with type 2 diabetes. Obes Rev. 2019;20(9):1205–1217. doi:10.1111/obr.12862
- Jimenez-Gutierrez GE, Martínez-Gómez LE, Martínez-Armenta C, et al. Molecular mechanisms of inflammation in sarcopenia: diagnosis and therapeutic update. Cells. 2022;11(15):2359. doi:10.3390/cells11152359
- Csapo R, Malis V, Sinha U, et al. Age-associated differences in triceps surae muscle composition and strength - an MRI-based cross-sectional comparison of contractile, adipose and connective tissue. BMC Musculoskelet Disord. 2014;15:209. doi:10.1186/1471-2474-15-209
- Ticinesi A, Nouvenne A, Cerundolo N, et al. Gut microbiota, muscle mass and function in aging: a focus on physical frailty and sarcopenia. Nutrients. 2019;11(7):1633. doi:10.3390/nu11071633
- Bouter KE, Van Raalte DH, Groen AK, et al. Role of the gut microbiome in the pathogenesis of obesity and obesity-related metabolic dysfunction. Gastroenterology. 2017;152(7):1671–1678. doi:10.1053/j.gastro.2016.12.048
- Utzschneider KM, Kratz M, Damman CJ, et al. Mechanisms linking the gut microbiome and glucose metabolism. J Clin Endocrinol Metab. 2016;101(4):1445–1454. doi:10.1210/jc.2015-4251
- Allin KH, Tremaroli V, Caesar R, et al. Aberrant intestinal microbiota in individuals with prediabetes. Diabetologia. 2018;61(4):810–820. doi:10.1007/s00125-018-4550-1
- Qin J, Li Y, Cai Z, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55–60. doi:10.1038/nature11450
- Kootte RS, Levin E, Salojarvi J, et al. Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 2017;26(4):611–619 e616. doi:10.1016/j.cmet.2017.09.008
- Strasser B, Wolters M, Weyh C, et al. The effects of lifestyle and diet on gut microbiota composition, inflammation and muscle performance in our aging society. Nutrients. 2021;13(6):2045. doi:10.3390/nu13062045
- Park S, Yuan H, Zhang T, et al. Long-term silk peptide intake promotes skeletal muscle mass, reduces inflammation, and modulates gut microbiota in middle-aged female rats. Biomed Pharmacother. 2021;137:111415. doi:10.1016/j.biopha.2021.111415
- Houghton MJ, Kerimi A, Mouly V, et al. Gut microbiome catabolites as novel modulators of muscle cell glucose metabolism. FASEB J. 2019;33(2):1887–1898. doi:10.1096/fj.201801209R
- Van Krimpen SJ, Jansen FAC, Ottenheim VL, et al. The effects of pro-, pre-, and synbiotics on muscle wasting, a systematic review-gut permeability as potential treatment target. Nutrients. 2021;13(4):1115. doi:10.3390/nu13041115
- Liu Y, Wang Y, Ni Y, et al. Gut microbiome fermentation determines the efficacy of exercise for diabetes prevention. Cell Metab. 2020;31(1):77–91 e75. doi:10.1016/j.cmet.2019.11.001
- Burini RC, Anderson E, Durstine JL, et al. Inflammation, physical activity, and chronic disease: an evolutionary perspective. Sports Med Health Sci. 2020;2(1):1–6. doi:10.1016/j.smhs.2020.03.004
- Liu HW, Chang SJ. Moderate exercise suppresses NF-kappaB Signaling and activates the SIRT1-AMPK-PGC1alpha axis to attenuate muscle loss in diabetic db/db mice. Front Physiol. 2018;9:636. doi:10.3389/fphys.2018.00636
- Dent E, Morley JE, Cruz-Jentoft AJ, et al. International clinical practice guidelines for sarcopenia (ICFSR): screening, diagnosis and management. J Nutr Health Aging. 2018;22(10):1148–1161. doi:10.1007/s12603-018-1139-9
- Dhillon RJ, Hasni S. Pathogenesis and management of sarcopenia. Clin Geriatr Med. 2017;33(1):17–26. doi:10.1016/j.cger.2016.08.002
- Reidy PT, Mahmassani ZS, Mckenzie AI, et al. Influence of exercise training on skeletal muscle insulin resistance in aging: spotlight on muscle ceramides. Int J Mol Sci. 2020;21(4):1514. doi:10.3390/ijms21041514
- Kimura T, Okamura T, Iwai K, et al. Japanese radio calisthenics prevents the reduction of skeletal muscle mass volume in people with type 2 diabetes. BMJ Open Diabetes Res Care. 2020;8(1):e001027. doi:10.1136/bmjdrc-2019-001027
- Zhao D, Shi W, Bi L, et al. Effect of short-term acute moderate-intensity resistance exercise on blood glucose in older patients with type 2 diabetes mellitus and sarcopenia. Geriatr Gerontol Int. 2022;22(8):653–659. doi:10.1111/ggi.14437
- Zhang XZ, Xie WQ, Chen L, et al. Blood flow restriction training for the intervention of sarcopenia: current stage and future perspective. Front Med. 2022;9:894996. doi:10.3389/fmed.2022.894996
- Shang X, Scott D, Hodge AM, et al. Dietary protein intake and risk of type 2 diabetes: results from the Melbourne Collaborative Cohort Study and a meta-analysis of prospective studies. Am J Clin Nutr. 2016;104(5):1352–1365. doi:10.3945/ajcn.116.140954
- Manders RJ, Little JP, Forbes SC, et al. Insulinotropic and muscle protein synthetic effects of branched-chain amino acids: potential therapy for type 2 diabetes and sarcopenia. Nutrients. 2012;4(11):1664–1678. doi:10.3390/nu4111664
- Low S, Wang J, Moh A, et al. Amino acid profile of skeletal muscle loss in type 2 diabetes: results from a 7-year longitudinal study in asians. Diabetes Res Clin Pract. 2022;186:109803. doi:10.1016/j.diabres.2022.109803
- Hamarsland H, Nordengen AL, Nyvik Aas S, et al. Native whey protein with high levels of leucine results in similar post-exercise muscular anabolic responses as regular whey protein: a randomized controlled trial. J Int Soc Sports Nutr. 2017;14:43. doi:10.1186/s12970-017-0202-y
- Jayanama K, Theou O, Godin J, et al. Association of fatty acid consumption with frailty and mortality among middle-aged and older adults. Nutrition. 2020;70:110610. doi:10.1016/j.nut.2019.110610
- Smith GI, Atherton P, Reeds DN, et al. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: a randomized controlled trial. Am J Clin Nutr. 2011;93(2):402–412. doi:10.3945/ajcn.110.005611
- Kiecolt-glaser JK, Belury MA, Andridge R, et al. Omega-3 supplementation lowers inflammation in healthy middle-aged and older adults: a randomized controlled trial. Brain Behav Immun. 2012;26(6):988–995. doi:10.1016/j.bbi.2012.05.011
- Ganapathy A, Nieves JW. Nutrition and sarcopenia-what do we know? Nutrients. 2020;12(6):1755. doi:10.3390/nu12061755
- Tamura Y, Omura T, Toyoshima K, et al. Nutrition management in older adults with diabetes: a review on the importance of shifting prevention strategies from metabolic syndrome to frailty. Nutrients. 2020;12(11):3367. doi:10.3390/nu12113367
- Hashimoto Y, Takahashi F, Kaji A, et al. Eating speed is associated with the presence of sarcopenia in older patients with type 2 diabetes: a cross-sectional study of the KAMOGAWA-DM cohort. Nutrients. 2022;14(4):759. doi:10.3390/nu14040759
- Hamstra SI, Roy BD, Tiidus P, et al. Beyond its psychiatric use: the benefits of low dose lithium supplementation. Curr Neuropharmacol. 2022. doi:10.2174/1570159X20666220302151224
- Atkinson RA, Srinivas-Shankar U, Roberts SA, et al. Effects of testosterone on skeletal muscle architecture in intermediate-frail and frail elderly men. J Gerontol A Biol Sci Med Sci. 2010;65(11):1215–1219. doi:10.1093/gerona/glq118
- Papanicolaou DA, Ather SN, Zhu H, et al. A phase IIA randomized, placebo-controlled clinical trial to study the efficacy and safety of the selective androgen receptor modulator (SARM), MK-0773 in female participants with sarcopenia. J Nutr Health Aging. 2013;17(6):533–543. doi:10.1007/s12603-013-0335-x
- Neto WK, Gama EF, Rocha LY, et al. Effects of testosterone on lean mass gain in elderly men: systematic review with meta-analysis of controlled and randomized studies. Age. 2015;37(1):9742. doi:10.1007/s11357-014-9742-0
- Narayanan R, Coss CC, Dalton JT. Development of selective androgen receptor modulators (SARMs). Mol Cell Endocrinol. 2018;465:134–142. doi:10.1016/j.mce.2017.06.013
- Dalton JT, Barnette KG, Bohl CE, et al. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled Phase II trial. J Cachexia Sarcopenia Muscle. 2011;2(3):153–161. doi:10.1007/s13539-011-0034-6
- Solomon ZJ, Mirabal JR, Mazur DJ, et al. Selective androgen receptor modulators: current knowledge and clinical applications. Sex Med Rev. 2019;7(1):84–94. doi:10.1016/j.sxmr.2018.09.006
- Wu CN, Tien KJ. The impact of antidiabetic agents on sarcopenia in type 2 diabetes: a literature review. J Diabetes Res. 2020;2020:9368583. doi:10.1155/2020/9368583
- Massimino E, Izzo A, Riccardi G, et al. The impact of glucose-lowering drugs on sarcopenia in type 2 diabetes: current evidence and underlying mechanisms. Cells. 2021;10(8):1958. doi:10.3390/cells10081958
- Aroda VR, Edelstein SL, Goldberg RB, et al. Long-term metformin use and vitamin B12 deficiency in the diabetes prevention program outcomes study. J Clin Endocrinol Metab. 2016;101(4):1754–1761. doi:10.1210/jc.2015-3754
- Cetrone M, Mele A, Tricarico D. Effects of the antidiabetic drugs on the age-related atrophy and sarcopenia associated with diabetes type II. Curr Diabetes Rev. 2014;10(4):231–237. doi:10.2174/1573399810666140918121022
- Mele A, Calzolaro S, Cannone G, et al. Database search of spontaneous reports and pharmacological investigations on the sulfonylureas and glinides-induced atrophy in skeletal muscle. Pharmacol Res Perspect. 2014;2(1):e00028. doi:10.1002/prp2.28
- Sanz-Canovas J, Lopez-Sampalo A, Cobos-Palacios L, et al. Management of type 2 diabetes mellitus in elderly patients with frailty and/or sarcopenia. Int J Environ Res Public Health. 2022;19(14):8677. doi:10.3390/ijerph19148677
- Yokota T, Kinugawa S, Hirabayashi K, et al. Pioglitazone improves whole-body aerobic capacity and skeletal muscle energy metabolism in patients with metabolic syndrome. J Diabetes Investig. 2017;8(4):535–541. doi:10.1111/jdi.12606
- Erdmann E, Charbonnel B, Wilcox RG, et al. Pioglitazone use and heart failure in patients with type 2 diabetes and preexisting cardiovascular disease: data from the PROactive study (PROactive 08). Diabetes Care. 2007;30(11):2773–2778. doi:10.2337/dc07-0717
- Sencan C, Dost FS, Ates Bulut E, et al. DPP4 inhibitors as a potential therapeutic option for sarcopenia: a 6-month follow-up study in diabetic older patients. Exp Gerontol. 2022;164:111832. doi:10.1016/j.exger.2022.111832
- Doucet J, Chacra A, Maheux P, et al. Efficacy and safety of saxagliptin in older patients with type 2 diabetes mellitus. Curr Med Res Opin. 2011;27(4):863–869. doi:10.1185/03007995.2011.554532
- Bamba R, Okamura T, Hashimoto Y, et al. Extracellular lipidome change by an SGLT2 inhibitor, luseogliflozin, contributes to prevent skeletal muscle atrophy in db/db mice. J Cachexia Sarcopenia Muscle. 2022;13(1):574–588. doi:10.1002/jcsm.12814
- Yabe D, Shiki K, Suzaki K, et al. Rationale and design of the EMPA-ELDERLY trial: a randomised, double-blind, placebo-controlled, 52-week clinical trial of the efficacy and safety of the sodium-glucose cotransporter-2 inhibitor empagliflozin in elderly Japanese patients with type 2 diabetes. BMJ Open. 2021;11(4):e045844. doi:10.1136/bmjopen-2020-045844
- Kumar A, Davuluri G, Welch N, et al. Oxidative stress mediates ethanol-induced skeletal muscle mitochondrial dysfunction and dysregulated protein synthesis and autophagy. Free Radic Biol Med. 2019;145:284–299. doi:10.1016/j.freeradbiomed.2019.09.031
- Lee SJ, Mcpherron AC. Regulation of myostatin activity and muscle growth. Proc Natl Acad Sci U S A. 2001;98(16):9306–9311. doi:10.1073/pnas.151270098
- Dasarathy S, Mccullough AJ, Muc S, et al. Sarcopenia associated with portosystemic shunting is reversed by follistatin. J Hepatol. 2011;54(5):915–921. doi:10.1016/j.jhep.2010.08.032
- Tsai CH, Huang PJ, Lee IT, et al. Endothelin-1-mediated miR-let-7g-5p triggers interleukin-6 and TNF-α to cause myopathy and chronic adipose inflammation in elderly patients with diabetes mellitus. Aging. 2022;14(8):3633–3651. doi:10.18632/aging.204034