Abstract
Purpose
Assessing the risks and preventable causes of maternal and neonatal mortality requires the availability of good-quality antenatal information. In Indonesia, however, access to reliable information on pregnancy-related results remains challenging. This research has proposed a research-based policy recommendation to improve availability and accessibility to vital information on antenatal examinations.
Patients and Methods
Descriptive statistics were used to characterize midwives’ capabilities in routinely gathering and recording antenatal information during pregnancy. The investigation was carried out among 19 midwives in South Kalimantan, Indonesia, from April 2016 to October 2017. Antenatal data on 4946 women (retrospective study) and 381 women (prospective study) have been accessed through a scientific and technical training program.
Results
To date, lack of timely access to antenatal information has hampered the process of reducing neonatal mortality in Indonesia. The post-training statistical analysis showed that the training has significantly improved midwives’ scientific knowledge and technical abilities in providing more reliable data on antenatal measurements.
Conclusion
Consistent scientific and technical training among midwives is required to update their knowledge and skills, particularly those relating to documenting the results of antenatal examinations at different stages of pregnancy and using that information to assess potential risks and identify necessary interventions. This should also be followed by routine monitoring on the quality of collected antenatal data. This can be one of the enabling actions to achieve the 2030 Sustainable Development Goals target in reducing neonatal mortality in Indonesia.
Introduction
Antenatal care (ANC) is a preventive intervention to promote optimal pregnancy results. Access to this has been recognized as one of the best mediations to avoid poor well-being. ANC services are required to conform to the quality standard of ANC to improve maternal and child health, including recording and reporting the after-effects of ANC assessments.Citation1,Citation2 This investment enables the provision of adequate data to be analyzed and utilized for informed planning, decision making and monitoring policy progress to lower the risks of having maternal and neonatal mortality.Citation3–Citation5
Suitable utilization of antenatal data and its methodical examination during different periods of pregnancy are vital when observing, identifying, and measuring the risks and preventable factors associated with maternal and neonatal mortality. In Indonesia, the access to routine information on pregnancy-related results, causes and the effects of intercessions is deficient. This prevents actions to diminish maternal, fetal, and neonatal mortality, such as planning programs, resource allocation, and decision making.Citation2,Citation6-Citation8
World-side improvement of antenatal data accessibility, consistency, and quality has turned out to be badly needed in improving well-being for babies.Citation4,Citation9-Citation11 Although good quality of data will not solely improve the life survival of both mother and infant,Citation5 it can enable medical practitioners to identify the signs of abnormalities; thus, appropriate interventions can be initiated in an expedient way.Citation4,Citation5,Citation9-Citation11
In Indonesia, midwives are the primary practitioners across territories.Citation12,Citation13 They are required to give comprehensive antenatal support to pregnant women and document the eventual outcomes of evaluations on local health recording and reporting systems.Citation2 They are moreover expected to detect alarm signals of potential risks and provide proper interventions or referrals during pregnancy and delivery. However, their abilities in reporting the results of antenatal evaluations are low both in hospitals (20%) and primary healthcare (PHC) centres (42.5%).Citation8 Unrecorded data or an inability to access local data on maternal, fetal, and neonatal characteristics have been perceived as a significant reason in hindering evidence-based interventions to evaluate the causes and predictable components related to maternal and newborn mortality.Citation10,Citation14
In developing countries, access to individual data of mother, fetus, and neonate during ANC remained inadequate. Although many factors influence the well-being of the fetus, routine and reliable measurement of fundal height (FH) at a given gestation age (GA) throughout pregnancy remains significant to estimate fetal weightCitation15 and monitor fetal growth during pregnancy.Citation16–Citation22 In Indonesia, the lack of these data hampered the development of standard growth charts for fetus and neonate.Citation23
The measurement of FH is straightforward and less expensive. It remains a significant screening tool during routine ANC, particularly in rural areas where ultrasound facilities are not always accessible.Citation20,Citation24 When the information of the last menstrual period (LMP) is unreliable and ultrasonic data are not available, FH can potentially be used as a proxy to estimate GA, which is one of the basic measures to recognize preterm birth and low birth weight (LBW).Citation4 Therefore, the examination of FH records can help midwives to improve the quality of maternal and neonatal care.Citation2,Citation25
The aim of this investigation was to propose a research-based policy recommendation to improve the awareness and ability among midwives on the importance of routinely recording essential data on antenatal measurements, essentially for FH measurements.
Materials and Methods
Research Aim and Design
This research is the extension of the two earlier published studies, the details have been provided elsewhere.Citation26,Citation27 The primary aim was to propose a research-based policy recommendation, based on the results of the two previously published research, to improve the awareness and ability among midwives on the importance of routinely recording essential data on antenatal measurements. This is particularly for FH measurements that are used to estimate fetal weight at different stages of pregnancy in the absence of ultrasound facilities.
This research has been conducted using a descriptive and exploratory design using both quantitative and qualitative methods. During the quantitative phase, local pregnancy registers were reviewed to evaluate midwives’ ability to collect and correctly record the ANC examination results before and after scientific and technical training. After the training program, electronic questionnaires were distributed to the midwives to qualitatively gather information regarding their opinions on the challenges of documenting routine ANC examination results.
Research Setting
This research was conducted between April 2016 and October 2017 in one province of Indonesia, South Kalimantan with the estimated population of almost 4 million in 2015.Citation28 This province is one of the five provinces with the highest neonatal mortality rates (NMR) in Indonesia.Citation29–Citation31 Administratively South Kalimantan is structured as two municipalities (urban areas) – Banjarmasin and Banjarbaru – and eleven districts (rural areas) – Banjar, Barito Kuala, Tapin, Hulu Sungai Selatan, Hulu Sungai Tengah, Hulu Sungai Utara, Balangan, Tabalong, Tanah Laut, Tanah Bumbu and Kotabaru. All these urban and rural areas were proportionally represented in the study.
In the capital of the province, public and private hospitals are available as tertiary health facilities. Each administrative area is served by hospitals as secondary health facilities that provide referral services in that area and health centres as primary health facilities. In this study, primary health care centres are the main concern because they are located near where people live and are the most commonly recommended and most cost-effective first level of health care in Indonesia, particularly in rural settings.Citation6,Citation32,Citation33
There are two common types of primary health care centres in South Kalimantan province: public health centres (Puskesmas or PHCs) and independent midwifery practices (Praktik Bidan Mandiri or IMPs). These facilities are situated in both urban and rural areas to ensure services are equally accessible and distributed across the communities. In total, there are 230 PHCsCitation28 and 187 IMPs (unpublished data from the provincial midwifery association in 2015) across the province.
Research Participants
For this study, 14 PHCs and 5 IMPs, proportionally distributed across the administrative areas of the province, were selected using multistage purposive sampling with the type of healthcare system and administrative location as the stratification factors. The selection criteria for the individual strata were based on the population density of the PHC’s catchment area, taken in conjunction with recommendations from the provincial midwifery association. Being administered by the Ministry of Health of the Republic of Indonesia, the participating health centres all followed the standard operational procedures for ANC.Citation34,Citation35 Therefore, it is assumed that the systematic variation between midwives’ measurements is not significant and has been ignored. The study also involved 19 midwives who had experienced in providing ANC services for a minimum of 5 years at the selected PHCs.
Research Instrument and Data Collection
The detailed information on research instrument and data collection procedures can be found in the formerly published studies.Citation26,Citation27 presents the summary.
Table 1 Summary of Research Instrument and Data Collection
Figure 1 Retrospective data for assessing training outcomes.
Notes: Adapted from Anggraini D, Abdollahian M, Marion K, et al. The impact of scientific and technical training on improving routine collection of antenatal care data for maternal and foetal risk assessment: a case study in the Province of South Kalimantan, Indonesia. J Pregnancy. 2018;2018. Copyright © 2018 Dewi Anggraini et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.Citation26
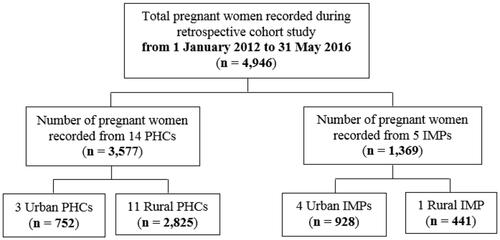
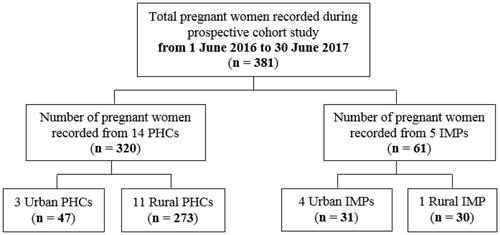
Figure 2 Prospective data for assessing training outcomes.
Notes: Adapted from Anggraini D, Abdollahian M, Marion K, et al. The impact of scientific and technical training on improving routine collection of antenatal care data for maternal and foetal risk assessment: a case study in the Province of South Kalimantan, Indonesia. J Pregnancy. 2018;2018. Copyright © 2018 Dewi Anggraini et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.Citation26
Statistical Analysis
The detailed information of statistical analysis used to assess the quality of routine ANC data collection can be found in the earlier published studies.Citation26,Citation27 The summary is given in .
Table 2 Summary of Statistical Analysis
Results
Baseline Characteristics of Midwives
Overall, the average age of the participating midwives was 41 years (29–56 years) with the average working experience in antenatal and midwifery services was 19 years. The descriptive summary of midwives’ characteristics is given in .
Table 3 Description of Midwives’ Characteristics (Number of Participating Midwives = 19)
General Information on the Study Population (Retrospective Study, n = 4946 Manually Recorded Data)
In total, 14 public health centres (PHCs) and 5 independent midwifery practices (IMPs) were involved in the study. Between 1 January 2012 and 31 May 2016, 4946 pregnant women received ANC services in these centres and their information was manually recorded (). Descriptive statistics on the baseline characteristics of mothers and newborns in the study population are presented in .
Table 4 Baseline Characteristics and Antenatal Events (Retrospective Study, Number of Pregnant Women = 4946)
Not all pregnant women included in the retrospective study attained the minimum recommendation of 4 ANC visits (range between 1 and 12 visits). Overall, the pregnant women were between 23 and 32 years old (43.2%), well nourished (60.1%) with middle-upper arm circumference (MUAC) ≥23.5 cm, nulliparous (24.4%) and had normal BMI at the first visit of ANC (between 18.5 and 24.9 kg/m2) (7.2%). Of these 4946 women, 1446 (29.2%) delivered the baby at term pregnancy, 560 (11.3%) delivered the baby through the spontaneous normal mode of delivery, 693 (14%) delivered preterm (<37 completed weeks of gestation); 1010 (20.4%) delivered male newborns, 967 (19.6%) delivered female newborns; and 987 (20.0%) were assisted by midwives. Before scientific and technical training among midwives, the information gap of each study population characteristic was substantial (from 17.2% to 88.9%) ().
Description of the Study Population (Prospective Study, n = 381)
Between 1 June 2016 and 30 June 2017, there were 381 pregnant women who enrolled, received ANC and were followed up until they gave birth at the study centers. Descriptive statistics on the baseline characteristics of mothers and newborns in the study population are presented in .
Table 5 Baseline Characteristics and Antenatal Events (Prospective Study, Number of Pregnant Women = 381)
Most pregnant women included in this study followed the minimum recommendation of ANC visits (average of 6 visits) with a range between 1 and 14 visits. Overall, the pregnant women were between 23 and 32 years old (61.2%), well-nourished (28.65%) with middle-upper arm circumference (MUAC) ≥23.5 cm, multiparous (71.4%) and had normal BMI at the first visit of ANC (between 18.5 and 24.9 kg/m2) (67.7%). Of these 381 women, 348 (91.3%) delivered the baby at term pregnancy, 346 (90.8%) delivered the baby through the spontaneous normal mode of delivery, 33 (8.7%) delivered preterm (<37 completed weeks of gestation); 213 (55.9%) delivered male newborns, 164 (43.0%) delivered female newborns; and 302 (79.3%) were assisted by midwives. After training, the information gap of each study population characteristic (prospective study) is smaller (from 0.32% to 68.5%) ().
The Impact of Scientific and Technical Training on Improving Routine Collection of ANC Data
Based on our reviews in the previous study,Citation26 scientific and technical training has generally and significantly improved the average amount (%) of recorded ANC data across all primary health care providers. A two-sample t-test shows an overall increase in data recorded from 17.5% to 62.1% (p-value <0.0005) ().
Table 6 Data Recorded Before and After Training
The outcomes also demonstrate a general improvement, especially in recording individual data, obstetric history, delivery plans, ANC utilization criteria, maternal measurements, provision of supplements, clinical fetal estimates and delivery time (from 0.8–47.9% to 57.4–99.2%). These results have been shown and discussed in the previous study.Citation26 Nonetheless, midwives’ responsiveness to the significance of gathering and recording the results of laboratory tests, maternal risk detection, ultrasonic fetal estimates and fetal risk detection provide an opportunity for improvement (<12%). A comprehensive investigation that compares the performance of midwives in documenting the results of the antenatal examinations before and after the training program has been done and published in the previous studies.Citation26,Citation27
The Impact of Scientific and Technical Training on Improving Routine Collection of FH Measurements
Scientific and technical training has also significantly improved the awareness and ability among midwives in recording FH measurements. shows that the training has increased the average amount of recorded FH information across PHC providers, from 37.1% to 58.5% (p-value < 0.0005).
Table 7 Fundal Height Assessment Recorded Before and After Training
In Indonesia, the Johnson-Toshach modelCitation37 that incorporates maternal FH and fetal station/descent level (FS) is well recognized and performed to predict fetal weight. In this study, however, the attention is not on the accuracy of the equation that has been assessed and talked about in the past investigation,Citation15 but on how accurately the information of FH was recorded on the manual register (before training) and electronic pregnancy register (after training).
presents the ability of the midwives in correctly recording FH measurements which expected to increase during the pregnancy.
Figure 3 FH and GA data available for fetal weight estimation. [Source: Anggraini, et al. Citation38]. (A) before training (retrospective data, n = 195). (B) after training (prospective data, n = 402).
![Figure 3 FH and GA data available for fetal weight estimation. [Source: Anggraini, et al. Citation38]. (A) before training (retrospective data, n = 195). (B) after training (prospective data, n = 402).](/cms/asset/2ec2ec39-8213-42fd-9e51-1d1dd2398a4d/djwh_a_12171254_f0003_c.jpg)
presents the ability of the midwives in correctly recording FH measurements which expected to increase during the pregnancy. (A) clearly shows that before training (retrospective cohort study), some of the recorded FH have not increased by GA. The relationship pattern between FH and GA could not be reliably seen when the data were retrospectively collected. This can be justified by the low correlation value between FH and GA recorded by r = 0.616 (p-value < 0.0005).
However, after training, the awareness and ability among midwives in correctly documenting FH information were significantly improved [(B)]. The increase in FH can now obviously be seen by the increase of GA when the data were prospectively collected. This can be justified by the higher correlation value between FH and GA recorded by r = 0.851 (p-value < 0.0005).
presents a similar pattern between FH and GA at different stages of pregnancy for prospective data.
Figure 4 Fundal height growth assessment during critical periods of pregnancy. [Source: Anggraini, et al. Citation38]. (A) 13–42 weeks (n = 402). (B) 20–40 weeks (n = 402). (C) 20–35 weeks (n = 399). (D) 24–40 weeks (n = 401)
![Figure 4 Fundal height growth assessment during critical periods of pregnancy. [Source: Anggraini, et al. Citation38]. (A) 13–42 weeks (n = 402). (B) 20–40 weeks (n = 402). (C) 20–35 weeks (n = 399). (D) 24–40 weeks (n = 401)](/cms/asset/c9889e59-f4ea-43b8-96c7-59e2ce31cdcb/djwh_a_12171254_f0004_c.jpg)
Factors Affecting Midwives in Completing Routine ANC Data Documentation Tasks
summarizes the main contributing factors affecting midwives in completing ANC data records and the main reasons for the delay in collecting and reporting routine ANC information.
Table 8 Factors and Reasons Influencing Midwives in Completing Routine Collection of ANC Data
The majority of the participating midwives (n = 17, 89.5%) responded to the effectiveness of the training and 52.6% of them responded to the importance of the developed electronic register, particularly if it could be computationally and automatically linked with the monthly ANC reporting formats. These results have been reported in the previously published work.Citation27
Discussion
Most of the midwives were senior midwives with working experience ranged between 6 and 36 years in ANC services across urban and rural primary health care facilities. They also followed the standard operational procedures for ANC measurements.Citation34,Citation35
Overall, the pregnant women, who were retrospectively and prospectively included in the study, were between 23 and 32 years old. They were well nourished with middle-upper arm circumference (MUAC) ≥ 23.5 cm, had normal BMI at the first visit of ANC (between 18.5 and 24.9 kg/m2), delivered the baby at term pregnancy through the spontaneous normal mode of delivery and were assisted by midwives.
Currently, local health registers, as opposed to periodic household surveys, are utilized to identify risks of maternal and neonatal mortality.Citation6,Citation7,Citation38 With the development of a decentralization approach in Indonesia,Citation6,Citation7 access to timely and reliable local information and the accumulation of maternal and fetal measurements during ANC is urgently required. Given appropriate education and training, monitoring and control as well as innovation; local maternal, fetal, and neonatal well-being information can be utilized as consistent baseline data to improve the quality of care services.Citation38
Access to individual data on maternal and fetal well-being during pregnancy and at delivery is fundamental to fortify routine health data frameworks. The analysis presented in the previous section clearly shows that scientific and technical training has equipped urban and rural midwives with the knowledge of the importance of ANC data measurement and documentation. The improvement of their essential midwifery care in recording the after-effects of ANC assessments was overall greater (62.1%) than the present national report (42.5%).Citation8 This was particularly evident in the gathering and recording of the critical attributes, such as FH, to evaluate the dangers during pregnancy. If the excellent quality of care can be related to its successful information accumulation during labor,Citation11 then it is also crucial to have real-time data collection at various phases of pregnancy to enhance the quality of ANC.
Moreover, the awareness of midwives in significantly recording FH at a certain GA to estimate the weight of fetus has fundamentally improved after the training (from 0–16.5% to 62.2–65.8%).Citation26 However, the information sufficiency stayed underneath 70%. A comparable pattern of low percentage in recording FH was found in rural Australia (range 36–76%).Citation39 Although many variables impact the well-being of the fetus, FH stays as one of the most prescribed and accessible indicators to estimate fetal weightCitation15 and screen fetal development during pregnancyCitation16–Citation22 to identify preterm birth and LBW.Citation4
The arrangement of the fetal growth chart during pregnancy is critical to evaluate the feasibility of the fetus at various phases of pregnancy and to guarantee neonatal endurance and prosperity. The chart is a compelling screening tool to help midwives in analyzing and identifying the dangers of fetal development variations from the norm, for example, prematurity and LBW. Subsequently, preventive activities and referrals can be adequately and timely initiated.
Indonesia was the fifth-worst for preterm births in the world with expected 154 preterm births for every 1000 live births.Citation40,Citation41 The event of prematurity, which for the most part, results in LBW, has increased by 3% between 1990 and 2013 and is the second most basic reason for death among neonates and children under five.Citation4,Citation8,Citation42 While maternal, neonatal, and child well-being projects have been started and executed to lessen the death ratio in Indonesia, mainly because of prematurity and LBW,Citation2,Citation43,Citation44 less consideration is given on gathering antenatal data and promoting surveillance tools for fetal development during pregnancy.
As far as we know, the scientific and technical training has, for the first time, prepared Indonesian midwives with the updated scientific knowledge and technology in regards to fetal development evaluation during pregnancy. The training has likewise expanded the midwives’ alertness to the significance of routine documentation of the vital information on attributes of mother and fetus in the present manual pregnancy registers.Citation26 The training also explored several factors triggering incomplete and delayed recording and reporting tasks of ANC data that have been discussed in the previous publication.Citation27 These include a high amount of work, lack of time for routine ANC examinations, inadequate skills and training, and lack of awareness about the importance of documenting the examination results. The result is consistent with those of Burke, Suswardany, Michener, Mazurki, Adair, Elmiyati, RaoCitation38 and Sibiya, Cele, NgxongoCitation45 who found similar factors hampering complete and reliable records of the recommended ANC examination results.
Therefore, the training has the following implications: it can fundamentally improve the quality of information gathered to develop Indonesia’s particular charts and protocols for the surveillance of fetal growth, it gives an understanding into how to execute such agreements to improve patients’ well-being and will prompt better and progressively reliable provision of local antenatal information to carry out evidence-based maternal and fetal hazard appraisal, pregnancy result review, as well as resource planning and allocation. The training focuses on the quality improvement of healthcare services.Citation46 Consistent education and training might then be an important part of investment programs in midwives to lessen maternal and neonatal mortality.Citation29,Citation47
Conclusion
Based on the comprehensive reviews of existing regulations on antenatal examinations, statistical analysis carried out in the previous research, the United Nations International Children’s Emergency Fund (UNICEF) recommendationsCitation30 the current project carried out by the National Research Council (USA) and the Indonesian Academy of Sciences (Indonesia),Citation6,Citation7 the following recommendations are proposed as enabling actions to achieve the 2030 SDGs target for the reduction of neonatal mortality:
Promote continuous scientific and technical training among midwives to update their knowledge and skills, particularly those relevant to measuring and documenting the results of antenatal examinations. As well as analyzing the information to assess potential risks and identify necessary interventions/referrals promptly.
Use the outcome of the information analysis to manage and distribute the health resources effectively.
Introduce the protocols of routine collection of antenatal examination results into clinical practice.
Assess/evaluate the adequacy and quality of antenatal information before and after the scientific and technical training to effectively design the structured content of the next training program.
Abbreviations
ANC, antenatal care; MCH, maternal and child health; PHC, primary healthcare center; FH, fundal height; FS, fetal station/descent level; GA, gestational age; LBW, low birth weight; UNICEF, United Nations International Children’s Emergency Fund.
Data Sharing Statement
Data underlying the findings of this study are included in the manuscript.
Ethics and Consent Statement
As part of a doctoral qualification, research approvals were obtained from the Indonesian national, provincial and local governments and two ethics’ clearances were provided by the Lambung Mangkurat University Medical Research Ethics Committee, Indonesia (reference: 018/KEPK-FK UNLAM/EC/III/2016) and the RMIT College Human Ethics Advisory Network (CHEAN), Australia (reference: ASEHAPP 19-16/RM No: 19974). Data about the reasons for the research and a consent form (written in both Bahasa Indonesia and English) for enrollment in the study were given to the chosen midwives and pregnant women (prospective study), who all consented to take part.
Author Contributions
DA, MA, and KM contributed to the conception and design of the study. DA, A, GTM, ASA provided the literature review and information summary on relevant research articles and policies in Indonesia. DA performed data collection, pre-processing data, analysis, and interpretation. DA prepared the manuscript. MA, KM, and A provided data analysis, advice, proofreading, and critical revision of the manuscript. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Acknowledgments
We are thankful to the Australian Agency for International Development (AusAID) for financing DA’s PhD grant in Mathematical Sciences at the School of Science, RMIT University, Melbourne, Australia.
We are also thankful to all medical practitioners and technical groups for their support in collecting the data used in this study, particularly the Head of the Provincial Health Department of South Kalimantan, Dr. Achmad Rudiansjah, Dr. Andy Yussianto, Nani Lidya, Tut Barkinah, Nurtjahaya, and Masjudah. The full list has been given in previous publications. Citation26,Citation27
Disclosure
The authors report no conflicts of interest in this work.
Additional information
Funding
References
- MoH. Mother’s Handbook of Health Services in Primary Health Facilities and Referral (Buku Saku Pelayanan Kesehatan Ibu Di Fasilitas Kesehatan Dasar Dan Rujukan. Republic of Indonesia: Ministry of Health; 2013.
- MoH. Regulation of the Minister of Health of the Republic of Indonesia Number 97 Year 2014 About Health Service Before Pregnant, Pregnancy, Labor, and Easy After Birth, Contraceptive Implementation Services, and Sexual Health Care Services (Peraturan Menteri Kesehatan Republik Indonesia Nomor 97 Tahun 2014 Tentang Pelayanan Kesehatan Masa Sebelum Hamil, Masa Hamil, Persalinan, Dan Masa Sesudah Melahirkan, Penyelenggaraan Pelayanan Kontrasepsi, Serta Pelayanan Kesehatan Seksual. Republic of Indoesia: Jakarta: Ministry of Health; 2014. 1–119.
- WHO. Strategic Plan for Strengthening Health Systems in the WHO Western Pacific Region. Manila: WHO Regional Office for the Western Pacific; 2008.
- Blencowe H, Cousens S, Chou D, et al. Born too soon: the global epidemiology of 15 million preterm births. Reprod Health. 2013;10(1):S2. doi:10.1186/1742-4755-10-S1-S2
- Lawn JE, Blencowe H, Waiswa P, et al. Stillbirths: rates, risk factors, and acceleration towards 2030. Lancet. 2016;387(10018):587–603. doi:10.1016/S0140-6736(15)00837-5
- Council NR. Reducing Maternal and Neonatal Mortality in Indonesia: Saving Lives. Saving the Future: National Academies Press; 2013.
- Hull TH. Reducing Maternal and Neonatal Mortality in Indonesia: Saving Lives, Saving the Future: Taylor & Francis; 2015.
- Achadi E, Jones G. Health Sector Review: Maternal. Neonatal and Child Health: Bappenas; 2014.
- Heazell AE, Siassakos D, Blencowe H, et al. Stillbirths: economic and psychosocial consequences. Lancet. 2016;387(10018):604–616. doi:10.1016/S0140-6736(15)00836-3
- Moxon SG, Ruysen H, Kerber KJ, et al. Count every newborn; a measurement improvement roadmap for coverage data. BMC Pregnancy Childbirth. 2015;15(2):S8. doi:10.1186/1471-2393-15-S2-S8
- de Bernis L, Kinney MV, Stones W, et al. Stillbirths: ending preventable deaths by 2030. Lancet. 2016;387(10019):703–716. doi:10.1016/S0140-6736(15)00954-X
- MoH. Basic Health Research (Riset Kesehatan Dasar). Republic of Indonesia: Jakarta: Balitbangkes, Ministry of Health; 2013.
- Abdullah A, Elizabeth E, Wungouw U, et al. Knowledge and Skills of Midwife Supervisors and Midwives in NTT and Changes Post Training. Jakarta: Australia Indonesia Partnership of Maternal and Neonatal Health (AIPMNH); 2015.
- Kerber KJ, Mathai M, Lewis G, et al. Counting every stillbirth and neonatal death through mortality audit to improve quality of care for every pregnant woman and her baby. BMC Pregnancy Childbirth. 2015;15(2):S9. doi:10.1186/1471-2393-15-S2-S9
- Anggraini D, Abdollahian M, Marion K. Foetal weight prediction models at a given gestational age in the absence of ultrasound facilities: application in Indonesia. BMC Pregnancy Childbirth. 2018;18(1):436. doi:10.1186/s12884-018-2047-z
- White LJ, Lee SJ, Stepniewska K, et al. Estimation of gestational age from fundal height: a solution for resource-poor settings. J Royal Soc Interface. 2012;9(68):503–510. doi:10.1098/rsif.2011.0376
- Parvin Z, Shafiuddin S, Uddin MA, Begum F. Symphysio fundal height (SFH) measurement as a predictor of birth weight. Faridpur Med College Jl. 2013;7(2):54–58. doi:10.3329/fmcj.v7i2.13498
- Siswosudarmo R, Titisari I. Developing a new formula for estimating birth weight at term pregnancy. Jurnal Kesehatan Reproduksi. 2014;1:2. doi:10.22146/jkr.5350
- Titisari HI, Siswosudarmo R. Risanto’s formulas is more accurate in determining estimated fetal weight based on maternal fundal height. Ind j Obstet Gynecol. 2013;1(3):149–151.
- Papageorghiou AT, Ohuma EO, Gravett MG, et al. International standards for symphysis-fundal height based on serial measurements from the Fetal Growth Longitudinal Study of the INTERGROWTH-21st Project: prospective cohort study in eight countries. BMJ. 2016;355:i5662. doi:10.1136/bmj.i5662
- Robert P, Ho J, Valliapan J, Sivasangari S. Symphysial fundal height (SFH) measurement in pregnancy for detecting abnormal fetal growth. Cochrane Database Systematic Review. 2015;9.
- Morse K, Williams A, Gardosi J. Fetal growth screening by fundal height measurement. Best Practice Res Clin Obstet Gynaecol. 2009;23(6):809–818. doi:10.1016/j.bpobgyn.2009.09.004
- Alisyahbana A, Chaerulfatah A, Usman A, Sutresnawati S. Anthropometry of newborns infants born in 14 teaching centers in Indonesia. Paediatr Indones. 1994;34(62):123.
- Goto E. Meta-analysis: identification of low birthweight by other anthropometric measurements at birth in developing countries. J Epidemiol. 2011;21(5):354. doi:10.2188/jea.JE20100182
- Ten Hoope-Bender P, de Bernis L, Campbell J, et al. Improvement of maternal and newborn health through midwifery. Lancet. 2014;384(9949):1226–1235. doi:10.1016/S0140-6736(14)60930-2
- Anggraini D, Abdollahian M, Marion K, et al. The impact of scientific and technical training on improving routine collection of antenatal care data for maternal and foetal risk assessment: a case study in the Province of South Kalimantan, Indonesia. J Pregnancy. 2018;2018.
- Anggraini D, Abdollahian M, Marion K, et al. The impact of scientific and technical training on improving databases’ adequacy for fetal growth chart development in limited-resource settings: a case study in the province of South Kalimantan, Indonesia. J Pregnancy. 2019;2019:10. doi:10.1155/2019/8540637
- MoH. Indonesia Health Profile (Profil Kesehatan Indonesia) 2015 Republic of Indonesia: Jakarta: Ministry of Health; 2016.
- Achadi E, Jones G. Health Sector Review: Maternal, Neonatal and Child Health. Republic of Indonesia: Jakarta: Ministry of National Development Planning/ Bappenas; 2014.
- UNICEF-Indonesia. Issues Briefs:. UNICEF Indonesia: Maternal and Child Health; 2012.
- MoH. Indonesia Health Profile (Profil Kesehatan Indonesia) 2012. Republic of Indonesia: Jakarta: Ministry of Health; 2013.
- Putera I, Pakasi TA, Karyadi E. Knowledge and perception of tuberculosis and the risk to become treatment default among newly diagnosed pulmonary tuberculosis patients treated in primary health care, East Nusa Tenggara: a retrospective study. BMC Research Notes. 2015;8(1):238. doi:10.1186/s13104-015-1209-6
- Achadi E, Jones G. Health Sector Review: Maternal, Neonatal, and Child Health. Republic of Indonesia: Jakarta: Ministry of National Development Planning/ Bappenas; 2014.
- MoH. Antenatal Integrated Guidelines (Pedoman Pelayanan Antenatal Terpadu). Jakarta: Directorate General of Public Health Directorate of Maternal Health. Republic of Indonesia: Ministry of Health; 2010.
- MoH. Antenatal Integrated Guidelines (Pedoman Pelayanan Antenatal Terpadu). Jakarta: Directorate General of Nutrition and Maternal and Child Health. Republic of Indonesia: Ministry of Health; 2012.
- Enomoto K, Aoki S, Toma R, Fujiwara K, Sakamaki K, Hirahara F. Pregnancy outcomes based on pre-pregnancy body mass index in Japanese women. PLoS One. 2016;11(6):e0157081. doi:10.1371/journal.pone.0157081
- Johnson RW. Calculations in estimating fetal weight. Am J Obstet Gynecol. 1957;74(4):929. doi:10.1016/0002-9378(57)90070-4
- Burke L, Suswardany DL, Michener K, et al. Utility of local health registers in measuring perinatal mortality: A case study in rural Indonesia. BMC Pregnancy Childbirth. 2011;11(1):20. doi:10.1186/1471-2393-11-20
- Rumbold AR, Bailie RS, Si D, et al. Delivery of maternal health care in Indigenous primary care services: baseline data for an ongoing quality improvement initiative. BMC Pregnancy Childbirth. 2011;11(1):16. doi:10.1186/1471-2393-11-16
- Lee AC, Katz J, Blencowe H, et al. National and regional estimates of term and preterm babies born small for gestational age in 138 low-income and middle-income countries in 2010. Lancet Global Heal. 2013;1(1):e26–e36. doi:10.1016/S2214-109X(13)70006-8
- Blencowe H, Cousens S, Oestergaard MZ, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet. 2012;379(9832):2162–2172. doi:10.1016/S0140-6736(12)60820-4
- WHO. World Health Statistics. 2015.
- MoH. Module (Book of Reference) Management of Low Birth Weight Infants (BBLR) for Village Midwives (Modul (Buku Acuan) Manajemen Bayi Berat Lahir Rendah (BBLR) Untuk Bidan Di Desa). Jakarta: Directorate of Nutrition and Maternal and Child Health (Direktorat Jenderal Bina Gizi dan Kesehatan Ibu dan Anak); 2011.
- MoH. Health Services Essential Neonatal: Basic Health Service Technical Guidelines (Pelayanan Kesehatan Neonatal Esensial: Pedoman Teknis Pelayanan Kesehatan Dasar). Jakarta: Directorate General of Health Development Society (Direktorat Jenderal Bina Kesehatan Masyarakat); 2010.
- Sibiya MN, Cele RJ, Ngxongo TSP. Assessment of the use of the new maternity case record in improving the quality of ante natal care in eThekwini District, KwaZulu-Natal. Int J Africa Nurs Sci. 2015;2:53–58. doi:10.1016/j.ijans.2015.04.002
- Gardosi J. Fetal growth standards: individual and global perspectives. Lancet. 2011;377(9780):1812–1814. doi:10.1016/S0140-6736(11)60507-2
- Van Lerberghe W, Matthews Z, Achadi E, et al. Country experience with strengthening of health systems and deployment of midwives in countries with high maternal mortality. Lancet. 2014;384(9949):1215–1225. doi:10.1016/S0140-6736(14)60919-3
