Abstract
Background
Food allergy (FA) is a significant public health problem. The therapeutic efficacy for FA is unsatisfactory currently. The breakdown of intestinal immune tolerance is associated with the pathogenesis of FA. Therefore, it is of great significance to develop novel therapeutic methods to restore immune tolerance in treating FA.
Methods
We proposed an oral administration strategy to treat FA by co-delivering food allergen epitope fragment (peptide: IK) and adjuvant R848 (TLR7 ligand) in the mPEG–PDLLA nanoparticles (PPLA-IK/R848 NPs). The generation of tolerogenic dendritic cells (DCs) and regulatory T cells (Tregs) induced by PPLA-IK/R848 NPs were evaluated in vitro and in vivo. The therapeutic effects of PPLA-IK/R848 NPs were also assessed in an OVA-induced FA model.
Results
PPLA-IK/R848 NPs could efficiently deliver IK to DCs to drive DCs into the tolerogenic phenotypes and promote the differentiation of Tregs in vitro and in vivo, significantly inhibited FA responses through the recovery of intestinal immune tolerance.
Conclusion
Oral administration of PPLA-IK/R848 NPs could efficiently deliver IK and R848 to intestinal DCs and stimulate DCs into allergen tolerogenic phenotype. These tolerogenic DCs could promote the differentiation of Tregs, which significantly protected mice from food allergic responses. This study provided an efficient formulation to alleviate FA through the recovery of immune tolerance.
Introduction
Food allergy (FA) is a T helper (Th)-2 dominant and IgE-mediated immune disorders that result in non-tolerance to a specific food. Both regulatory T cells (Tregs) deficiency and dysfunction were found to be associated with FA.Citation1 FA is a serious public health problem that adversely affects the people’s quality of life and the economy.Citation2 FA is more prevalent in young people with 6–8% incidence compared to 3–4% in adults.Citation3 More than 90% of FA are due to eight main foods: milk, egg, peanut, tree, soy, wheat, fish and shellfish.Citation4 The management of FA is restricted to avoid touching or taking food allergens. Indeed, at present, there are no Food and Drug Administration (FDA) approved treatments for FA. Despite allergen-specific immunotherapyCitation5–Citation7 has been employed in the treatment of FA, it still carry unacceptable risks, such as IgE-mediated adverse effects.Citation8 Thus, improved modalities of delivering allergen-specific immunotherapy to least IgE-mediated allergic reaction are needed.
T cell epitopes of allergen-based immunotherapy has been employed in the treatment of allergic diseases.Citation9 In this strategy, peptides (T cell or B cell epitope fragments) were selected from the allergens. These allergen fragments retain the immunological ability to induce immune response, and they are too short to cross-link IgE on mast cells compared with the whole allergen molecule,Citation10,Citation11 thereby decrease the IgE-mediated side effects. However, the immune responses induced by these small peptides are limited unless combining with an adjuvant. Resiquimod (R848), a known adjuvant with similar functions of Toll-like receptor (TLR)7 ligand, can effectively reduce the respiratory symptoms of allergic rhinitis and asthma.Citation12,Citation13 Therefore, R848 could be a potential adjuvant candidate to treat FA if it is combined with allergen epitope fragments.
However, R848 is a hydrophobic imidazoquinoline compound. The poor solubility will lead to poor immunological responses thus more doses of adjuvant are required. To solve this problem, nanoscale drug delivery systems are usually preferred. Amphiphilic methoxy poly(ethylene glycol)-poly(D,L-lactide) (mPEG-PDLLA) copolymers are promised as biocompatible, biodegradable and nontoxic amphiphilic block copolymer.Citation14,Citation15 They offer the potential to achieve high solubility of hydrophobic drugs. For many peptide drugs larger than 1000 Da, the optimized delivery systems are necessary to enhance the peptide permeation across the physiological barriers.Citation16 mPEG-PDLLA could also act as highly effective peptide delivery systems and offer multiple advantages, such as the function to control and extend the release of peptideCitation17,Citation18 and protect the encapsulated peptide from degradation after systemic injection.Citation19
In this study, an ovalbumin (OVA) induced FA mouse model was developed. An OVA epitope fragment peptide OVA33-47 (IAIMSALAMVYLGAK, IK in short) and R848 were co-encapsulated in mPEG-PDLLA nanoparticles (PPLA-IK/R848 NPs) in the treatment of experimental FA via oral administration (). We hypothesize that the PPLA-IK/R848 NPs can efficiently deliver IK and R848 to the intestine and be phagocytosed by dendritic cells (DCs) to induce tolerogenic DCs. These tolerogenic DCs could promote the differentiation of Tregs in intestine (), thereby, to alleviate FA.
Scheme 1 A scheme of the conformation of PPLA-IK/R848 NPs and the role of it in the FA inhibition by inducing tolerogenic DCs and Foxp3+ Tregs in the intestine.
Notes: PPLA-IK/R848 NPs can efficiently deliver the IK and R848 to the intestine and be phagocytosed by dendrtic cells (DCs) to induce TGF-β-producing tolerogenic DCs and to promote the differentiation of Tregs in intestine.
Abbreviations: OVA, ovalbumin; PPLA-IK/R848 NPs, OVA epitope fragment IK and R848 co-delivery nanoparticles; FA, food allergy; DCs, dendritic cells; TGF-β, transforming growth factor-β; Foxp3, Forkhead box protein 3; Tregs, regulatory T cells; Th2, T-helper 2 cells.
Materials and methods
Materials
mPEG2000-PDLLA2000 was purchased from Jinan Daigang Biomaterial Co., Ltd (Jinan, China). OVA33-47: IAIMSALAMVYLGAK (IK) and FITC labeled IK (FITC-IK) were synthesized by ONTERES biotechnologies Co., Ltd (Hangzhou, China). TLR7 agonist R848 was purchased from MedChem Express (Monmouth Junction, NJ). The normal saline was supplied by Shandong Qidu pharmaceutical Co., Ltd (Shandong, China). The Float-A-Lyzer®G2 dialysis cassette was obtained from Spectrum Labs (CA, USA). Mouse GM-CSF and IL-4 were purchased from Sino Biological, China. RPMI 1640 was developed by Roswell Park Memorial Institute. Penicillin-streptomycin and fetal bovine serum (FBS) were purchased from Thermo Fisher Scientific. All the other organic solvents and chemicals were of the highest degree of purity commercially available. The water used in the experiment was deionized.
Preparation of PPLA-IK/R848 NPs
PPLA-IK/R848 NPs were prepared by w/o/w double emulsion solvent evaporation as described before with some modifications.Citation20 Briefly, 2 mg of IK was dissolved in 500 µl of methanol and distilled water mixed solution (v:v 1:1), then subsequently added to 1mL of dichloromethane (DCM) containing 10 mg R848 and 40 mg mPEG-PDLLA with ultrasonication at 250 W for 5 mins (KunShan Ultrasonic Instruments Co., Ltd, Kunshan, China) to prepare a water-in-oil emulsion (W1/O). Next, the primary emulsion was added to 10 mL of an aqueous tween 80 solution (2.5% w/v) and the mixture was emulsified again by constant ultrasonication, followed by evaporation of methanol and DCM under vacuum until no organic solvent remained. Then the nanoparticles were collected by centrifugation for 20 min at 20,000 g, washed with pyrogen-free water and resuspended in pyrogen-free water. The fluorescent labeling of PPLA-IK/R848 NPs (FITC-IK/R848 NPs for short) was prepared using FITC-IK as described above.
Characterization of PPLA-IK/R848 NPs
The mean particle size, polydispersity index (PDI), and zeta potential of PPLA-IK/R848 NPs were measured by dynamic light scattering (Zetasizer Nano ZS; Malvern Instruments, Malvern, UK) at 25 °C. Each sample was measured in triplicates with 12 scans each. The morphology of PPLA-IK/R848 NPs was observed using a JEM-1400 electron microscope (JEOL, Tokyo, Japan). One drop of the PPLA-IK/R848 NPs was placed on a 300-mesh copper grid, air-dried, and negatively stained with 2% (w/v) uranyl acetate for electron microscope observation.
Stability of PPLA-IK/R848 NPs in stimulated gastrointestinal fluids
PPLA-IK/R848 NPs (containing 0.2 mg/mL of IK and 1mg/mL of R848) were mixed (1:4, v/v) with simulated gastric fluid (containing 1.0% pepsinum in 1mol/L diluted HCl, pH=1,4) or simulated intestinal fluid (containing 1.0% pancreatin in pH 6.8 phosphate buffer saline (PBS, 0.01 M)) and incubated at 37 °C. At specific time intervals, 1 mL of sample was removed and analyzed (size change and particle distribution). Each experiment was performed in triplicate.
In vitro drug release behavior
To evaluate the in vitro release behavior of IK and R848 from NPs, 1 mL FITC-IK/R848 NPs (containing 0.5 mg/mL of FITC-IK and 2.5 mg/mL of R848) were placed in Float-A—Lyzer dialysis cassettes (molecular weight cut off 20 kDa; Spectrum Labs, Rancho Dominguez, CA, USA), immersed into 1 L of PBS (pH 7.4, 0.1 mol/L), and incubated at 37 °C with stirring (100 rpm). FITC-IK and R848 physical mixture suspensions (0.5 mg FITC-IK and 2.5 mg R848 were directly dispersed in 1 mL of water at 37 °C with ultrasonication at 250 W for 15 mins) were used as a control and were treated under the same condition. At specific time intervals, 50 μL of samples were withdrawn from the cassettes, and the cassettes were replenished with 50μL of fresh PBS solution. The cumulative release of both FITC-IK and R848 were estimated by the reduction of the quantity inside the dialysis bag with the HPLC method. The release medium was replaced every 24 hrs to ensure sink conditions. All the assays were performed in triplicate.
In vitro cell uptake by DC 2.4 cells
DC2.4 cells (ATCC, Manassas, VA. 5×105 cells/well) were seeded into 12-well plates and maintained at 37 °C in 5% CO2 in RPMI 1640 medium supplemented with 10% FBS and 100 U/mL penicillin and 0.1 mg/mL streptomycin overnight. The medium was replaced by the same amount of FITC-IK NPs and free FITC-IK DMSO solution (40 μg/mL or 80 μg/mL, 1 mL). After 12 hr of incubation, the supernatant in each well was removed. The cells were washed 3 times with cold PBS and fixed with 4% paraformaldehyde for 20 min. Then the cells were washed 3 times again with PBS and stained with DAPI for 5min. The cellular uptake of FITC-IK NPs and free FITC-IK solution were monitored by fluorescence microscope (Olympus, Tokyo, Japan).
PPLA-IK/R848 NPs primed DCs and Treg induction
Bone marrow-derived dendrtic cells (BMDC) were prepared as previously reported.Citation21 Briefly, bone marrow cells were obtained from the bone marrow of femurs and iliac bones of Balb/c mice. The cells were seeded in 6-well plates with complete RPMI-1640 culture medium in the presence of IL-4 (10 ng/mL) and GM-CSF (20 ng/mL). BMDCs were cultured with blank-NPs, the same amount of IK/R848 mixture or PPLA-IK/R848 NPs (the corresponding concentration of IK and R848 is 1.6 μg/mL and 8 μg/mL, respectively). The whole culture medium with indicated reagents was replaced on day 3 and half of the culture medium was replaced on day 5 and day 7. On day 8, all cells were collected. A part of BMDCs was used for DC phenotypes analysis by flow cytometry and Western blot .
Splenocytes were harvested by mincing spleen tissue and passing the cells through a 200-mesh screen. Naïve CD4+ T cells were isolated from splenocytes by magnetic cell sorting (MACS) kit purchased from the Miltenyi Biotech (Bergisch Gladbach, Germany). The primed BMDCs (4×105 cells/well) were co-cultured with naive CD4+ T cells (8×106 cells/well) in 24-well plates which were coated with an anti-CD3 (1 μg/mL) and stimulated with anti-CD28 (2 μg/mL) and OVA (20 μg/mL). The cells were collected 3 days later and analyzed by flow cytometry.
In vivo cell uptake by intestinal DCs
For easier detection of cell uptake by intestinal DCs, naïve mice were orally administered with 1mg of FITC-IK and FITC-IK NPs, respectively. After 6 h, small intestine was removed and flow cytometry analysis of the intestinal lamina propria mononuclear cells (LPMCs) was performed. The cell suspensions were stained with PE-conjugated anti-CD11c and APC-conjugated anti-CD103 (BD Biosciences Pharmingen). Isotype IgG was used as control antibodies. CD11c+CD103+ cells were gated for intestinal DCs and then FITC intensity was directly analyzed using a FACS Calibur Flow cytometer (BD Biosciences). The data were analyzed using Flowjo software (7.6.1).
For fluorescence analysis of in vivo cell uptake, intestinal cryosections of mice receiving free FITC-IK and FITC-IK NPs were prepared. The sections were incubated with DAPI in order to visualize nuclei. Images of positive staining were taken using a fluorescence microscope (Olympus, Tokyo, Japan).
Sensitization and challenge of mice
Animals
Female Balb/c mice (6 weeks) were purchased from the Experimental Animal Center (Guangzhou, China) and maintained in a specific pathogen-free environment. Mice were allowed to access food and water freely. The animal studies were carried out in accordance with the Institutional Guidelines for Animal Care and Use of Laboratory Animals at Shenzhen University and approved by the Animal Ethic Committee at Shenzhen University.
SIT treatment
Mice were randomly divided into 5 groups (10 mice per group): A) Control group: Mice were treated with normal saline (NS); B) Model group: Mice were sensitized to OVA and treated with NS; C) Blank NPs group: Sensitized mice were treated with blank NPs; D) IK/R848 physical mixture group: Sensitized mice were treated with a mixture of IK and R848; E) PPLA-IK/R848 NPs group: Sensitized mice were treated with PPLA-IK/R848 NPs. Each mouse of group D and E was received 50 μg IK and 250 μg R848 each time. All the groups were received oral administration every day for 6 times after sensitization (more information is available in supplementary information).
Sensitization and challenge of mice
FA mouse model was established based on previous studies with some modifications.Citation22–Citation24 Briefly, mice were immunized with 0.1 mg OVA per mouse in 0.2 mL NS plus 2 mg of Alum (Sigma, USA) via subcutaneous injection on day 0, 3 and 7. Then mice were challenged by oral administration of 50 mg OVA on day 14, 16 and 18. The control group was treated with an equal volume of NS. The mice temperatures and body weight were monitored and the emergence of allergic diarrhea was also observed within 30 min after challenge. 24 hrs after the final challenge, blood samples (500 μL) were taken from the mice eyes with local anesthesia before sacrifice and the serum was isolated for the measurement of OVA-specific antibodies. The small intestines were removed, fixed in 4% paraformaldehyde immediately for histopathological analysis or frozen at −80 °C for enzyme-linked immunosorbent assay (ELISA).
Flow cytometry analysis for Tregs
LPMCs from different groups were stained with FITC-conjugated anti-CD4 and APC-conjugated anti-CD25 (BD Biosciences Pharmingen) before fixation and intracellular labeling using a PE-conjugated Foxp3 antibody (BD Biosciences Pharmingen). Isotype IgG was used as control antibodies. Cells were analyzed using a FACS Calibur Flow cytometer (BD Biosciences). The data were analyzed using Flowjo software (7.6.1).
Statistical analysis
Statistical analysis was performed with SPSS 19.0 statistics software. Both Student’s t test and ANOVA two ways test were used to evaluate the differences between groups, and P<0.05 or less was considered significant.
Results and discussion
Preparation and characterization of PPLA-IK/R848 NPs
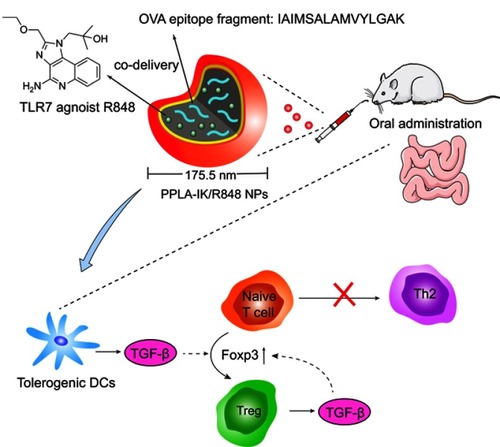
An OVA epitope fragment IAIMSALAMVYLGAK (IK) was synthesized with purity ≥95% and the sequences were confirmed by HPLC and LC-MS spectrometry analysis (Figure S1). PPLA-IK/R848 NPs were prepared with mPEG-PDLLA and Tween 80. They exhibited an average diameter of 175.5±6.5 nm and was about 110 nm larger than that of blank NPs (). The polydispersity index (PDI) value was 0.158±0.013 with a zeta potential of +2.81 mV. Cationic particles were effectively taken into DCs due to the attraction to negatively charged cell membranes.Citation25 Transmission electron microscope (TEM) image of PPLA-IK/R848 NPs showed a spherical and smooth surface (), which was smaller than that observed by dynamic light scattering (DLS) examination because the drying process resulted in particle shrinkage. The obtained PPLA-IK/R848 NPs nearly maintained their original size and no aggregation were observed in simulated gastric or intestinal fluids during the 12 h incubation. (), indicating that the obtained PPLA-IK/R848 NPs were suitable for oral administration. The size of the obtained NPs in our study is optimal for transport across the gut barrier and uptake by immune cells in the intestine.Citation26,Citation27 The in vitro drug release behavior of FITC-IK/R848 NPs was compared with FITC-IK/R848 physical mixture (dispersed directly in water) and the cumulative dissolution profile is shown in . The cumulative release profiles of FITC-IK/R848 NPs exhibited a two-phase release pattern. In the initial quick release phase, 68.1% of encapsulated FITC-IK and 46.8% of encapsulated R848 were released within 4h. This may be attributed to their adhesion to the outer layer of the NPs that are not being tightly bound and consequently are more easily released. This type of release is advantageous for NPs to stimulate the immune system because of the limited period of exposure to the mucosal surface.Citation28 The second phase consisted of constant continuous release of FITC-IK and R848 up to 72 h (3 days). Such a slow but continuous release is beneficial to build a long-lasting immune response because low dose of allergen immunotherapy favors the induction of local Tregs.Citation29 The results are in agreement with the previously published data of other nano-vaccine prepared by PLGA, which were shown to be retained for considerable periods of time, allowing for gradual and extended release of the encapsulation before biodegradation.Citation30,Citation31
Figure 1 Characterization of PPLA-IK/R848 NPs.
Notes: (A) The particle size distribution of PPLA-IK/R848 NPs and blank NPs. Each sample was measured in triplicate (12 scans); (B) TEM image of PPLA-IK/R848 NPs. The scale bar is 100 nm. (C) The stability of PPLA-IK/R848 NPs in simulated gastrointestinal fluids at 37 °C. (D) The in vitro cumulative release profile of FITC-IK and R848 in each formulation. The data are presented as mean ± SD, n=3.
Abbreviations: PPLA-IK/R848 NPs, OVA epitope fragment IK and R848 co-delivery nanoparticles; DLS, dynamic light scattering; TEM, Transmission electron microscope; SD, standard deviation.
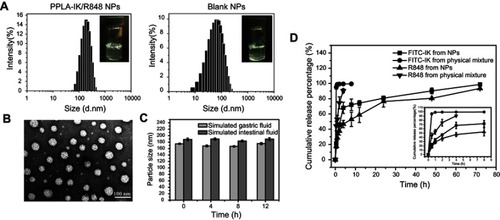
FITC-IK NPs can be highly taken by DC2.4 cells
FITC labeled IK was used to trace the cell internalization of IK in both, NPs and free in solution, by DC 2.4 cells. The green and blue fluorescence indicate FITC-IK and DAPI, respectively. The uptake of FITC-IK NPs and free FITC-IK by DC2.4 cells was in a concentration-dependent manner (). DC2.4 cells treated with FITC-IK NPs showed brighter green fluorescence than FITC-IK solution treated cells under the same concentration of FITC-IK, suggesting that FITC-IK NPs are more likely to be taken by DC2.4 cells. As reported by Silva et alCitation34, NPs that are approximately 200 nm or less can transit across lymphatic barrier and can be taken up by DCs. This may be attributed to one of the features of NPs that can attach to the cell membrane nonspecifically and then are internalized in the DCs via endocytosis.Citation32 DCs have superior capacity to cross-present exogenous antigen and are considered the major target of vaccines. Therefore, IK-carrying NPs can improve antigen processing, presentation and T cell priming by DCs compared to using free IK.Citation33,Citation34
Figure 2 Uptake of FITC-IK NPs and free FITC-IK by DC2.4 in vitro.
Notes: The nuclei are stained with DAPI (blue). The green staining indicates FITC-IK. The scale bars is 50 μm.
Abbreviations: FITC-IK NPs, FITC decorated IK loaded nanoparticles; DAPI, 2-(4-Amidinophenyl)-6-indolecarbamidine dihydrochloride.
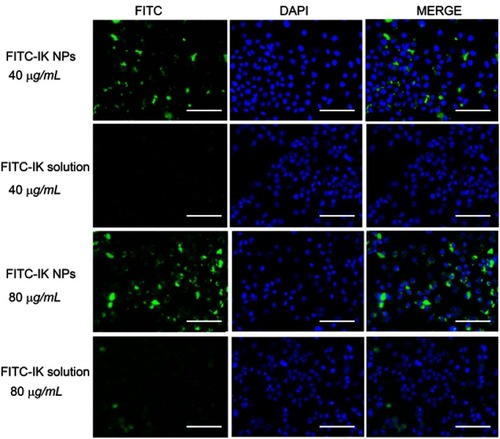
Tolerogenic DCs and Tregs can be induced by PPLA-IK/R848 NPs in vitro
DCs play a central role in the activation and polarization of T cells in vivo. To understand the effects of PPLA-IK/R848 NPs on DC’s functions and their roles in the induction of Tregs, bone marrow-derived DCs (BMDCs) were prepared with the bone marrows from naïve Balb/c mice and stimulated with blank NPs, IK/R848 mixture and PPLA-IK/R848 NPs in the culture, respectively. As shown by , exposure to PPLA-IK/R848 NPs significantly increased the percentage of TGF-β+DCs compared to IK/R848 mixture (10.70 vs 3.89%, P<0.001). The results of Western blot () also showed that DCs treated with PPLA-IK/R848 NPs expressed higher protein levels of TGF-β. TGF-β was reported to promote the conversion of naïve T cells into Foxp3+ Tregs.Citation35–Citation37 To test this, BMDCs were co-cultured with naïve CD4+ T cells. As expected (), co-culture of naïve CD4+ T cells with PPLA-IK/R848 NPs primed DCs induced about 11.2% Foxp3+Tregs, while the IK/R848 mixture primed DCs induced only 7.10% Foxp3+Tregs.
Figure 3 PPLA-IK/R848 NPs treated DCs display a tolerogenic phenotype and promote the differentiation of Tregs in vitro.
Notes: (A) The flow cytometry analysis of DCs after exposure to different formulations (denoted above each subpanel).The gated dot plots indicates CD11c+TGF-β+ cells. (B) The TGF-β levels in the protein extract of DCs after exposure to different formulations measured by Western blot. (C) The flow cytometry analysis of Tregs in CD4+T cells after co-cultured with different formulations primed-DCs. The data are presented as mean ± SD (**P<0.01, ***P<0.001).
Abbreviations: PPLA-IK/R848 NPs, OVA epitope fragment IK and R848 co-delivery nanoparticles; DC, dendritic cells; Tregs, regulatory T cells; TGF-β, transforming growth factor-β; SD, standard deviation.
The essential functions of DCs are to present the antigen information to T cells to initiate immune responses in the body. The induced types of immune responses are dependent on the activated DC phenotypes. TGF-β producing DCs have tolerogenic properties since TGF-β is an important factor in Treg development and suppressor functions.Citation38 The above results demonstrated that PPLA-IK/R848 NPs can induce much more tolerogenic DCs than IK/R848 mixture, which may be attributed to that more NPs are internalized into DCs via endocytosis. Several NPs-based drug delivery systems have emerged in DCs-based immunotherapy. For example, NPs chemically conjugated to antibodies against the c-type lectin receptor DEC205Citation39 or mannose receptor DC-SIGN that can target to human DCsCitation40 and the robust humoral or cellular responses can be achieved consequently. However, most NPs target to DCs focused on the activation of DCs and their immunostimulatory potential for DC is applied to tumor immunotherapy. Few studies have been reported to induce tolerogenic DCs and maintain immune tolerance in NPs-based drug delivery system. To our knowledge, this is the first description of NPs that promote the development of TGF-β producing tolerogenic DCs and the differentiation of Tregs via oral administration.
PPLA-IK/R848 NPs can be endocytosed by intestinal DCs
To determine if PPLA-IK/R848 NPs can be uptaken by intestinal DCs in vivo, naïve mice were orally administered with FITC-IK or FITC-IK NPs. After 6 h later, small intestine was removed and flow cytometry analysis of the LPMCs was performed. The results showed intestinal DCs captured more FITC-IK NPs than free FITC-IK (Figure S2A). Fluorescence microscope analysis of small intestinal tissue also demonstrated that more FITC-IK NPs were uptaken by intestinal cells than FITC-IK (Figure S2B). The results indicated that FITC-IK NPs are more likely endocytosed by intestinal DCs than free FITC-IK, which was in consistent with in vitro uptake study. The molecular weight of IK is 1552 Da. For many peptide drugs larger than 1000 Da, the optimized delivery systems are required to enhance the peptide permeation across the intestine barriers.Citation16 R848 is a hydrophobic imidazoquinoline compound. The solubilization of the drug would be a rate-limiting factor for intestine absorption.Citation41 Besides, the intestine content such as dietary residues, intestinal secretions, mucus, or colonic bacterial enzymes may have a negative influence on the stability of the drugs.Citation42 Polymeric particles are able to protect the encapsulated drug from gastrointestinal degradation and can bind to and possibly cross the epithelial barrier. The adhesion between the NPs and the intestines prolonged the contact between PPLA-IK/R848 NPs and the absorbing membranes of the gut and thus promote the cellular uptake in the intestine.Citation43,Citation44 Moreover, Pegylated NPs can protect against aggregation in the gut lumen and decrease the extent of enzymatic degradation of protein or peptide-like drugs.Citation45
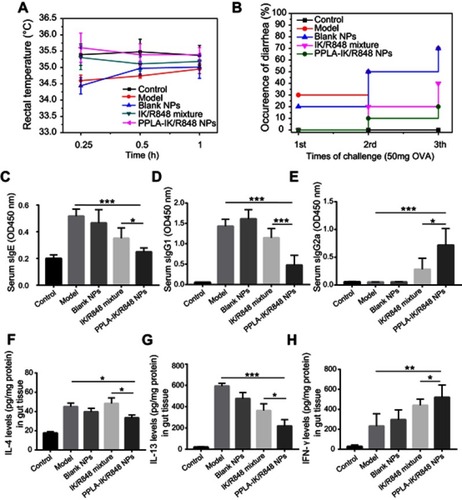
PPLA-IK/R848 NPs protected mice from OVA-induced allergic-inflammation in the intestine
To further test the therapeutic effect of PPLA-IK/R848 NPs in vivo, a mouse model of FA was developed. (Figure S3) The therapy in our study was executed by oral administration because it is a preferred route of immunotherapy in human subjects. The results showed that besides inducing the general allergic responses (, including the drop of core temperature, diarrhea and the enhancement of serum OVA-specific IgE), mice in model group and blank NPs treated group also showed other allergy-relevant responses, including increases in Th2 cytokines (), infiltration of mononuclear cells and eosinophils in the intestinal tissue (jejunum part, Figure S4). The results proved the FA model is successful and no therapeutic effects were resulted using blank materials. In mice treated with PPLA-IK/R848 NPs, allergic reactions were markedly suppressed. The PPLA-IK/R848 NPs showed little toxicity since there was no obvious body weight loss (Figure S5) and the pathological examinations of stomach, duodenum and liver exhibited insignificant differences between PPLA-IK/R848 NPs and control group (Figure S6). Serum IgG1 levels were closely associated with Th2-type responses.Citation46 PPLA-IK/R848 NPs group also showed a relatively lower level of OVA-sIgG1 compared with other groups that were sensitized and challenged with OVA (, P<0.001). Reversely, OVA-sIgG2a level (, a Th1-associated immunoglobulin in mice) and IFN-γ level were () significantly higher in PPLA-IK/R848 NPs group compared with model group. In conclusion, PPLA-IK/R848 NPs could shift immune response from a Th2 to a Th1 pattern. The results are consistent with a previous study in which mice received immunotherapy with a nanodispersion system carrying T cell epitope peptide and R848.Citation47 The hydrophilic T cell epitope peptide was delivered through mouse skin using solid-in-oil (S/O) nanodispersion system to treat pollinosis while PPLA-IK/R848 NPs was delivered by oral administration in our study to treat FA. Several reports indicate that R848 have the ability to shift the Th2 response toward Th1‐type response through activating Th1‐related lymphocytes.Citation48 Secretion of IL‐12, induction of IgG2a and reduction of IgE were reported after administration of R848,Citation49,Citation50 while T cell epitope itself also enhances the production of IgG subclass antibodies that are related to the Th1‐type immune response.Citation9
Figure 4 PPLA-IK/R848 NPs suppressed OVA-induced allergic reaction.
Notes: (A) The core temperature was recorded 30 min after the challenge with OVA. (B) Mice with diarrhea within 2 h after the challenge with OVA. (C–E) Serum levels of OVA-specific IgE (C), OVA-specific IgG1 (D) and OVA-specific IgG2a (E). (F–H) The protein levels of IL-4 (F), IL-13 (G) and IFN-γ (H) levels in intestine tissue. The data are presented as mean ± SD (*P<0.05, **P<0.01, ***P<0.001).
Abbreviations: PPLA-IK/R848 NPs, OVA epitope fragment IK and R848 co-delivery nanoparticles; SD, standard deviation.
Treatment with PPLA-IK/R848 NPs increases ova-specific Tregs in the intestine
The switch from Th2 to Th1 immune response needs to be well coordinated, since an uncontrolled bias towards Th1 response may lead to other disease.Citation51 Induction of Tregs may suppress excessive proliferation of both Th1 and Th2 cells due to its immune suppressive ability on both T cell subsets.Citation52 To investigate the effects of PPLA-IK/R848 NPs on modulating the immune response, LPMCs were isolated from the small intestine and the frequency of Tregs was assessed by flow cytometry (). The results showed that treatment with PPLA-IK/R848 NPs significantly increased the frequency of Tregs as compared with the model group (P<0.001) and IK/R848 mixture treated group (P<0.05).
Figure 5 PPLA-IK/R848 NPs increase OVA-specific Tregs in the intestine.
Notes: (A) Isolated LPMCs from different groups were staining for Tregs by flow cytometric analysis. The bars show summarized data of Tregs as shown by the dot plots. (B) The protein levels of TGF-β and IL-10 levels in intestine tissue. The data are presented as mean ± SD (*P<0.05, **P<0.01, ***P<0.001).
Abbreviations: PPLA-IK/R848 NPs, OVA epitope fragment IK and R848 co-delivery nanoparticles; LPMCs, lamina propria mononuclear cells; Tregs, regulatory T cells; TGF-β, transforming growth factor-β; Foxp3, Forkhead box protein 3; SD, standard deviation.
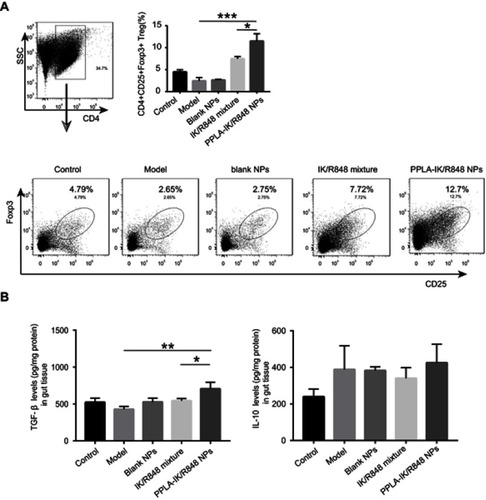
It is known that immune suppression for tolerance and maintenance of immune homeostasis depends on immunosuppressive cytokines such as IL-10 or TGF-β, which are produced by Tregs,Citation53,Citation54 we also assessed the protein level of TGF-β and IL-10 in the small intestine (). Surprisingly, there are no significant differences in the levels of IL-10 among these groups, whereas significantly higher production of TGF-β was detected in mice treated with PPLA-IK/R848 NPs. This might be explained by that R848 induced Tregs mediated suppression of allergic disease mainly through a TGF-β-dependent pathway.Citation12 Although our study lacks the analysis of DC phenotypes in vivo, we assumed that PPLA-IK/R848 NPs described in the present study which could be more easily taken in by DCs were capable of inducing more tolerogenic DCs that promote the differentiation of allergen-specific Tregs compared with IK/R848 mixture.
Conclusion
In summary, the encapsulation in PPLA-IK/R848 NPs showed a slower release and a higher uptake by DC2.4 cells compared to IK/R848 mixture. Oral administration of PPLA-IK/R848 NPs could efficiently deliver the IK and R848 to the intestine to be phagocytosed by DCs so that they could stimulate DCs into allergen tolerogenic phenotype and promote the differentiation of Tregs in the intestine via a TGF-β-dependent mechanism, which significantly protected mice from food allergic responses. These findings demonstrated that the co-delivery strategy of allergen epitope fragments and R848 using NPs can significantly protect mice from FA through the recovery of immune tolerance.
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 81460252, No.31570932); Basic Research Project of Science and Technology Program of Shenzhen (No.JCYJ20160429171931438, No.JCYJ20180306171552582) and China Postdoctoral Science Foundation (2017M622662).
References
- Dang TD, Allen KJ, J Martino D, Koplin JJ, Licciardi PV, Tang ML. Food-allergic infants have impaired regulatory T-cell responses following in vivo allergen exposure. Pediatr Allergy Immunol. 2016;27(1):35–43. doi:10.1111/pai.1249826456457
- Venter C, Arshad SH. Epidemiology of food allergy. Pediatr Allergy Immunol. 2011;58(2):42–50.
- Mansoor DK, Sharma HP. Clinical presentations of food allergy. Pediatr Clin North Am. 2011;58(2):315–326. doi:10.1016/j.pcl.2011.02.00821453804
- Sicherer SH. Epidemiology of food allergy. Pediatr Allergy Immunol. 2011;127(3):594.
- Varshney P, Jones SM, Scurlock AM, et al. A randomized controlled study of peanut oral immunotherapy: clinical desensitization and modulation of the allergic response. J Allergy Clin Immunol. 2011;127(3):654–660. doi:10.1016/j.jaci.2010.12.111121377034
- Chin SJ, Vickery BP, Kulis MD, et al. Sublingual versus oral immunotherapy for peanut-allergic children: a retrospective comparison. J Allergy Clin Immunol. 2013;132(2):476–478. doi:10.1016/j.jaci.2013.02.01723534975
- Anagnostou K, Clark A, King Y, Islam S, Deighton J, Ewan P. Efficacy and safety of high-dose peanut oral immunotherapy with factors predicting outcome. Clin Exp Allergy. 2011;41(9):1273–1281. doi:10.1111/j.1365-2222.2011.03699.x21414048
- Wai CYY, Leung NYH, Leung PSC, Chu KH. Immunotherapy of food allergy: a comprehensive review. Clin Rev Allergy Immunol. 2019; 57(1):55–73. doi:10.1007/s12016-017-8647-y.
- Prickett SR, Rolland JM, O’Hehir RE. Immunoregulatory T cell epitope peptides: the new frontier in allergy therapy. Clin Exp Allergy. 2015;45(6):1015–1026. doi:10.1111/cea.1255425900315
- Chhiba KD, Singh AM, Bryce PJ. New developments in immunotherapies for food allergy. Immunotherapy. 2015;7(8):913–922. doi:10.2217/IMT.15.5526268849
- Gupta K, Kumar S, Das M, Dwivedi PD. Peptide based immunotherapy: A pivotal tool for allergy treatment. Int Immunopharmacol. 2014;19(2):391–398. doi:10.1016/j.intimp.2014.01.03024530919
- Van LP, Bardel E, Gregoire S, et al. Treatment with the TLR7 agonist R848 induces regulatory T-cell-mediated suppression of established asthma symptoms. Eur J Immunol. 2011;41(7):1992–1999. doi:10.1002/eji.20104091421480211
- Qu S, Qin T, Li M, et al. The effects of resiquimod in an ovalbumin-induced allergic rhinitis model. Int Immunopharmacol. 2018;59:233–242. doi:10.1016/j.intimp.2018.04.01529665497
- Hao Y, Dong M, Zhang T, et al. A novel approach of using near-infrared responsive PEGylated gold nanorod coated poly (L-Lactide) microneedles to enhance the antitumor efficiency of docetaxel loaded MPEG-PDLLA micelles for treating A431 tumor. ACS Appl Mater Interfaces. 2017;9(18):15317. doi:10.1021/acsami.7b0360428418236
- Xiang GH, Hong GB, Wang Y, Cheng D, Zhou JX, Shuai XT. Effect of PEG-PDLLA polymeric nanovesicles loaded with doxorubicin and hematoporphyrin monomethyl ether on human hepatocellular carcinoma HepG2 cells in vitro. International Journal of Nanomedicine 2013;8(1):4613–4622.
- Doak BC, Over B, Giordanetto F, Kihlberg J. Oral druggable space beyond the rule of 5: insights from drugs and clinical candidates. Chem Biol. 2014;21(9):1115–1142. doi:10.1016/j.chembiol.2014.08.01325237858
- Yuan J, Guo L, Wang S, et al. Preparation of self-assembled nanoparticles of epsilon-polylysine-sodium alginate: a sustained-release carrier for antigen delivery. Colloids Surf B Biointerfaces. 2018;171:406–412. doi:10.1016/j.colsurfb.2018.07.05830071482
- Peng X, Liang Y, Yin Y, Liao H, Li L. Development of a hollow mesoporous silica nanoparticles vaccine to protect against house dust mite induced allergic inflammation. Int J Pharm. 2018;549(1–2):115–123. doi:10.1016/j.ijpharm.2018.07.04730040973
- Shen J, Burgess DJ. Drugs for Long Acting Injections and Implants. US: Springer; 2012.
- Rahimian S, Fransen MF, Kleinovink JW, et al. Polymeric nanoparticles for co-delivery of synthetic long peptide antigen and poly IC as therapeutic cancer vaccine formulation. J Control Release. 2015;203:16–22. doi:10.1016/j.jconrel.2015.02.00625660830
- Wang H, Mo L, Xiao X, et al. Pplase of Dermatophagoides farinae promotes ovalbumin-induced airway allergy by modulating the functions of dendritic cells in a mouse model. Sci Rep. 2017;7:43322. doi:10.1038/srep4332228240301
- Castillo-Courtade L, Han S, Lee S, Mian FM, Buck R, Forsythe P. Attenuation of food allergy symptoms following treatment with human milk oligosaccharides in a mouse model. Allergy. 2015;70(9):1091–1102. doi:10.1111/all.1265025966668
- Yamaki K, Yoshino S. Preventive and therapeutic effects of rapamycin, a mammalian target of rapamycin inhibitor, on food allergy in mice. Allergy. 2012;67(10):1259–1270. doi:10.1111/all.1200022913509
- Yamashita H, Takahashi K, Tanaka H, Nagai H, Inagaki N. Overcoming food allergy through acquired tolerance conferred by transfer of Tregs in a murine model. Allergy. 2012;67(2):201–209. doi:10.1111/j.1398-9995.2011.02742.x22050332
- Skwarczynski M, Toth I. Recent advances in peptide-based subunit nanovaccines. Nanomedicine. 2014;9(17):2657–2669. doi:10.2217/nnm.14.18725529569
- Gaumet M, Gurny R, Delie F. Localization and quantification of biodegradable particles in an intestinal cell model: the influence of particle size. Eur J Pharm Sci. 2009;36(4):465–473. doi:10.1016/j.ejps.2008.11.01519124077
- Griffin BT, Guo J, Presas E, Donovan MD, Alonso MJ, O’Driscoll CM. Pharmacokinetic, pharmacodynamic and biodistribution following oral administration of nanocarriers containing peptide and protein drugs ☆. Adv Drug Deliv Rev. 2016;106(Pt B):367. doi:10.1016/j.addr.2016.06.00627320644
- Aguado MT, Lambert PH. Controlled-release vaccines-biodegradable Polylactide/Polyglycolide (PL/PG) microspheres as antigen vehicles. Immunobiology. 1992;184(2–3):113–125. doi:10.1016/S0171-2985(11)80470-51587538
- Maeta A, Matsushima M, Muraki N, et al. Low-dose oral immunotherapy using low-egg-allergen cookies for severe egg-allergic children reduces allergy severity and affects allergen-specific antibodies in serum. Int Arch Allergy Immunol. 2018. doi:10.1159/000485891
- Joshi VB, Adamcakovadodd A, Jing X, et al. Development of a poly (lactic-co-glycolic acid) particle vaccine to protect against house dust mite induced allergy. Aaps J. 2014;16(5):975–985. doi:10.1208/s12248-014-9624-524981892
- Marazuela EG, Prado N, Moro E, Villalba M, Rodríguez R, Batanero E. Intranasal vaccination with poly(lactide‐co‐glycolide) microparticles containing a peptide T of Ole e 1 prevents mice against sensitization. Clin Exp Allergy. 2008;38(3):520–528. doi:10.1111/j.1365-2222.2007.02922.x18205856
- Tonigold M, Mailänder V. Endocytosis and intracellular processing of nanoparticles in dendritic cells: routes to effective immunonanomedicines. Nanomedicine. 2016;11(20):nnm-2016–0195. doi:10.2217/nnm-2016-0195
- Tacken P, De-Vries IR, Figdor C. Dendritic-cell immunotherapy: from ex vivo loading to in vivo targeting. Nat Rev Immunol. 2007;7(10):790–802. doi:10.1038/nri217317853902
- Silva AL, Rosalia RA, Varypataki E, Sibuea S, Ossendorp F, Jiskoot W. Poly-(lactic-co-glycolic-acid)-based particulate vaccines: particle uptake by dendritic cells is a key parameter for immune activation. Vaccine. 2015;33(7):847–854. doi:10.1016/j.vaccine.2014.12.05925576216
- Chen W, Konkel JE. Development of thymic Foxp3(+) regulatory T cells: TGF-β matters. Eur J Immunol. 2015;45(4):958–965. doi:10.1002/eji.20144499925684698
- Eusebio M, Kuna P, Kraszula L, Kupczyk M, Pietruczuk M. Allergy-related changes in levels of CD8+CD25+FoxP3(bright) Treg cells and FoxP3 mRNA expression in peripheral blood: the role of IL-10 or TGF-beta. J Biol Regul Homeost Agents. 2014;28(3):461–470.25316133
- Coombes JL, Siddiqui KRR, Arancibiacárcamo CV, et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β– and retinoic acid–dependent mechanism. J Exp Med. 2007;204(8):1757–1764. doi:10.1084/jem.2007059017620361
- Jia L, Lu J, Zhou Y, et al. Tolerogenic dendritic cells induced the enrichment of CD4+Foxp3+ regulatory T cells via TGF-β in mesenteric lymph nodes of murine LPS-induced tolerance model. Clin Immunol.2018;197:118–129. doi:10.1016/j.clim.2018.09.010.
- Dhodapkar MV, Mario S, Biwei Z, et al. Induction of antigen-specific immunity with a vaccine targeting NY-ESO-1 to the dendritic cell receptor DEC-205. Sci Transl Med. 2014;6(232):232ra251. doi:10.1126/scitranslmed.3008068
- Fan Y, Moon JJ. Nanoparticle drug delivery systems designed to improve cancer vaccines and immunotherapy. Vaccines. 2015;3(3):662–685. doi:10.3390/vaccines303066226350600
- Ulapane KR, Kopec BM, Meg M, Siahaan TJ. Peptides and drug delivery. Oxygen Transp Tissue XXXIII. 2017;1030:167–184. doi:10.1007/978-3-319-66095-0_8.
- Philip AK, Philip B. Colon targeted drug delivery systems: a review on primary and novel approaches. Oman Med J. 2010;25(2):79–87. doi:10.5001/omj.2010.24
- Lautenschläger C, Schmidt C, Lehr CM, Fischer D, Stallmach A. PEG-functionalized microparticles selectively target inflamed mucosa in inflammatory bowel disease. Eur J Pharm Biopharm. 2013;85(3):578–586. doi:10.1016/j.ejpb.2013.09.01624084650
- Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery ☆. Adv Drug Deliv Rev. 2016;99(Pt A):28–51. doi:10.1016/j.addr.2015.09.01226456916
- Pawar VK, Meher JG, Singh Y, Chaurasia M, Reddy BS, Chourasia MK. Targeting of gastrointestinal tract for amended delivery of protein/peptide therapeutics: strategies and industrial perspectives. J Controlled Release. 2014;196:168–183. doi:10.1016/j.jconrel.2014.09.031
- Ochel M, Vohr HW, Pfeiffer C, Gleichmann E. IL-4 is required for the IgE and IgG1 increase and IgG1 autoantibody formation in mice treated with mercuric chloride. J Immunol. 1991;146(9):3006–3011.2016536
- Kitaoka M, Naritomi A, Kawabe Y, Kamihira M, Goto M. Transcutaneous pollinosis immunotherapy using a solid-in-oil nanodispersion system carrying T cell epitope peptide and R848. Bioeng Transl Med. 2017;2(1):102–108. doi:10.1002/btm2.1004829313026
- Brugnolo F, Sampognaro S, Liotta F, et al. The novel synthetic immune response modifier R-848 (Resiquimod) shifts human allergen-specific CD4+ TH2 lymphocytes into IFN-gamma-producing cells. J Allergy Clin Immunol. 2003;111(2):380–388. doi:10.1067/mai.2003.10212589360
- Shen E, Lu L, Wu C. TLR7/8 ligand, R-848, inhibits IgE synthesis by acting directly on B lymphocytes. Scand J Immunol. 2010;67(6):560–568. doi:10.1111/j.1365-3083.2008.02105.x
- Siebeneicher S, Reuter S, Krause M, et al. Epicutaneous immune modulation with Bet v 1 plus R848 suppresses allergic asthma in a murine model. Allergy. 2014;69(3):328–337. doi:10.1111/all.1232624329861
- Cui J, Pazdziorko S, Miyashiro JS, et al. T H 1-mediated airway hyperresponsiveness independent of neutrophilic inflammation. J Allergy Clin Immunol. 2005;115(2):309–315. doi:10.1016/j.jaci.2004.10.04615696086
- Stock P, Dekruyff RH, Umetsu DT. Inhibition of the allergic response by regulatory T cells. Curr Opin Allergy Cli Immunol. 2006;6(1):12–16. doi:10.1097/01.all.0000200502.69672.44
- Palomares O, Yaman G, Azkur AK, Akkoc T, Akdis M, Akdis CA. Role of Treg in immune regulation of allergic diseases. Eur J Immunol. 2010;40(5):1232–1240. doi:10.1002/eji.20094004520148422
- Du PM, Samsom JN. Adaptive T-cell responses regulating oral tolerance to protein antigen. Allergy. 2011;66(4):478–490. doi:10.1111/j.1398-9995.2010.02519.x21143239
