?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
Solid lipid nanoparticles (SLNs) of amikacin were designed in this study for pulmonary delivery to reduce the dose or its administration intervals leading to reduction of its toxicities especially in long term treatment. Nanoparticles of amikacin were prepared from cholesterol by solvent diffusion technique and homogenization. The size, zeta potential, loading efficiency, and release profile of the nanoparticles were studied. The conventional broth macrodilution tube method was used to determine the minimum inhibitory concentration (MIC) and minimum bacteriostatic concentration (MBC) of amikacin SLNs with respect to Pseudomonas aeruginosa in vitro. To guarantee the stability of desired SLNs, they were lyophilized using cryoprotectants. Results showed that considering the release profile of amikacin from the studied nanocarrier, MIC and MBC of amikacin could be about two times less in SLNs of amikacin compared to the free drug. Therefore, fewer doses of amikacin in SLNs can clear the infection with less adverse effects and more safety. Particle size enlargement after lyophilization of desired SLNs after two months storage was limited in comparison with non-lyophilized particles, 996 and 194 nm, respectively. Zeta potential of lyophilized particles was increased to +17 mV from +4 mV before lyophilization. Storage of particles in higher temperature caused accelerated drug release.
Introduction
Aminoglycosides are the mainstay in the treatment of serious Gram-negative systemic infections. The use of aminoglycosides can be limited because of their adverse effects, mainly oto- and nephrotoxicity which happens in 15%–17% of patients, hearing loss (8%), and vestibular toxicity (3%). Also retina toxicity was observed with gentamycin in vitro.Citation1–Citation3 Controlling aminoglycosides concentration is critical because of their narrow therapeutic range.Citation3 Aminoglycosides are used for treatment of many infections, such as severe pulmonary infections like cystic fibrosis and skin infections, because of their beneficial effects.Citation4 Many studies have been carried out so far to reduce these toxicities such as production of liposomal amikacin dry powder inhaler, intra-tracheal delivery strategy of gentamycin, and thiolated chitosan nanoparticles of amikacin.Citation5–Citation7 Production of solid lipid nanoparticles (SLNs) of amikacin for pulmonary delivery was also reported by Varshosaz et al.Citation8
There are some differences between liposomes and SLNs. By definition, a liposome is a tiny bubble (vesicle) made out of the same material as a cell membrane (lipid bilayer). Liposomes can be filled with drugs and be used to deliver drugs for cancer and other diseases. Membranes are usually made of phospholipids which are molecules that have a head group and a tail group. The head is hydrophilic and the tail, which is made of a long hydrocarbon chain, is hydrophobic. In nature, phospholipids are found in stable membranes composed of two layers (a bilayer). The heads are attracted to water and line up to form a surface facing the water, whereas the tails are repelled by water and line up to form a surface away from it. When membrane phospholipids are disrupted, they can reassemble themselves into tiny spheres, smaller than a normal cell, either as bilayers or monolayers. The bilayer structures are liposomes. The monolayer structures are called micelles. Liposomes can be composed of naturally-derived phospholipids with mixed lipid chains, like egg phosphatidylethanolamine, or of pure surfactant components, like dioleoylphosphatidylethanolamine.Citation9 SLNs as colloidal carrier systems combine the advantages of traditional systems, but avoid some of their major disadvantages. In contrast to liposomes, SLNs do not have a bilayer structure. They may have a matrix structure or encapsulate drugs. They are much more stable than liposomes. Some advantages of SLNs are the possibility of controlling drug release and drug targeting, increased drug stability, high drug payload, possibility of the incorporation of lipophilic and hydrophilic drugs, lack of biotoxicity of the carrier, no problems with respect to large-scale production, sterilization possibility, and good tolerability. However, some of the major drawbacks of SLNs are low drug loading, unpredictable drug release, and the risk of gelation due to polymorphism of the solid lipids. The main ingredients used to produce SLNs include solid lipid(s), emulsifier, and water.
There are many studies on SLNs, even for hydrophilic drugs and peptide delivery. The desired SLNs were made from cholesterol with one layer structure, smaller particle size, simple preparation method, and good stability that candidates makes then for success in scale up in future.Citation10,Citation11
Amikacin is 6-O-(3-amino-3-deoxy-α-1)-glucopyranosyl)- 4-O-(6-amino-6-deoxy-α-D-glucopyranosyl)-N1-[(2,S)- 4-amino- 2-hydroxybutanoyl]-2-deoxy-D-streptamine, a substance obtained from kanamycin A. It has positive charge. Amikacin sulphate having a molar ratio of amikacin to H2SO4 of 1:2 contains the equivalent of not less than 674 μg and not more than 786 μg of amikacin (C22H43 N5O13) per mg, calculated on the dried basis. It has the following molecular formula C22H43N5O132H2SO4 with a molecular weight of 781.75. Aminoglycosides like amikacin “irreversibly” bind to specific 30S subunit proteins and 16S rRNA. Amikacin inhibits protein synthesis by binding to the 30S ribosomal subunit to prevent the formation of an initiation complex with mRNA. Specifically amikacin binds to four nucleotides of 16S rRNA and a single amino acid of protein S12. This interferes with the decoding site in the vicinity of nucleotide 1400 in 16S rRNA of 30S subunit. This region interacts with the wobble base in the anticodon of tRNA. This leads to interference with the initiation complex and misreading of mRNA so incorrect amino acids are inserted into the polypeptide leading to nonfunctional or toxic peptides and the breakup of polysomes into nonfunctional monosomes. Amikacin is a semisynthetic broad spectrum aminoglycoside antibiotic derived from kanamycin A by acetylation. It is commonly administered parenterally for the treatment of Gram-negative infections resistant to gentamycin, kanamycin, and tobramycin because the amikacin molecule has fewer points susceptible to enzymatic attack than the other aminoglycosides.Citation4,Citation9,Citation12 Commonly used aminoglycosides include tobramycin, gentamycin, and amikacin.Citation2 For treatment of cystic fibrosis, combination therapy with an aminoglycoside, a broad spectrum betalactam antibiotic is given intravenously for 14–21 days.Citation4
The minimum inhibitory concentration (MIC) of amikacin is 8 μg/mL for Pseudomonas aeruginosa.Citation1 In previous studies it has been reported that liposomal encapsulated tobramycin showed considerable antimicrobial effect at concentrations below the MIC of the free antibiotic in vitro.Citation2,Citation13 Therefore, reducing the effective concentration of aminoglycosides can be a challenge to reduce their adverse effects.
In the present study the minimum effect of SLNs of amikacin was investigated and compared with MIC of the free drug. The SLNs of amikacin were prepared for minimum size and maximum drug-loading efficiency using cholesterol as the dipodic ingredient. At first, the release profile of the drug from SLNs was studied, then a screening was performed to show the MIC of SLNs of amikacin.
To investigate the stability condition of desired nanoparticles, three different conditions were selected and desired SLNs were stored for two months and their physicochemical properties investigated for both particle dispersion and freeze-dried particles. The antimicrobial efficiency of both dispersion and dried SLNs was the same in first step of production.
Material and methods
Materials
Cholesterol, Tween 80, ethanol, acetone, and Muller-Hinton agar were all from Merck Chemical Company (Darmstadt, Germany). P. aerogynosa (American Type Culture Collection [ATCC] 9027) was obtained from the microbiology laboratory in Tehran University of Medical Sciences (Tehran, Iran).
Preparation of SLNs of amikacin
The SLNs of amikacin were prepared as our previously reported methodCitation8 to achieve optimized particles from a particle size and drug-loading efficiency point of view. Briefly, 160 mg of amikacin powder was dissolved in deionized water containing 1% w/w Tween 80 and homogenized at 11,000 rpm (T 18 basic Ultra-Turrax; IKA Werke GmbH, Staufen, Germany). Then 314 mg of cholesterol (0.81 mmol) as lipid phase was dissolved in 24 mL of the mixture of ethanol/acetone with the ratio of 3:1 (v/v) (equal to 18 mL ethanol and 6 mL acetone) by heating to 70°C and stirring. Then hot oily phase was added to aqueous phase in 25°C under homogenization at 11,000 rpm for 6 min. The prepared emulsion was sonicated in bath sonicator (Tecno-Gaz Ultrasonic system; Tecna S.p.A, Bologna, Italy) and cooled to room temperature to achieve nanoparticles. SLNs were produced in optimum level and type of surfactant, optimum rate and time of homogenization, and ratio of lipid:drug to achieve smallest particles with maximum drug-loading efficiency using central composite design.Citation8
Particle size and zeta potential of produced SLNs were measured using photon correlation spectroscopy (Zetasizer Nano ZS 3000; Malvern Instruments, Worcestershire, UK).
Drug-loading efficiency
Loading efficiency of amikacin in SLNs was calculated by EquationEq. 1(1) .Citation8,Citation14 In this method the concentration of entrapped amikacin was calculated from the difference between the total amount of drug used for preparing SLNs and the free remaining drug in the medium. To do this, after preparing SLNs the emulsion containing SLNs was centrifuged (Sigma Laboratories, Osterode, Germany) at 35,000 rpm for 45 min at −4°C and concentration of amikacin in supernatant was analyzed by high-performance liquid chromatography (HPLC).Citation8
Release study
Release study was performed using dialysis method. Five mL of the optimized formulation before freeze-drying was placed in DO405 dialysis tubing 23 × 15 mm (cut-off: 10–12 KD; Sigma Laboratories, Osterode, Germany) immersed in 50 mL of phosphate buffer solution (pH 7.4). One mL sample was withdrawn at predetermined time intervals and drug concentration was analyzed using pre-column derivatization by HPLC method.Citation8 Drug release profile was studied in different temperature conditions for SLNs of amikacin solution in first dispersion and redispersed freeze-dried nanoparticles after storage at 4°C, 25°C, and 40°C for 60 days.
Freeze drying
Lyophilization was used to prolong stability of amikacin loaded SLNs. Lyophilization of particles was done using cryoprotectants like sucrose, dextrose, and mannitol to limit the risk of aggregation of particles. The process was optimized using D-optimal experimental design reported previously.Citation15
Stability study
The initial particle size and zeta potential of the amikacin loaded nanoparticles dispersion were measured immediately after preparation and after freeze drying, and redispersion of particles in deionized water by bath sonication for 5 s using Zetasizer Nano ZS3000 (Malvern, UK). This batch was divided into three sample sets and each sample set was stored at 4°C, 25°C, and 40°C–4°C being refrigerator conditions, 25°C for room temperature, and 40°C as the indicative hot climate areas that the drug may be stored during distribution from the manufacturer to the end user.
All samples were stored in plain glass vials (USP type 1). Samples were withdrawn after 1, 3, 7, 15, 30, 45, and 60 days and subjected to particle size and zeta potential measurements. Polydispersity index (PDI) and drug released in deionized water containing 1% w/w Tween 80 were studied. However, measurement of released fraction of drug was checked just after 60 days as the samples were dry and had no risk of drug release.Citation16
Morphology study
Morphology of the nanoparticles was characterized by scanning electron microscopy (SEM). The nanoparticles were mounted on aluminum stubs, sputter-coated with a thin layer of Au/Pd, and examined using an SEM (Philips XL30; Philips, Almelo, The Netherlands).
Antimicrobial activity of SLNs
To determine if there is any relationship between the activity of SLNs of amikacin and drug release profile from colloidal vehicle, and also to compare between the activity of nanoparticles of amikacin (directly after preparing particles in original medium and after lyophilization of SLNs and then dispersed in water) with that of free drug, the “well diffusion test” was carried out using P. aeruginosa (ATCC 9027) as the Gram-negative pathogenic strain.
The bacterial suspensions with a cell density equivalent to 0.5 McFarland (1.5 × 108 CFU/mL) were transferred individually onto the surface of Muller–Hinton agar plates using sterile cotton swabs. Wells with 8 mm diameters were prepared by punching a sterile cork borer onto agar plates and removing the agar to form a well. Aliquots of 100 μl of each of two control solutions, free-drug and blank-SLNs, were delivered into the wells. A third well was full with SLNs of amikacin as the test sample. After incubation time for about 24–48 h, at 35°C–37°C, the zones of inhibition around the wells were measured in mm using a caliper.
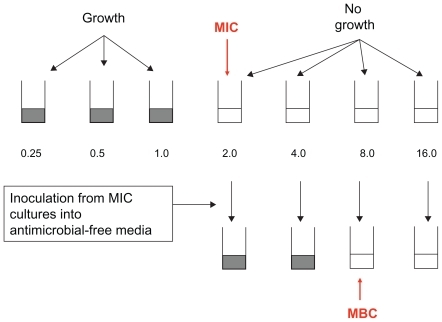
Determination of MIC and minimum bacteriostatic concentration of SLNs against the pathogen
The conventional broth macrodilution tube method was used to determine MIC and minimum bacteriostatic concentration (MBC) of SLNs of amikacin with respect to P. aeruginosa as Gram-negative pathogenic bacteria in vitro.Citation17,Citation18 A stock solution of free amikacin was prepared in sterile water (64 μg/mL) that was further diluted in Muller–Hinton broth to reach a concentration range of 0.125 to 32 μg/mL. SLNs were also dispersed in Muller–Hinton broth to reach an equal concentration of free amikacin as 0.125 to 32 μg/mL according to the percent of drug loading.
Final concentration of bacteria in individual tubes was adjusted to about 5 × 106 CFU/mL. Control tubes contained only culture media without any antimicrobial agent, culture media with SLNs of amikacin due to probable contamination. Other test tubes included culture media with pathogenic strain (in 5 × 106 CFU/mL), culture media with amikacin SLNs and pathogenic strain (in 5 × 106 CFU/mL), and culture media with P. aeruginosa (in 5 × 106 CFU/mL) with free amikacin in MIC (8 μg/mL), as was previously measured.Citation1,Citation2
After 24 and 48 h incubation at 35°C–37°C, the test tubes were examined for possible bacterial turbidity and MBC and MIC of each test compound was determined respectively as lowest concentration that could stop and inhibit visible bacterial growth for 24 h and 72 h.Citation12,Citation13 demonstrates the method of the MIC and MBC testing. A study was done on both freeze-dried and nonfreeze-dried SLNs of amikacin.
Results and discussion
Particle size
The size and zeta potential of the particles before freeze drying were 150 ± 4 nm and + 4 mV, respectively, and after freeze drying these increased to 190 ± 7 nm and + 16 mV. The percentage of drug-loading efficiency was 87% ± 4%.
Drug release profiles and antimicrobial activity
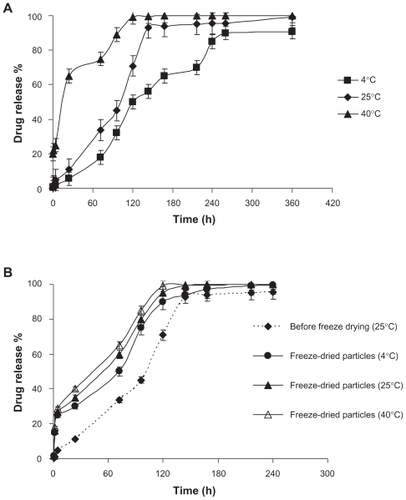
The release profile of amikacin from SLNs dispersion is shown in . shows the percentage of the drug released from the cholesterol carrier which was sustained for 70, 144, and 240 h at 40°C, 25°C, and 4°C, respectively. The differences between different temperatures before lyophilization are quite statistically significant (), but after lyophilization the differences are only significant between 4°C and 40°C, and release was accelerated by increasing the storage temperature from 4°C to 40°C (). Desired freeze-dried SLNs were stored at 4°C, 25°C, and 40°C, and drug release studies were done on each sample. Results show that after freeze drying only an early time burst release could be observed. By increasing the storage temperature, drug release was also accelerated.
Figure 2 Release profiles of amikacin from SLNs at three different storage temperatures A) before and B) after freeze drying.
The antimicrobial activity of SLNs of amikacin after preparation in first dispersion is shown in and antimicrobial activity of lyophilized particles after redispersion is shown in . It can be seen that SLNs of amikacin possessed antimicrobial activity against P. aeruginosa. Although the antimicrobial activity of drug-loaded SLNs was less than that of free drug (),
Table 1 MBC and MIC of free amikacin and SLNs of amikacin before and after freeze drying with respect to Pseudomonas aeruginosa
Figure 3 Photographs of the zone of inhibition produced by free amikacin and its SLNs in A) primary dispersion of SLNs and B) after freeze drying and re-dispersion of SLNs.
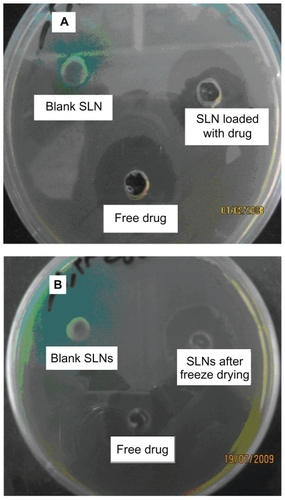
Previously, Beaulac et al, reported that the liposome formulation composed of dipalmitoylphosphatidylcholine and dimyristoylphosphatidylglycerol encapsulating tobramycin showed a considerable antibiotic effect at concentration below the MIC of the free antibiotic in vitro.Citation13 They argued that the enhanced antimicrobial effect may be due to a fusion mechanism of liposome formulation with the bacteria cell wall. Other studies were carried out concerning intracellular bacteria; they reported the possibility of further improving liposomal drug efficacy toward infected cells.Citation19,Citation20 It seems that SLNs and liposomal structures are both composed of lipids and may be comparable.
Freeze drying
The optimum condition for lyophilization was evaluated for the type and percentage of cryoprotectant and freezing temperature, the best results achieved with 12% w/w of sucrose as cryoprotectant at −80°C. As the results are shown elsewhere,Citation16 in this condition the increase in particle size was minimum, the release profile changes were minimum, and SEM pictures showed spherical particles similar prior to freeze drying. Also increasing the charge of particles caused more stability after redispersion.Citation16
Morphology study
SEM pictures of amikacin loaded SLNs are shown in . , relates to the SLNs of amikacin which were stored at 4°C for 60 days, aggregation of particles was not seen. shows SEM pictures of amikacin loaded nanoparticles stored at 25°C and 40°C, respectively. Aggregation of particles and particle size enlargement were seen in these temperatures due to melting of cholesterol. shows SEM picture of lyophilized redispersed particles, this figure confirms that freeze drying doesn’t have any significant impact on the shape and size of SLNs.
Stability study
Table 2 Results of stability study on SLNs of amikacin before and after freeze drying
Conclusion
The aim of this study was to evaluate the efficacy of the amikacin SLNs after preparation in the form of primary dispersion and redispersed freeze-dried particles in comparison with free drug. The loaded drug showed less MIC and MBC than free amikacin for both primary SLN dispersion and redispersed lyophilized particles. Some probable reasons were the lipophilic nature of SLNs which enhanced cellular entrance of drug into bacterial membrane and the small size of particles. Consequently it could be concluded that aminoglycosides might be administered in lower doses or longer intervals by delivering as solid lipid nanoparticles to reduce their side effects.
Acknowledgments
The authors would like to thank the Research Vice Chancellor of Isfahan University of Medical Sciences for financial support of this project. Technical assistance of Mr Jamali, Mrs S Norouzi, Mrs H Saabani, and Mr R Jafarian is appreciated. Thanks are given to Mrs Yousefi for English editing of the manuscript.
References
- BartalCDanonASchlaefferFPharmacokinetic dosing of aminoglycosides: a controlled trialAm J Med2003114319419812637133
- SchiffelersRStormGBakker-WoudenbergILiposome-encapsulated aminoglycosides in pre-clinical and clinical studiesJ Antimicrob Chemother200148333334411532996
- WilsonJFDavisACTobinCMEvaluation of commercial assays for vancomycin and aminoglycosides in serum: a comparison of accuracy and precision based on external quality assessmentJ Antimicrob Chemother2003521788212805260
- HammondLJGaldwellSCampbellPWCystic fibrosis, intravenous antibiotics, and home therapyJ Pediatr Health Care19915124301990112
- ShahSMisraALiposomal amikacin dry powder inhaler: effect of fines on in vitro performanceAAPS Pharm Sci Tech200454e65
- CullenABCoxCAHippSJWolfsonMRShafferTHIntra-tracheal delivery strategy of gentamicin with partial liquid ventilationResp Med19999311770778
- AtyabiFTalaieFDinarvandRThiolated chitosan nanoparticles as an oral delivery system for amikacin: in vitro and ex vivo evaluationsJ Nanosci Nanotechnol2009984593460319928123
- VarshosazJGhaffariSKhoshayandMRAtyabiFAzarmiSKobarfardFDevelopment and optimization of amikacin solid lipid nanoparticles by central composite designJ Liposome Res20102029710419621981
- BaraniHMontazerMA review on applications of liposomes in textile processingJ Liposome Res200818324926218770074
- MullerHMaderKGohlaSSolid lipid nanoparticles (SLN) for controlled drug delivery – a review of the state of the artEur J Pharm Biopharm20005016117710840199
- ZhangNPingQHuangGXuWChengYHanXLecitin-modified solid lipid nanoparticles as carriers for oral administration of insulinInt J Pharm20063271215315916962267
- SweetmanSCThe Complete Drug Reference35th edLondon, UKChicago Pharmaceutical Press (PhP)2007
- BeaulacCSachetelliSLagaceJIn vitro bactericidal efficacy of sub-MIC concentration of liposome encapsulated antibiotic against gram- negative and gram- positive bacteriaJ Antimicrob Chemother199841135419511035
- GazoriTKhoshayandMRAziziEYazdizadePNomaniAHaririanIEvalution of alginate/chitosan nanoparticles as antisense delivery vector: formulation, optimization, and in vitro characterizationCarbohyd Polym2009773599606
- VarshosazJGhaffariSKhoshayandMRAtyabiFOptimization of freeze-drying condition of amikacin solid lipid nanoparticles using D-optimal experimental designPharm Dev Tech2010 In press
- VivekKReddyHMurthyRInvestigation of the effect of the lipid matrix on drug entrapment, in vitro release, and physical stability of olanzapine-loaded solid lipid nanoparticlesAAPS Pharm Sci Tech2007841624
- LarsonTAPetersonLRGerdingDNMicrodilution aminoglycoside susceptibility testing of Pseudomonas aeruginosa and Escherichia coli: correlation between MICs of clinical isolates and quality control organismsJ Clin Microbiol19852258198213932459
- SiddiqiSHeifetsLBCynamonMRapid broth macrodilution method for determination of MICs for Mycobacterium avium isolatesJ Clin Microbiol1993319233223388408551
- BermudezLEWuMYoungLSIntracellular killing of Mycobacterium avium complex by rifapentine and liposome-incapsulated amikacinJ Infect Dis198715635105133611835
- PoynerEAAlparHOAlmeidaAJGambleMDBrownMRWA comparative study on the pulmonary delivery of tobramycin encapsulated into liposomes and PLA microspheres following intravenous and endotracheal deliveryJ Control Rel19953514148