Abstract
Premalignant lesions arise from cells that abnormally proliferate and have a tendency to become cancerous. Developing methods to specifically target and remove these premalignant lesions is imperative to the prevention of malignant progression into gastrointestinal (GI) tumors. However, accurate detection and diagnosis of GI precancerous lesions is challenging, as these lesions show little or no structural change. Thus, this prevents early intervention and reduces the success rate of therapy. In this review, we performed a systematic analysis of the technological advancements in the combined application of nanovesicles (NVs) and the human interstitial fluid (HIF) to specifically target GI premalignant lesions. NVs, which include quantum dots (QDs), are small membranous vehicles of a nanometer diameter that are widely used as drug delivery vectors, therapeutic effectors and diagnostic sensors. HIF is the fluid that is present in human interstitial tissues (HITs) in which signaling molecules and agents travel and can be found throughout the body. HIF is exploited by tumor cells for their invasion, migration and spread. Because the HITs span the entire submucosa of the gastrointestinal tract, they have been increasingly targeted in GI tumor therapy. The challenges involved in the combined application of NVs and HIF in the detection, diagnosis, prognosis and therapy of GI premalignant lesions are also discussed.
Introduction
The appearance of premalignant lesions, which arise from abnormal cell growth, indicates a precancerous condition that has the potential to develop into tumors, including benign and malignant. The presence of such lesions is associated with a high risk of tumors development. The occurrence of these premalignant lesions are usually accompanied by non-characteristic symptoms, such as fatigue, mild discomfort, and loss of appetite. Thus, this poses a challenge for the accurate detection, diagnosis and administration of early intervention therapy. In this review, we focus on the gastrointestinal (GI) premalignant lesions in GI precancerous conditions, such as early gastric cancer, atrophic gastritis, intestinal metaplasia and dysplasia, which develop in a multi-step process.Citation1
The early detection and diagnosis of structural or molecular changes in premalignant lesions in GI tumor screening and surveillance is critical for early intervention and follow-up. Early diagnosis can substantially reduce the prevalence and mortality rate of GI tumors. Common detection methods of GI premalignant lesions include endomicroscopy and histology biopsy, which are invasive and time-consuming methods that typically span over 3–5 days. Clinicians base the diagnosis on the physical appearance of the abnormal lesions.Citation2 Because both of these methods are invasive, their clinical application is limited by the patient age and the size of the lesion. Thus, there is a need for the improvement of and the development of novel diagnostic and therapeutic methods that can circumvent these limitations.
The human interstitium is a newly discovered organ that comprises fluid-filled compartments between tissues in the dermis and submucosae, and other fibrous connective tissues that are found throughout the body, especially those that span the entire GI tract.Citation3 The interstitiumis well-known to be the primary source of lymph. The fluid-filled channels of human interstitial tissues (HITs) connect to form a collagenous network, which is compressible and distensible, thereby enabling it to function as a shock absorber. This unique characteristic of HITs supports the occurrence of mechanical processes such as peristalsis in the GI tract. The continuous structure of the network enables these channels to function as a molecular conduit, which is exploited by tumor cells to enable their invasion, migration and spread.Citation3 The transport and distribution of pro-cancerous molecules and invasive tumor cells via the human interstitial fluid (HIF) in the submucosae channels in the GI tract promotes cancer progression and metastasis. Tumor interstitial fluid (TIF), which is found at tumor sites, comprises a high concentration of tumor-related substances. Thus, the direct sampling of TIF is an effective tumor detection and diagnostic strategy.Citation4
Here, nanovesicles (NVs) have two meanings: synthetic vesicles that combined with nanomaterials, such as QDs;natural vesicles of nanometer diameter. Specifically, NVs are those synthesized that integrated small, structurally diverse vesicles with QDs and those natural that diameters ranging from 25 to 1,000 nanometers (nm). Therefore, NVs are grouped into two types: body-derived exosomes,Citation5 and syntheticCitation6 or biomimetic liposomes.Citation7 Exosomes can be found in human body fluids and they function in antigen presentation, immune response, intercellular communication, and RNA and protein transfer.Citation8 As molecular messengers in signal transduction between cells, they can influence molecular physiology and promote cancer development. Thus, they can be used as biomarkers in the early diagnosis of GI tumors.Citation9 Synthetic NVs can be sub-divided into two groups: a conjugate mixture of nanoparticles (NPs) and vesicles such as the quantum dots (QDs) which are widely used in biomedical sensing and imaging applications;Citation10 and human-engineered exosomes with diameters ranging from 50 to 200nm.Citation11 The tumor-derived exosomes (TEXs), which carry proteins and RNAs (including microRNAs and messenger RNAs), are malignant vesicles that are prevalent in the serum of cancer patients. These exomes hold promise in disease diagnosis and prognosis as they can protect labile cancer biomarkers from degradation.Citation12 These TEXs can escape from the tumor’s microenvironment and reach the cerebrospinal fluid and systemic blood circulation via the HIF.Citation13 Hence, the HIF and TIF are rich sources for the discovery of novel serum biomarkers.Citation14 Targeted inhibition of TEXs may be a viable therapeutic strategy for controlling tumor progression.Citation15
In this review, we first discuss the developments in the field regarding the separate biological applications of NVs and HIF in diagnostics, cancer monitoring, therapy and drug delivery in GI premalignant lesion diagnosis. Following that, the latest research developments regarding the application of the novel synergistic therapeutic approach combining NVs and HIF in detection, diagnosis, treatment and follow-up of GI premalignant lesions are discussed. In closing, we outline the challenges to be addressed and the future directions of subsequent research in the field.
Individually Targeted NVs or HIF Against GI Premalignant Lesions
Both body-derived and synthesized NVs are commonly used in the detection,Citation16–Citation21 diagnosis,Citation22–Citation27 treatmentCitation28–Citation33 and prognosisCitation34–Citation39 of primary GI tumors, especially inpremalignant lesions (). It could be deduced from these studies that the success of NV therapeutic strategies lies in the encapsulated content which includes proteins, RNAs and NPs. As a delivery tool, however, is also a foreign substance, the NV is easily absorbed and destroyed by the liver and spleen mediated by reticuloendothelial system (RES) once enter the body. The conjugated QDs could intend to be a blanket, thereby shielding the drug from harsh gastrointestinal conditions, whereas the NVs ensure high encapsulation efficiency of their contents and passive targeting to GI premalignant lesions, and ultimately improve the gastrointestinal resistance, mucoadhesion properties, and tumor growth inhibition. Carboxymethyl chitosan coud provide a gastric resistant coating and impart a stealth character to lipid vesicles that they tethered, thereby preventing their RES clearance, and greatly improve the ability of the encapsulated paclitaxel to against tumors.Citation40 The intrinsic properties of QDs, including easy functionalization, biocompatibility, and tumor passive targeting, made them suitable for use as vehicles of NVs biogenesis. The use of NVs in tumor therapy also takes advantage of their stability, biocompatibility, and targeting ability for targeting the delivery of proteins, RNAs, or therapeutic chemotherapeutic drugs by passive or an active loading. Passive loading consists of the overexpression of RNAs, including miRNAs, shRNAs, or mRNA in a cell culture and active loadingconsists of the directed introduction of the desired molecule into purified NVs via electroporation.Citation41 Hence, careful selection of the specific NV cargo and conjugated QDs to target certain GI precancerous lesions is a key consideration.
Table 1 Targeting NVs Against GI Premalignant Lesions
Because HIF present in HITs is a novel medium that has been recently identified to be a rich source of tumor cells and tumor-related substances, there are only a few studies investigating its application in tumor detection, diagnosis, treatment, and prognosisto dateCitation42–Citation45 (). HIF that is comprised of disproportionately high levels of tumor cells, tumor-specific markers, and tumor-related substances compared to the normal interstitial fluid is referred to as TIF.Citation4 TIF represents a potential source of biomarkers, they could influence both cell growth and the invasive potential of tumoral cells, influences the communication between cells themselves, between tumoral cells and both healthy surrounding tissues and the systemic environment. The mechanism through which the TIF influences tumor development has not been clearly ascertained to date, however, this may closely related to the niform pressure existence, flow diminishes, different secreted substances, cytokines and chemotactic factors, wich leading to drugs accumulation decline, waste removal reduce, tumoral tissue expand, tumoral cells growth, etc.Citation46 TIF is not readily available, until now, no standard method has been identified for their isolation. Now several methods have been developed: capsule implantation, wick-in-needle technique, microdialysis,wick method, glass capillary, capillary ultrafiltration, tissue elution and tissue centrifugation.Citation46 Proteomic approaches could be used to quantify known bioactive compounds in the TIF and could open new frontiers and opportunities in the discovery of cancer biomarkers.
Table 2 Targeting HIF Against GI Premalignant Lesions
These preliminary studies focused on determining whether an association exists between the presence of a tumor and a certain substance found in the TIF. Important details, such as the identity of the substance in the TIF and its expression level, have yet to ber clarified. Nonetheless, these preliminary findings demonstrate the significance of TIF to against tumors and suggest the possibility of combining the use of HIF with NVs to target GI premalignant lesions. In particular, the advantages of both could be combined to maximize the anti-tumor effect.
Combined Targeting of NVs and HIF Against GI Premalignant Lesions
Detection, Diagnosis and Prognosis Strategies
TEXs, which are abundant in the TIF in cancer patients,Citation4 are exosomes used for packaging and transporting proteins, mRNAs, and miRNAs that are differentially expressed by tumor cells.Citation47 These exosomes can be found in the peripheral blood as they have been reported to be able to cross the blood-brain barrier intact.Citation48 In addition, TEXs have been detected in human serum and saliva, which are considered unconventional diagnostic body fluids.Citation49 As mentioned earlier, as a partial and integral relationship, TIF and HIF are interflowed. TEXs can also be found in HIF and their encapsulated miRNAs can be used to predict the molecular features and pathological and physiological status of the primary tumor cells. Hence, they can be used to determine tumor stagingCitation47 and can serve as novel diagnostic tumor markers.Citation13,Citation50 TEXs are isolated and derived from TIF. Conventional methods for isolating exosomes include ultracentrifugation, size exclusion chromatography, polymer precipitation, immunocapture assays and microfluidic methods.Citation51 Recently developed novel methods, for example the analytical microfluidic platform (EVHB-Chip), have significantly reduced the time taken for the isolation of tumor-specific exosomes.Citation52 TEXs are nano-scale messengers of tumor cells loaded with bio-molecular cargo of DNA, RNA, and proteins. Since the core content of TEXs is the nucleotides and proteins it encapsulates, it is necessary to use genomics and proteomics techniques for tumour analysis and characterization. The contents of TEXs can be released through membrane permeabilization or lysis solution; after centrifugation and extraction, the nucleotides can be analysis by genomics technology to confirm whether these exomsomes are tumor-specific (). Upon successful validation of the isolated TEX candidates, a detection and diagnosis protocol can be executed to monitor the GI premalignant lesions. The specific proteins of TEXs are becoming new biomarkers for tumor monitoring and diagnosis. The quantity and type of TEXs and their encapsulated specific proteins are also factors for consideration in early cancer detection, diagnosis and prognosis. After centrifugation and extraction, proteomics technology is typically used to distinguish, determine and characterize the type and quantity of specific proteins in TEXs detected and whether they are tumor-specific (). This is further confirmed by morphology analysis and surface biomarker testing.
Figure 1 Using TEXs to detect, diagnose and predict GI premalignant lesions. TEXs are isolated from the TIF and analyzed. The encapsulated contents within the TEXs are released via membrane permeabilizers or lysis solution. The type and quantity of TEXs and their encapsulated specific proteins with respect to a specific tumor are confirmed using proteomics technology. The nucleotides (DNAs, RNAs) relationship with a specific tumor is subsequently analyzed and confirmed using genomics technology. Finally, conduct the multi-omics techniques analysis for tumour characterization.
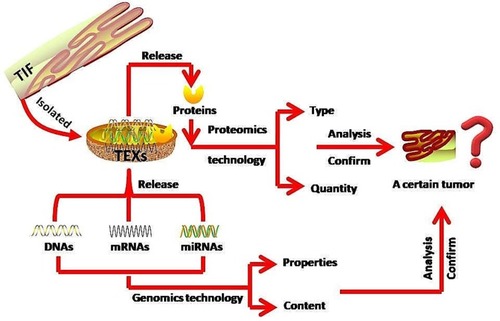
Other than TEXs, tumor-specific proteins are also easily detected in biofluidsCitation51 and show promise as potential biomarkers.Citation53 Current proteomics technology for tumor detection and monitoring are sufficiently sensitive in the detection of plasma-diluted tumor-specific proteins and biomarker candidates that were initially present at high concentrations when first secreted into the TIF.Citation54 After detecting the tumor-specific markers, functional assays are then performed to understand their biological mechanisms. Simultaneously, a vesicle database search (EVpedia, Exocarta, and Vesiclepedia) is conducted to determine their novelty. Upon confirmation of novelty, ELISA and Western blot are performed to determine the potential clinical relevance of the marker, with the application of a cut-off value.Citation51 TEX and tumor-specific marker identification is complicated by the abundance of other tumor-related substances and tumor-associated macrophages (TAMs) in the tumor interstitium, which are found to be associated with a poor tumor prognosis.Citation55 Hence, careful and detailed analysis of the substances that are detected in the TIF is necessary to ensure the accurate identification of biomarker candidates.
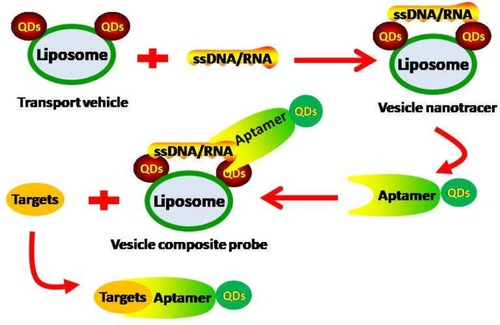
NVs can be conjugated with QDs to function as vehicles to transport specific therapeutic cargo (eg aptamer) to target GI premalignant lesions directly.Citation19,Citation56 A vesicle composite probe is constructed in multiple steps to carry a specific aptamer for direct detection of the lesion (). First, a transport vehicle comprising QDs that are conjugated to a liposome is constructed. Because the biophysical interactions between different liposomes and QDs vary, specific QDs should be designed to enable conjugation with the liposome of choice. Next, the transport vehicle is conjugated to vesicle nanotracers, such as a labeled single stranded DNA/RNA binding protein. In the final construction step, the aptamer that is labeled with other QDs is combined to complete the vesicle composite probe. The designed aptamer should bind specifically to the target and be released at the appropriate time to enable target detection. Localized surface plasmon resonance biosensors with self-assembled gold nanoislands (SAM-AuNIs) has been demonstrated to be another direct method for direct target detection and this method may be able to distinguish exosomes from different sources.Citation57 QD-conjugated NVs encapsulated with NPs can also be used as various imaging contrast agents, in combination with computer tomography and other imaging tools, as an indirect method to identify GI lesions.Citation19–Citation21
Figure 2 Using QD-conjugated NVs to directly detect GI premalignant lesions. A transport vehicle is first constructed by conjugating a liposome to QDs. Next, a vesicle nanotracer is constructed by conjugating a labeled single stranded DNA/RNA binding protein to the transport vehicle. Following this, a vesicle composite probe is formed by combining the vesicle nanotracer with an aptamer that is labeled with other QDs. The designed aptamer should be able to recognize and bind to the labeled protein in the vesicle nanotracer and to the in vivo target. Upon encountering the in vivo target, the aptamer binds to it preferentially and dissociates from the vesicle nanotracer.
Treatment Strategies
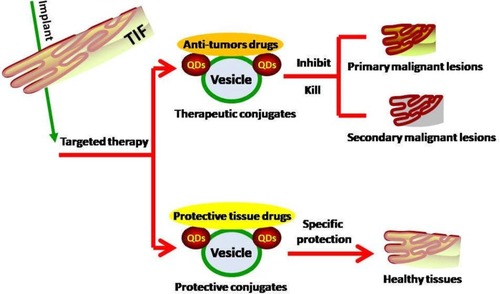
After detection of the precancerous lesion, targeted therapy can be initiated. Synthetic NVs combined with QDs and specific anti-tumor drugs can be designed to disrupt the tumor microenvironment to suppress tumor growth and metastasis or to destroy the primary premalignant lesions or secondary malignant lesions via NV implantation in the TIF (). For healthy tissues or cells, protective conjugates can be used to avoid secondary malignant transformation. Timely follow-up after treatment should subsequently be scheduled.
Figure 3 NV implantation in the TIF to target GI premalignant lesions. NVs carrying either an anti-tumor drug to suppress or kill the primary lesions or secondary malignant lesions, or protective conjugates to shield healthy tissues or cells from the effects of the tumor, thereby avoiding secondary malignant transformation, can be implanted into the TIF.
Once malignancy occurs, tumor-specific proteins are released into the HIF and migrate to invade the downstream tissues or organs, or via an active transport route to an upstream location, resulting in tumor spread and metastasis. Current treatment strategies against tumor-specific proteins utilize molecular targeting such as blocking synthesis and secretion,Citation53,Citation58 inhibiting activity, reducing the number, or disrupting biological activity. Imatinib,Citation53 and ZNF545, which inhibit promoter activity and reduce translation efficiency,Citation58 have been reported to inhibit the biogenesis of tumor-specific proteins. An epidermal growth factor receptor-specific masked chimeric antigen receptor has been shown to specifically recognize and cleave tumor-associated antigens, and induce high levels of antitumor activity.Citation59 QD-conjugated NVs that carry tumor-specific proteases could be implanted into the TIF to disrupt the tumorigenic activities. For example, NVs that carry collagenases could be used to control collagen overexpression in the TIF to disrupt the tumor microenvironment.Citation60
TEXs are one of the abundant tumor-related substances that are present in the TIF of patients. TEXs play a key role in the mediation of paracrine communication between tumor cells that promote tumor progression and metastasis.Citation61,Citation62 TEX targeting strategies include gene silencing to reduce synthesis, suppression of internalization by normal cells or tissues, reducing the number of TEXs and inhibiting their biological activity. QDs functionalized with antibodies against overexpressed receptors and inhibitors, such as siRNAs and hairpins, have been shown to reduce the biogenesis, release and secretion of TEXs.Citation47 QDs functionalized with inhibitors of uptake machinery (eg protein receptors) enable the modulation of the tumor microenvironment, thereby preventing secondary damage to other normal cells or tissues.Citation47 TEXs in the TIF could first be identified by QD-conjugated NVs through medical imaging techniques (eg flow cytometry, atomic force microscopy and transmission electron microscopy),Citation63 and then selectively captured and removed from the entire circulatory system by ultracentrifugation and hemofiltration.Citation64,Citation65 Hydrolases and other antagonists can be used to inhibit or prevent the activity of TEXs, thereby blocking further tumor communication and metastasis. Heparanase inhibitors and its neutralizing monoclonal antibodies could inhibit heparanase activity and knock it down, markedly attenuating tumor progression, metastasis and chemoresistance.Citation66 Additionally, a similar report suggests that chemical and molecular inhibition of Bcl-xL could lead to a significant decrease in the uptake TEXs in hematopoietic malignant cells.Citation67 Suppression of TEX production should be the first therapeutic approach used to treat GI premalignant lesions before considering other, more complicated, strategies. However, accurate identification of the genes associated with specific TEXs for gene silencing therapy has proven challenging. Hence, future research should be directed towards resolving this issue.
Therapeutic NV targeting is a common treatment strategy. It is a biofunctionalized drug delivery system prepared by biomimetic strategies, specific for the therapeutic transport of miRNA, mRNA, siRNA, lncRNA, peptides, and other synthetic drugs.Citation68,Citation69 Therapeutic NVs require high colloidal stability, biocompatibility, and ligand conjugation chemistry properties, to covalently or electrostatically conjugate with a variety of biomolecules, such as nucleic acids, peptides, proteins, antibodies, fluorophores and drugs, thereby giving them labile, therapeutic, and diagnostic capabilities. In addition, NVs should ideally not display significant cytotoxicity or activate the complement system.Citation70 The implantation of NVs carrying therapeutic cargo could be performed by indirect (permeation via concentration gradient)Citation47 or direct methods (electroporation)Citation71 to increase the specificity and efficiency of the therapy. Most solid tumors possess high vascular densities with extensive permeability, impaired lymphatic clearance, and large fenestrations into the tumors’ interstitial space. Thus, these inherent structural defects of tumor tissues enable the selective penetration and retention of NVs, introduced through passive targeting, at the tumor site but not in normal tissues.Citation47 Intravenous injection can be used to effectively enrich therapeutic NVs at tumor sites to improve drug delivery efficiency and antitumor efficacy.Citation72 Monoclonal antibodies, peptides/proteins, folic acid, carbohydrates, and DNA/RNA, which function in ligand binding, are employed in the active targeting to the overexpressed cell-surface receptors of tumor cells.Citation47 A direct method for NV implantation in the TIF is nanochannel electroporation (NEP). This method involves the direct injection of NVs into the cytoplasm, bypassing the endocytosis route. For example, a “3DNEP chip” has been engineered to deliver extremely small biological elements to huge plasmids with near zero negative impact on the targeted cells.Citation73 The bioavailability of therapeutic NVs affects their internalization into the tumor. Conjugating biofunctionalized peptides to the surfaces of NVs could significantly improve their uptake into the tumor lesion, and, thus, improve the treatment outcome.Citation74
Strategies to Optimize Therapeutic NVs
NVs as potential diagnostic and prognostic biomarkers of GI premalignant lesions offer a minimally invasive clinical approach. There is ongoing research investigating the molecular impact of NVs in the body. For example, there are studies investigating the metabolism, acute toxicity, bioavailability, biological clearance, pharmacological delivery, and chronic exposure safety of NVs. In addition, efforts are also being directed towards the standardization of protocols to characterize NV-based medicine and the biological effects, to increase their production while ensuring reproducibility and quality.
Methods to Modify Therapeutic NVs
Therapeutic NVs should ideally be stable with a strong water resistant membrane and they should be chemically inert so that there are no unintended effects regarding their contents or their timed release, allowing them to exert their anti-tumor function on their target. Having excellent biocompatibility in vivo is another desirable property that enables NVs to cross biological barriers and efficiently deliver bioactive cargo, and can this even help in evading immune recognition.Citation75
The development of modification of the approach of therapeutic NVs in GI lesion therapy has focused on the “modular design” of every component, such as the improvement of the lipophilicity of encapsulated agents, the modification of the lipid bilayer with amphiphilic molecules, the modification of the surface proteins, internal proteins and nucleic acids through cell engineering.Citation76 For example, modified NVs have been demonstrated to exhibit improved thermal, pH and proteolytic stability and increased enzymatic activity.Citation77 In addition to a good colloidal stability and a controlled size, having a high loading capacity for hydrophilic and hydrophobic drugs is a critical characteristic of therapeutic NVs that ensures the effectiveness of the delivery system. Hydrophobic therapeutics (such as hyaluronan) can be loaded in to their membranes to increase the drug stability and efficacy, while hydrophilic therapeutics (such as RNAs) can be encapsulated to improve their cellular delivery.Citation78 Polyethylene glycol, cholesterol and egg phosphatidylcholine, have been reported to modify encapsulated drugs.Citation74 The drugs encapsulated in synthetic NVs should not be limited to only one or a few types, but should instead contain a variety of drugs, such as compound drugs, that include two or more anti-tumor drugs, to increase the therapeutic efficacy. For example, the triple drug-loaded NVs including cisplatin, doxorubicin (DOX), and camptothecin, showed enhanced anti-tumor efficacy.Citation79
It has been reported that NV stability primarily depends on the morphology and not the size.Citation80 In addition, nanodiscs, rather than uniformly-sized NVs, could lead to an improved delivery syste, in terms of cellular internalization.Citation80 There is no best stand-alone method todate for the improvement of the biocompatibility and bioavailability of therapeutic NVs. Various methods should instead be integrated to enable optimal NV internalization. For example, low-dose photodynamic therapy can enhance the uptake of liposomal cisplatin in pleural malignancies, provided that the photo-induction conditions are tightly controlled to minimize side effects.Citation81 In another example of a multipronged NV synthesis strategy that significantly enhanced tumor inhibition, protamine was used to improve specific NV internalization and reduce non-specific uptake, heparin was used to enhance stability, and calcium carbonate was used to improve drug loading and confer pH sensitivity.Citation82
Optimization of NV Preparation Methods
Next, we discuss an alternative approach for theoptimization of therapeutic NVs: optimization of the NV synthesis methods. Hydration and ultrasonication methods have been shown to improve nanovesicular delivery, thereby enabling the transfer of the vesicular encapsulated contents. For example, NVs coupled with transferrin were reported to function as an efficient delivery vehicle for tocotrienols in breast cancer therapy.Citation83 Nanoprecipitation has been demonstrated to effectively improve NV internalization, thereby significantly improving anticancer efficacy.Citation84 For example, glutathione-responsive paclitaxel dimer NVs with ultrahigh drug content that were prepared using nanoprecipitation, exhibited good colloid stability, effective cellular uptake and comparable cytotoxicity to Taxol.Citation85 Rehydration and dehydration methods can be applied to ensure NV compositional uniformity.Citation86 For example, subjecting NVs that were engineered to carry three different types of venoms to rehydration and dehydration methods, led to improved stability and anti-cancer efficacy.Citation87 Through self-assembly/sol-gel techniques, vesicles can be conjugated with certain QDs to form multi-functional organic/inorganic hybrid NVs that display a unique morphology and reasonable stability under physiological conditions.Citation88 These hybrid NVs can be used for accurate cancer diagnosis and monitoring. QDs can be implanted on the bilayer membrane surface or inner core of the vesicle to form conjugated NVs, which can be labeled with fluorescent QDs and tracked by specific marker antibodiesCitation89 to define their physical characteristics and determine their concentration.Citation90 NVs that are synthesized through the sol-gel method possess hydrophilic and hydrophobic microdomains, which enable the co-delivery of both water-soluble and water-insoluble drugs for maximum anti-tumor efficacy.Citation89 For example, the topotecan (TPT)-loaded liposome-QD hybrid was shown to be able to transport both therapeutic and diagnostic agents simultaneously, thereby functioning as a multifunctional delivery vehicle.Citation89 Ensuring a uniform size of therapeutic NVs during synthesis is also an area that has been studied. The cross-flow method was developed to ensure size consistency of NVs.Citation91 In addition, NVs should be engineered to be of an adequate size, ideally around 400–800nm, to transport sufficient therapeutic drugs while also being small enough to move through the TIF and infiltrate the tumor. Nanodroplets that are capable of tumor internalization have been designed for extravascular imaging.Citation92 This method could potentially be applied in a similar manner for the early diagnosis and treatment of GI premalignant lesions.
The QDs and the encapsulated drugs within NVs can also affect the overall performance of therapeutic NVs.Citation89 Because QDs are not naturally present in the body, they can produce toxic effects, have poor photochemical properties and a short lifetime. Designing NVs to carry QDs within them was found to effectively improve their biocompatibility, photostability, and improve tumor internalization.Citation93
Combined Targeting of Therapeutic NVs and TIF
A corollary approach to enhance the efficacy of therapeutic NVs in GI tumor therapy is to exploit the intrinsic nature of the TIF. The methods that have been devised to date, such as increasing the local tissue temperature or taking pro-cyclical drugs to increase body fluid metabolism, thereby indirectly increasing its transport capacity, rely on the fluidity of the TIF to drive implanted therapeutic NVs towards tumor sites. For example, temperature-sensitive liposomes (TSLs) have been designed to carry heat-triggered drugs, wherein the rate of drug release depends on the presence or absence of unbound proteins when heated, to target the tumor interstitium.Citation94
Challenges in the Combined Targeting of NVs and TIF Against GI Premalignant Lesions
The ideal therapeutic NVs shouldbe able to carry specific drugs, have an excellent stability, biocompatibility and maximum anti-tumor efficacy. For example, QD-conjugated NVs can be constructed by simply mixing oppositely charged carbon QDs that are coordinated by cerium (Ce3+) ions and DNA in water, to enable their application in the biological setting.Citation95 Despite extensive research into the improvement of the target-specificity of anti-tumor drugs used in therapy and the developments regarding their ability to overcome tumor heterogeneity, these issues still remain a challenge.Citation96 In addition, better NV implantation approaches that do not affect the stability and therapeutic properties of the QD-conjugated NVs have to be developed. This is especially important for the targeting of cancer cell types that internalize the therapeutic conjugates through receptor-mediated endocytosis.Citation97
Previously, biomarkers were thought to be generally applicable among different cancer types, and that none were designated as representative of a specific tumor type. However, research on the TIF revealed that different cancers express different tumor-specific proteins, which show little overlap.Citation54 Screening for biomarker candidates that are specific for different tumors is, thus, possible. For example, specific exosomal miRNA clusters could potentially be used as biomarkers to diagnose gastric cancer at an early stage, and determine the cancer stage and metastasis.Citation34 In the event of malignancy, tumor cells or tissues secrete TEXs and also secrete tumor-related substances into the TIF. Because both TEXs and the TIF arise from the same tumor source, this raised the question of the existence of overlapping substances being secreted. Indeed, a preliminary study demonstrated that intracellular proteins in the TIF may actually be derived from TEXs.Citation54 However, this observation must be confirmed through additional studies. TIF can accumulate tumor by-products in a relatively narrow space, due to perfusion of tumorigenic tissues or cells. Thus, the TIF is a rich resource for tumor biomarkers and therapeutic targets and should be extracted from the HIF for detailed analysis and identification. There is currently no consensus regarding a method for the extractionand characterization onTIF.Citation98 In fact, it is thought that different extraction and analysis methods should be used for TIF with diverse origins, and that multiple methods should be used if necessary.
Even after resolving the issues regarding the possibility of identifying the same candidate from the TIF and TEXs, and deciding the strategies to adopt for TIF extraction and analysis, there remains the challenge of deciding the best treatment method along with other problems that may arise during the course of therapy. For example, issues such as the selection of drugs that can bind specifically to the identified target, understanding the mechanism of transport and metabolism of the therapeutic conjugates in the TIF and their influence on the tumors’ microenvironment. Tumor-specific proteins in the TIF and TEXs promote GI precancerous lesion and malignant tumor proliferation and invasion.Citation3,Citation99 Because the TIF is fluid, the determination of the correct location to extract it to reliably reflect the local tumor microenvironment and provide accurate early tumor detection and effective monitoring is a challenge. In most tissues and conditions, the TIF is not readily available. Tumor-specific proteins and TEXs are usually present at different concentrations in different locations. Typically, they can be found at high concentrations in tumors and at low concentrations in the plasma.Citation54
Perspectives and Conclusions
We have systematically reviewed the existing technologies and developments in GI premalignant lesion therapy using NVs and HIF individually. In addition, we have discussed the combined application of NVs and HIF in early GI premalignant lesion detection, diagnosis, prognosis and treatment. From our comprehensive analysis, we have noted several unavoidable and currently unresolved problems.
Although preliminary research has been performed to determine the role of TEXs in the mediation ofthe communication between tumors and normal tissues, thereby promoting their spread and metastasis,Citation100 further studies should be conducted to elucidate their genetic basis and precise mechanism of action.
Efforts should also be directed towards the improvement of NV synthesis and implantation therapeutic strategies. For example, issues to consider in the use of synthetic NVs conjugated with NPs include the potential toxicity of the NPs used, the stability of their conjugates, as well as the ease of tracking, monitoring and follow-up. In addition, there should be a compromise between the NP size and biocompatibility when selecting NPs for use inconjugation with NVs. One viable strategy is to tag NP surfaces with specific ligands to target more aggressive tumor cells in the digestive tract, thereby improving the selectivity and efficacy of the nanodelivery vehicle while reducing the side effects.Citation72 Multiple NV implantation methods are currently being used. However, there is no consensus regarding the best method among them. It is also not known whether different implantation methods should be combined to yield the best results. Hence, further research should be performed to address these issues.
The macro-control of NV metabolism after implantation in the TIF is another area that warrants further study. Strategies shoul dencourage the accelerated flow of NVs in the TIF to indirectly increase drug release rate and protect the unaffected downstream tissues or organs, while ensuring the maintenance of a fixed therapeutic local concentration to minimize off-target effects. This seems to be an irreconcilable contradiction, but it does need to be resolved.
Lastly, there is no consensus regarding the detection threshold for the use of tumor-specific markers as biomarkers in early detection. Tumour-specific markers can be detected in both precancerous lesions and malignant tumors.Citation101 Thus, a threshold value should be set for a more accurate diagnosis of the tumor stage and prognosis.
In conclusion, our analyses support the use of a combined application of NVs and HIF in targeting GI premalignant lesions and their continued development and evolution. Improvement of the stability, biocompatibility, and amenability of therapeutic NVs is necessary for their clinical application. Therapy should not be limited to a single method and should instead combine several compatible approaches to achieve maximum efficacy.
Abbreviations
GI, gastrointestinal; NV(s), nanovesicle(s);NP(s), nanoparticle(s); QD(s), quantum dot(s); HIF, human interstitial fluid; HITs human interstitial tissues; TIF tumor interstitial fluid; nm, nanometers; TEXs, tumor derived exosomes; miRNAs, microRNAs; mRNAs, messenger RNAs; RES, reticuloendothelial system; TAMs, tumor-associated macrophages; NEP, nanochannel electroporation; DOX, doxorubicin; TPT, topotecan; μm, micrometer; TSLs, temperature-sensitive liposomes.
Disclosure
The authors report no conflicts of interest in this work.
Acknowledgment
This study was supported by grants from the National Natural Science Foundation of China (81660743 and 81860790).
References
- Rentien AL, Lévy M, Copie-Bergman C, et al. Long-term course of precancerous lesions arising in patients with gastric MALT lymphoma. Dig Liver Dis. 2018;50(2):181–188. doi:10.1016/j.dld.2017.10.01429102522
- Li X, Li H, He X, et al. Spectrum- and time-resolved endogenous multiphoton signals reveal quantitative differentiation of premalignant and malignant gastric mucosa. Biomed Opt Express. 2018;9(2):453–471. doi:10.1364/BOE.9.00045329552386
- Benias PC, Wells RG, Sackey-Aboagye B, et al. Structure and distribution of an unrecognized interstitium in human tissues. Sci Rep. 2018;8(1):4947. doi:10.1038/s41598-018-23062-629588511
- Onsurathum S, Haonon O, Pinlaor P, et al. Proteomics detection of S100A6 in tumor tissue interstitial fluid and evaluation of its potential as a biomarker of cholangiocarcinoma. Tumour Biol. 2018;40(4):1010428318767195. doi:10.1177/101042831876719529629840
- Zhu Q, Heon M, Zhao Z, et al. Microfluidic engineering of exosomes: editing cellular messages for precision therapeutics. Lab Chip. 2018;18(12):1690–1703. doi:10.1039/c8lc00246k29780982
- Li P, Yao Q, Lü B, et al. Visible Light-induced supra-amphiphilic switch leads to transition from supramolecular nanosphere to nanovesicle activated by pillar[5]arene-based host-guest interaction. Macromol Rapid Commun. 2018;39(20):e1800133. doi:10.1002/marc.20180013329786904
- Danaei M, Dehghankhold M, Ataei S, et al. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics. 2018;10(2):E57. doi:10.3390/pharmaceutics1002005729783687
- Sun B, Peng J, Wang S, et al. Applications of stem cell-derived exosomes in tissue engineering and neurological diseases. Rev Neurosci. 2018;29(5):531–546. doi:10.1515/revneuro-2017-005929267178
- Atay S, Wilkey DW, Milhem M, et al. Insights into the proteome of gastrointestinal stromal tumors-derived exosomes reveals new potential diagnostic biomarkers. Mol Cell Proteomics. 2018;17(3):495–515. doi:10.1074/mcp.RA117.00026729242380
- Bhat A, Huan K, Cooks T, et al. Probing interactions between AuNPs/AgNPs and Giant Unilamellar Vesicles (GUVs) using hyperspectral dark-field microscopy. Int J Mol Sci. 2018;19(4):E1014. doi:10.3390/ijms1904101429597298
- Arenaccio C, Chiozzini C, Ferrantelli F, et al. Exosomes in therapy: engineering, pharmacokinetics and future applications. Curr Drug Targets. 2019;20(1):87–95. doi:10.2174/138945011966618052110040929779478
- Boyiadzis M, Whiteside TL. The emerging roles of tumor-derived exosomes in hematological malignancies. Leukemia. 2017;31(6):1259–1268. doi:10.1038/leu.2017.9128321122
- Giusti I, Di Francesco M, Dolo V. Extracellular vesicles in glioblastoma: role in biological processes and in therapeutic applications. Curr Cancer Drug Targets. 2017;17(3):221–235. doi:10.2174/156800961666616081318295927528364
- Zhang J, Hao N, Liu W, et al. In-depth proteomic analysis of tissue interstitial fluid for hepatocellular carcinoma serum biomarker discovery. Br J Cancer. 2017;117(11):1676–1684. doi:10.1038/bjc.2017.34429024941
- Ludwig N, Whiteside TL. Potential roles of tumor-derived exosomes in angiogenesis. Expert Opin Ther Targets. 2018;22(5):409–417. doi:10.1080/14728222.2018.146414129634426
- Lin LY, Yang L, Zeng Q, et al. Tumor-originated exosomal lncUEGC1 as a circulating biomarker for early-stage gastric cancer. Mol Cancer. 2018;17(1):84. doi:10.1186/s12943-018-0834-929690888
- Wang J, Liu Y, Sun W, et al. Plasma exosomes as novel biomarker for the early diagnosis of gastric cancer. Cancer Biomark. 2018;21(4):805–812. doi:10.3233/CBM-17073829400660
- McMullen JRW, Selleck M, Wall NR, et al. Peritoneal carcinomatosis: limits of diagnosis and the case for liquid biopsy. Oncotarget. 2017;8(26):43481–43490. doi:10.18632/oncotarget.1648028415645
- Zhang K, Du X, Yu K, et al. Application of novel targeting nanoparticles contrast agent combined with contrast-enhanced computed tomography during screening for early-phase gastric carcinoma. Exp Ther Med. 2018;15(1):47–54. doi:10.3892/etm.2017.538829387181
- Feng ST, Li H, Luo Y, et al. Molecular targeted magnetic resonance imaging of human colorectal carcinoma (LoVo) cells using novel superparamagnetic iron oxide- loaded nanovesicles: in vitro and in vivo studies. Curr Cancer Drug Targets. 2016;16(6):551–560. doi:10.2174/156800961666616060312361627262319
- Blanco VM, Latif T, Chu Z, et al. Imaging and therapy of pancreatic cancer with phosphatidylserine-targeted nanovesicles. Transl Oncol. 2015;8(3):196–203. doi:10.1016/j.tranon.2015.03.01126055177
- Yan L, Dong X, Gao J, et al. A novel rapid quantitative method reveals stathmin-1 as a promising marker for esophageal squamous cell carcinoma. Cancer Med. 2018;7(5):1802–1813. doi:10.1002/cam4.144929577639
- Wang Y, Zhang C, Zhang P, et al. Serum exosomal microRNAs combined with alpha-fetoprotein as diagnostic markers of hepatocellular carcinoma. Cancer Med. 2018;7(5):1670–1679. doi:10.1002/cam4.139029573235
- Huang Z, Zhang L, Zhu D, et al. A novel serum microRNA signature to screen esophageal squamous cell carcinoma. Cancer Med. 2017;6(1):109–119. doi:10.1002/cam4.97328035762
- Davis HW, Hussain N, Qi X. Detection of cancer cells using SapC-DOPS nanovesicles. Mol Cancer. 2016;15(1):33. doi:10.1186/s12943-016-0519-127160923
- Ding J, Feng M, Wang F, et al. Targeting effect of PEGylated liposomes modified with the Arg-Gly-Asp sequence on gastric cancer. Oncol Rep. 2015;34(4):1825–1834. doi:10.3892/or.2015.414226238930
- Hoshino I, Maruyama T, Fujito H, et al. Detection of peritoneal dissemination with near-infrared fluorescence laparoscopic imaging using a liposomal formulation of a synthesized indocyanine green liposomal derivative. Anticancer Res. 2015;35(3):1353–1359.25750285
- Li S, Yao J, Xie M, et al. Exosomal miRNAs in hepatocellular carcinoma development and clinical responses. J Hematol Oncol. 2018;11(1):54. doi:10.1186/s13045-018-0579-329642941
- Pan JH, Zhou H, Zhao XX, et al. Role of exosomes and exosomal microRNAs in hepatocellular carcinoma: potential in diagnosis and antitumour treatments (Review). Int J Mol Med. 2018;41(4):1809–1816. doi:10.3892/ijmm.2018.338329328436
- Zhang H, Wang Y, Bai M, et al. Exosomes serve as nanoparticles to suppress tumor growth and angiogenesis in gastric cancer by delivering hepatocyte growth factor siRNA. Cancer Sci. 2018;109(3):629–641. doi:10.1111/cas.1348829285843
- George J, Yan IK, Patel T. Nanovesicle-mediated delivery of anticancer agents effectively induced cell death and regressed intrahepatic tumors in athymic mice. Lab Invest. 2018;98(7):895–910. doi:10.1038/s41374-018-0053-429748614
- Borrelli DA, Yankson K, Shukla N, et al. Extracellular vesicle therapeutics for liver disease. J Control Release. 2018;273:86–98. doi:10.1016/j.jconrel.2018.01.02229373816
- Thapa RK, Ku SK, Choi HG, et al. Vibrating droplet generation to assemble zwitterion-coated gold-graphene oxide stealth nanovesicles for effective pancreatic cancer chemo-phototherapy. Nanoscale. 2018;10(4):1742–1749. doi:10.1039/c7nr07603g29308494
- Sun ZP, Li AQ, Jia WH, et al. MicroRNA expression profiling in exosomes derived from gastric cancer stem-like cells. Oncotarget. 2017;8(55):93839–93855. doi:10.18632/oncotarget.2128829212193
- Tokuhisa M, Ichikawa Y, Kosaka N, et al. Exosomal miRNAs from peritoneum lavage fluid as potential prognostic biomarkers of peritoneal metastasis in gastric cancer. PLoS One. 2015;10(7):e0130472. doi:10.1371/journal.pone.013047226208314
- Matsumura T, Sugimachi K, Iinuma H, et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br J Cancer. 2015;113(2):275–281. doi:10.1038/bjc.2015.20126057451
- Guo J, Fan KX, Xie LI, et al. Effect and prognostic significance of the KAI1 gene in human gastric carcinoma. Oncol Lett. 2015;10(4):2035–2042. doi:10.3892/ol.2015.360426622792
- Zheng NG, Mo SJ, Li JP, et al. Anti-CSC effects in human esophageal squamous cell carcinomas and Eca109/9706 cells induced by nanoliposomal quercetin alone or combined with CD 133 antiserum. Asian Pac J Cancer Prev. 2014;15(20):8679–8684. doi:10.7314/apjcp.2014.15.20.867925374189
- Wang B, Zhuang X, Deng ZB, et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther. 2014;22(3):522–534. doi:10.1038/mt.2013.19023939022
- Joshi N, Saha R, Shanmugam T, et al. Carboxymethyl-chitosan-tethered lipid vesicles: hybrid nanoblanket for oral delivery of paclitaxel. Biomacromolecules. 2013;14(7):2272–2282. doi:10.1021/bm400406x23721348
- EL Andaloussi S, Andaloussi S, Mäger I, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347–357. doi:10.1038/nrd397823584393
- Fujie S, Matsubayashi H, Ishiwatari H, et al. Intraductal tubulopapillary epithelial proliferation associated with type 1 autoimmune pancreatitis. J Gastrointestin Liver Dis. 2018;27(1):83–87. doi:10.15403/jgld.2014.1121.271.fuj29557419
- Liang L, Fanzong L, Peixi Z, et al. Plexiform angiomyxoid myofibroblastic tumor of the stomach: a case report. Diagn Cytopathol. 2017;45(1):55–58. doi:10.1002/dc.2357227561459
- Reichel D, Curtis LT, Ehlman E, et al. Development of halofluorochromic polymer nanoassemblies for the potential detection of liver metastatic colorectal cancer tumors using experimental and computational approaches. Pharm Res. 2017;34(11):2385–2402. doi:10.1007/s11095-017-2245-928840432
- Atochina-Vasserman EN, Guo CJ, Abramova E, et al. Surfactant dysfunction and lung inflammation in the female mouse model of lymphangioleiomyomatosis. Am J Respir Cell Mol Biol. 2015;53(1):96–104. doi:10.1165/rcmb.2014-0224OC25474372
- Ura B, Di Lorenzo G, Romano F, et al. Interstitial fluid in gynecologic tumors and its possible application in the clinical practice. Int J Mol Sci. 2018;19(12):E4018. doi:10.3390/ijms1912401830545144
- Roma-Rodrigues C, Raposo LR, Cabral R, et al. Tumor microenvironment modulation via gold nanoparticles targeting malicious exosomes: implications for cancer diagnostics and therapy. Int J Mol Sci. 2017;18(1):E162. doi:10.3390/ijms1801016228098821
- García-Romero N, Carrión-Navarro J, Esteban-Rubio S, et al. DNA sequences within glioma-derived extracellular vesicles can cross the intact blood-brain barrier and be detected in peripheral blood of patients. Oncotarget. 2017;8(1):1416–1428. doi:10.18632/oncotarget.1363527902458
- Sun Y, Liu S, Qiao Z, et al. Systematic comparison of exosomal proteomes from human saliva and serum for the detection of lung cancer. Anal Chim Acta. 2017;982:84–95. doi:10.1016/j.aca.2017.06.00528734369
- Ferguson Bennit HR, Gonda A, McMullen JRW, et al. Peripheral blood cell interactions of cancer-derived exosomes affect immune function. Cancer Microenviron. 2019;12(1):29–35. doi:10.1007/s12307-018-0209-129603062
- Li A, Zhang T, Zheng M, et al. Exosomal proteins as potential markers of tumor diagnosis. J Hematol Oncol. 2017;10(1):175. doi:10.1186/s13045-017-0542-829282096
- Reátegui E, van der Vos KE, Lai CP, et al. Engineered nanointerfaces for microfluidic isolation and molecular profiling of tumor-specific extracellular vesicles. Nat Commun. 2018;9(1):175. doi:10.1038/s41467-017-02261-129330365
- Berglund E, Daré E, Branca RM, et al. Secretome protein signature of human gastrointestinal stromal tumor cells. Exp Cell Res. 2015;336(1):158–170. doi:10.1016/j.yexcr.2015.05.00425983130
- Wagner M, Wiig H. Tumor interstitial fluid formation, characterization, and clinical implications. Front Oncol. 2015;5:115. doi:10.3389/fonc.2015.0011526075182
- Woo SR, Corrales L, Gajewski TF. Innate immune recognition of cancer. Annu Rev Immunol. 2015;33:445–474. doi:10.1146/annurev-immunol-032414-11204325622193
- Miao YB, Ren HX, Gan N, et al. A homogeneous and “off-on” fluorescence aptamer-based assay for chloramphenicol using vesicle quantum dot-gold colloid composite probes. Anal Chim Acta. 2016;929:49–55. doi:10.1016/j.aca.2016.04.06027251948
- Thakur A, Qiu G, Ng SP, et al. Direct detection of two different tumor-derived extracellular vesicles by SAM-AuNIs LSPR biosensor. Biosens Bioelectron. 2017;94:400–407. doi:10.1016/j.bios.2017.03.03628324860
- Cheng Y, Liang P, Geng H, et al. A novel 19q13 nucleolar zinc finger protein suppresses tumor cell growth through inhibiting ribosome biogenesis and inducing apoptosis but is frequently silenced in multiple carcinomas. Mol Cancer Res. 2012;10(7):925–936. doi:10.1158/1541-7786.MCR-11-059422679109
- Han X, Bryson PD, Zhao Y, et al. Masked chimeric antigen receptor for tumor-specific activation. Mol Ther. 2017;25(1):274–284. doi:10.1016/j.ymthe.2016.10.01128129121
- Dolor A, Szoka FC Jr. Digesting a path forward: the utility of collagenase tumor treatment for improved drug delivery. Mol Pharm. 2018;15(6):2069–2083. doi:10.1021/acs.molpharmaceut.8b0031929767984
- Rajagopal C, Harikumar KB. The origin and functions of exosomes in cancer. Front Oncol. 2018;8:66. doi:10.3389/fonc.2018.0006629616188
- Huang Y, Liu K, Li Q, et al. Exosomes function in tumor immune microenvironment. Adv Exp Med Biol. 2018;1056:109–122. doi:10.1007/978-3-319-74470-4_729754177
- Madhankumar AB, Mrowczynski OD, Patel SR, et al. Interleukin-13 conjugated quantum dots for identification of glioma initiating cells and their extracellular vesicles. Acta Biomater. 2017;58:205–213. doi:10.1016/j.actbio.2017.06.00228583903
- Yamashita T, Takahashi Y, Nishikawa M, et al. Effect of exosome isolation methods on physicochemical properties of exosomes and clearance of exosomes from the blood circulation. Eur J Pharm Biopharm. 2016;98:1–8. doi:10.1016/j.ejpb.2015.10.01726545617
- Urabe F, Kosaka N, Yoshioka Y, et al. The small vesicular culprits: the investigation of extracellular vesicles as new targets for cancer treatment. Clin Transl Med. 2017;6(1):45. doi:10.1186/s40169-017-0176-z29238879
- Vlodavsky I, Singh P, Boyango I, et al. Heparanase: from basic research to therapeutic applications in cancer and inflammation. Drug Resist Updat. 2016;29:54–75. doi:10.1016/j.drup.2016.10.00127912844
- Vardaki I, Sanchez C, Fonseca P, et al. Caspase-3-dependent cleavage of Bcl-xL in the stroma exosomes is required for their uptake by hematological malignant cells. Blood. 2016;128(23):2655–2665. doi:10.1182/blood-2016-05-71596127742710
- Barile L, Vassalli G. Exosomes: therapy delivery tools and biomarkers of diseases. Pharmacol Ther. 2017;174:63–78. doi:10.1016/j.pharmthera.2017.02.02028202367
- Zhang P, Zhang L, Qin Z, et al. Genetically engineered liposome-like nanovesicles as active targeted transport platform. Adv Mater. 2018;30:7. doi:10.1002/adma.201705350
- Tagalakis AD, Maeshima R, Yu-Wai-Man C, et al. Peptide and nucleic acid-directed self-assembly of cationic nanovehicles through giant unilamellar vesicle modification: targetable nanocomplexes for in vivo nucleic acid delivery. Acta Biomater. 2017;51:351–362. doi:10.1016/j.actbio.2017.01.04828110069
- Fernandes E, Ferreira JA, Andreia P, et al. New trends in guided nanotherapies for digestive cancers: a systematic review. J Control Release. 2015;209:288–307. doi:10.1016/j.jconrel.2015.05.00325957905
- Yang K, Liu Y, Liu Y, et al. Cooperative assembly of magneto-nanovesicles with tunable wall thickness and permeability for mri-guided drug delivery. J Am Chem Soc. 2018;140(13):4666–4677. doi:10.1021/jacs.8b0088429543442
- Chang L, Bertani P, Gallego-Perez D, et al. 3D nanochannel electroporation for high-throughput cell transfection with high uniformity and dosage control. Nanoscale. 2016;8(1):243–252. doi:10.1039/c5nr03187g26309218
- Ye Z, Zhang T, He W, et al. Methotrexate-loaded extracellular vesicles functionalized with therapeutic and targeted peptides for the treatment of glioblastoma multiforme. ACS Appl Mater Interfaces. 2018;10(15):12341–12350. doi:10.1021/acsami.7b1813529564886
- Kooijmans SAA, Schiffelers RM, Zarovni N, et al. Modulation of tissue tropism and biological activity of exosomes and other extracellular vesicles: new nanotools for cancer treatment. Pharmacol Res. 2016;111:487–500. doi:10.1016/j.phrs.2016.07.00627394168
- Tao SC, Guo SC, Zhang CQ. Modularized extracellular vesicles: the dawn of prospective personalized and precision medicine. Adv Sci (Weinh). 2018;5(2):1700449. doi:10.1002/advs.20170044929619297
- Wan S, He D, Yuan Y, et al. Chitosan-modified lipid nanovesicles for efficient systemic delivery of l-asparaginase. Colloids Surf B Biointerfaces. 2016;143:278–284. doi:10.1016/j.colsurfb.2016.03.04627022867
- Hood JL. Post isolation modification of exosomes for nanomedicine applications. Nanomedicine (Lond). 2016;11(13):1745–1756. doi:10.2217/nnm-2016-010227348448
- Deshpande NU, Jayakannan M. Cisplatin-stitched polysaccharide vesicles for synergistic cancer therapy of triple antagonistic drugs. Biomacromolecules. 2017;18(1):113–126. doi:10.1021/acs.biomac.6b0141128064505
- Aresh W, Liu Y, Sine J, et al. The morphology of self-assembled lipid-based nanoparticles affects their uptake by cancer cells. J Biomed Nanotechnol. 2016;12(10):1852–1863. doi:10.1166/jbn.2016.229229359898
- Cavin S, Wang X, Zellweger M, et al. Interstitial fluid pressure: a novel biomarker to monitor photo-induced drug uptake in tumor and normal tissues. Lasers Surg Med. 2017;49(8):773–780. doi:10.1002/lsm.2268728544068
- Gong MQ, Wu JL, Chen B, et al. Self-assembled polymer/inorganic hybrid nanovesicles for multiple drug delivery to overcome drug resistance in cancer chemotherapy. Langmuir. 2015;31(18):5115–5122. doi:10.1021/acs.langmuir.5b0054225927163
- Tan DM, Fu JY, Wong FS, et al. Tumor regression and modulation of gene expression via tumor-targeted tocotrienol niosomes. Nanomedicine (Lond). 2017;12(20):2487–2502. doi:10.2217/nnm-2017-018228972460
- Feng C, Han X, Chi L, et al. Synthesis, characterization, and in vitro evaluation of TRAIL-modified, cabazitaxel -loaded polymeric micelles for achieving synergistic anticancer therapy. J Biomater Sci Polym Ed. 2018;29(14):1729–1744. doi:10.1080/09205063.2018.148361629851539
- Pei Q, Hu X, Zhou J, et al. Glutathione-responsive paclitaxel dimer nanovesicles with high drug content. Biomater Sci. 2017;5(8):1517–1521. doi:10.1039/c7bm00052a28191576
- Oropeza-Guzman E, Ruiz-Suárez JC. Dehydration/rehydration cycles for mixing phospholipids without the use of organic solvents. Langmuir. 2018;34(23):6869–6873. doi:10.1021/acs.langmuir.8b0079929779384
- Al-Asmari AK, Ullah Z, Al Balowi A, et al. In vitro determination of the efficacy of scorpion venoms as anti-cancer agents against colorectal cancer cells: a nano-liposomal delivery approach. Int J Nanomedicine. 2017;12:559–574. doi:10.2147/IJN.S12351428144138
- Niu D, Wang X, Li Y, et al. Facile synthesis of magnetite/perfluorocarbon co-loaded organic/inorganic hybrid vesicles for dual-modality ultrasound/magnetic resonance imaging and imaging-guided high-intensity focused ultrasound ablation. Adv Mater. 2013;25(19):2686–2692. doi:10.1002/adma.20120431623447424
- Seleci M, Ag Seleci D, Scheper T, et al. Theranostic liposome-nanoparticle hybrids for drug delivery and bioimaging. Int J Mol Sci. 2017;18(7):E1415. doi:10.3390/ijms1807141528671589
- McNicholas K, Michael MZ. Immuno-characterization of exosomes using nanoparticle tracking analysis. Methods Mol Biol. 2017;1545:35–42. doi:10.1007/978-1-4939-6728-5_327943205
- Park YH, Lee DH, Um E, et al. On-chip generation of monodisperse giant unilamellar lipid vesicles containing quantum dots. Electrophoresis. 2016;37(10):1353–1358. doi:10.1002/elps.20160003526920999
- Xu X, Song R, He M, et al. Microfluidic production of nanoscale perfluorocarbon droplets as liquid contrast agents for ultrasound imaging. Lab Chip. 2017;17(20):3504–3513. doi:10.1039/c7lc00056a28933795
- Samadikhah HR, Nikkhah M, Hosseinkhani S. Enhancement of cell internalization and photostability of red and green emitter quantum dots upon entrapment in novel cationic nanoliposomes. Luminescence. 2017;32(4):517–528. doi:10.1002/bio.320727767252
- Al-Ahmady ZS, Hadjidemetriou M, Gubbins J, et al. Formation of protein corona in vivo affects drug release from temperature-sensitive liposomes. J Control Release. 2018;276:157–167. doi:10.1016/j.jconrel.2018.02.03829522832
- Wang L, Wang Y, Sun X, et al. Versatile self-assembly and biosensing applications of DNA and carbon quantum dots coordinated cerium ions. Chemistry. 2017;23(43):10413–10422. doi:10.1002/chem.20170170928580665
- Kim HJ, Oh SC. Novel systemic therapies for advanced gastric cancer. J Gastric Cancer. 2018;18(1):1–19. doi:10.5230/jgc.2018.18.e329629216
- Liang G, Kan S, Zhu Y, et al. Engineered exosome-mediated delivery of functionally active miR-26a and its enhanced suppression effect in HepG2 cells. Int J Nanomedicine. 2018;13:585–599. doi:10.2147/IJN.S15445829430178
- Gromov P, Gromova I. Characterization of the tumor secretome from Tumor Interstitial Fluid (TIF). Methods Mol Biol. 2016;1459:231–247. doi:10.1007/978-1-4939-3804-9_1627665563
- Liu H, Chen W, Zhi X, et al. Tumor-derived exosomes promote tumor self-seeding in hepatocellular carcinoma by transferring miRNA-25-5p to enhance cell motility. Oncogene. 2018;37(36):4964–4978. doi:10.1038/s41388-018-0309-x29786077
- Chiba M, Watanabe N, Watanabe M, et al. Exosomes derived from SW480 colorectal cancer cells promote cell migration in HepG2 hepatocellular cancer cells via the mitogen-activated protein kinase pathway. Int J Oncol. 2016;48(1):305–312. doi:10.3892/ijo.2015.325526647805
- Tanaka M, Ishikawa S, Ushiku T, et al. EVI1 modulates oncogenic role of GPC1 in pancreatic carcinogenesis. Oncotarget. 2017;8(59):99552–99566. doi:10.18632/oncotarget.2060129245923
