Abstract
Purpose
Ailanthus altissima is one of the world’s most invasive species with a globally problematic spread. Pollen is dispersed locally and partially airborne. We aimed at investigating if (i) A. altissima pollen can be detected in relevant quantity in the air and if (ii) sensitization to A. altissima can be detected in patients with seasonal exacerbation of atopic diseases.
Patients and Methods
We recorded distribution of A. altissima in Leipzig, Germany. In 2019 and 2020, pollen was collected with a Hirst-type pollen trap placed on the roof of the University Hospital. Specific IgE investigations were performed in children and adults with history of atopic diseases with deterioration between May and July. We analysed specific IgE for A. altissima, Alternaria sp., birch, grasses, profilins, polcalcins and crossreacting carbohydrates.
Results
We found abundant growth of A. altissima and pollen was detected from early June to mid-July with a maximum pollen concentration of 31 pollen/m3. Out of 138 patients (63 female, 69 children/adolescents), 95 (69%) had seasonal allergic rhinitis, 84 (61%) asthma, and 43 (31%) atopic dermatitis. Sensitization to A. altissima was shown in 59 (42%). There were no significant differences between age groups. In 59% of patients sensitized (35/59), there was no sensitization to possibly cross-reacting structures.
Conclusion
Sensitization to A. altissima pollen could be detected in 42% of our patients with atopic diseases, suggesting allergenic potential of this neophyte. In the context of further spread with climate change, eradication strategies and population-based sensitization studies are needed.
Introduction
Climate change and changing environmental conditions are altering habitats, mainly in urban areas, leading to the spread of invasive alien plant species. In parallel, atopic diseases have increased tremendously in the last decades and have become a global challenge imposing significant socioeconomic burden.Citation1–8 Indeed, allergy has been suggested as a sentinel measure of planetary health and biodiversity loss.Citation9,Citation10 While multiple factors of climate change lead to higher allergenicity of pollen, invasive alien plant species may result in new sensitizations and more severe clinical signs and symptoms.Citation11,Citation12
A globally spreading and in many countries highly invasive species of concern is A. altissima,Citation13,Citation14 an undemanding deciduous tree of the Simaroubaceae family, native to northeastern and central China, benefiting from the mild urban climate. The name is thought to derive from the Moluccan term “Aylanto”, meaning “tree of heaven”, referring to its rapid growth and height, which can reach 15 m in 25 years.Citation15 The spread of the tree is mainly limited to urban heat islands, as the seedlings are sensitive to low temperatures.Citation15,Citation16 The flowering period of A. altissima is predominantly in June and lasts around 27 days after the gradual opening of the male flowers.Citation15,Citation17
Its success in Europe since its first introduction as an ornamental tree in 1751 is likely due to several factors.Citation13,Citation16,Citation18 A. altissima contains allelopathic chemicals such as ailanthone, which suppresses the growth of other plant species through phytotoxic effects.Citation15,Citation19 Once the tree is established, it spreads aggressively vegetatively and by seeds; its pollen is mainly dispersed locally but also airborne. Control of the tree requires an extremely high level of effort, as it resprouts vigorously after pruning. It also grows in nutrient-poor soils and is resistant to drought, salt, and herbicides, which are major reasons why it is planted as an urban ornamental tree.Citation15,Citation16
In China, where the tree is native, 30% of patients tested for suspected allergic rhinitis had positive skin prick tests for A. altissima.Citation20 In Europe, there is one investigation among 54 allergic adult patients in Italy, as well as in some case reports dating back as far as 80 years.Citation21–26 However, there is a lack of comprehensive studies on sensitization to A. altissima in regions where the tree is spreading invasively. High exposure is particularly expected in cities, where population density is high and warmer temperatures provide optimal growing conditions for the tree. In our study, we aimed at investigating if (i) A. altissima pollen can be detected in relevant quantity and if (ii) sensitization to A. altissima can be detected in patients with seasonal signs and symptoms of their atopic diseases during its pollen season in Leipzig, Germany.
Materials and Methods
Distribution of Ailanthus altissima and Pollen Concentration
The tree registry of the City of Leipzig is publicly accessible online (https://www.leipzig.de/umwelt-und-verkehr/umwelt-und-naturschutz/baeume-und-baumschutz/stadtbaeume/) and was used to determine planting dates and location of planted trees of A. altissima in Leipzig (Supplemental Figure 1). We inspected fifteen 600×600 m grids along an east-west transect including city center and suburbs of approx. 10 km distance where the number of A. altissima plants had already been counted in 1982Citation27 and documented the current number of trees. When making counts, it is important to note that A. altissima trees form root runners. This means that around a big tree many small sprouts can emerge from the roots. As in 1982,Citation27 these shoots are counted as one plant with the tree from which they originate from. Also, groups of young trees in which the individual stems are located within a few square meters, are counted as one tree due to root fruiting. A seven-day recording volumetric Hirst-type pollen and spore trap (Burkhard Manufacturing Ltd., Hertfordshire, UK) (Supplemental Figure 2A) was placed at the roof of the University hospital in Leipzig (51.330847N 12.387845E, height 17.3 m asl) starting in April 2019. The power of the pump was set to a volumetric flow rate of approximately 10 L/min and the drum was replaced once a week. An analysis of the daily mean pollen concentration including all identifiable pollen types was achieved by a defined movement of the vaseline-covered strip on the drum. Based on a defined volumetric flow rate, pollen counts can be related to m3 of air per day. The analysis followed the European regulation DIN EN 16868 for pollen and fungal spore sampling.
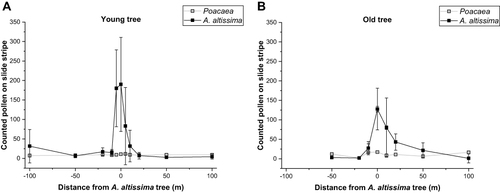
In addition, vaseline-covered slides (Supplemental Figure 2B and 2C) were exposed to individual trees at specific intervals along a transect for one week, and exposure was repeated for one and two additional weeks, respectively. The adhering A. altissima and grass pollen were analyzed with 200x magnification on an area of 2.6 mm x 60 mm in the middle of the cover glass (Counted pollen on slide stripe) according to recommendations of R. Wachter (http://www.dr-wachter.de/Messstellen/Messmethode%20f.htm). For an old tree (location: 51.35233482599263, 12.429702588848789, Breast height diameter (BHD) 1.02m, count data from two consecutive weeks (16.-30.06.2020) were averaged and measured in −50, −20, −10, 0, 10, 20, 50, 100 m distance to the tree, while for a younger tree (location: 51.328712546333406, 12.333695635475502, BHD 0.13m), three consecutive weeks (16.06.-07.07.2020) were used as the mean and measured in −100, −50, −20, −10, 0, 10, 20, 50, 100 m distance to the tree.
Study Population and IgE Measurement
In 2019 and 2020, pediatric and adult residents from Leipzig and neighboring communities who presented to our Comprehensive Allergy Center were enrolled if they had seasonal (May-July) allergic rhinitis (AR) and/or asthma and/or worsening of their atopic dermatitis. We recorded history of atopic diseases as well as known IgE sensitizations. The study was approved by the Medical Ethics Committee of the Medical Faculty of the University of Leipzig (reference number 082–10-19042, 10). All patients gave informed consent. The patients’ data were processed in compliance with the Hospital Law of the Federal State of Saxony (§ 34 Abs.1 SächsKHG). Blood samples were collected and sera were stored at −20℃ for a maximum of 3 months until further use. Specific IgE measurements for A. altissima (research allergen SAS 535 tree of heaven, same batch for all), Betula verrucosa 2/4 (Bet v 2/4, t221), and cross-reactive carbohydrate determinants (CCD; o214) were performed using ImmunoCAP® (Thermo Fisher, Freiburg, Germany). Values >/= 0.35kU/l (CAP1) were classified as positive. Intra- and interday coefficient of correlation was determined for research allergen SAS 535. Proportions of sensitization were plotted against place of residence (city center/suburbs).
Results
Ailanthus altissima in Leipzig
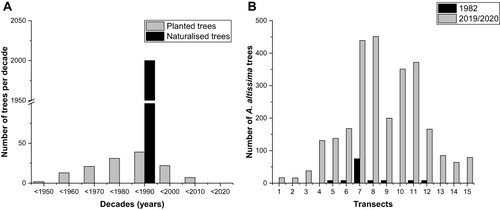
In Leipzig, Germany, there are significant populations of A. altissima and evidence of plantings dates back to 1817.Citation27,Citation28 In the tree cadastre of the city of Leipzig, targeted plantings have been documented since 1920 (Figure S1). Thus, A. altissima has been present in Leipzig for at least 100 years. From 1950 to 1990, new trees were increasingly planted in the city center ( and ). While 67 planted trees were counted until 1980, a detailed vegetation mapping in 1983 showed about 2000 feral trees, resulting in A. altissima being called the “character tree” of the city center.Citation27 An inventory of a subset of records revealed another massive increase from 123 trees in one transect in 1982 to 2715 trees in 2019/2020, representing a 22-fold increase in 37 years (). Abundant numbers of plants were found mainly within the city center.
Figure 1 Ailanthus altissima in front of the Institute of Laboratory Medicine of the University Hospital Leipzig, young feral tree in the city center, and details of the leaves and flower of the tree.

Figure 2 (A) Number of Ailanthus altissima trees planted in Leipzig per decade (gray) and feral trees identified in a detailed mapping by Gutte et al (1987).Citation27 (B) Numbers of A. altissima plants in 1982Citation27 and in 2019/2020, counted along an east-west transect with grid sizes of 600×600 m in Leipzig. Sites 6–10 were located at the city center.
Pollen Concentrations
In 2019 and 2020, A. altissima pollen was recorded from early June to mid-July with a maximum pollen concentration of 31 pollen/m3 on June 15th, 2019 and 21 pollen/m3 on June 30th, 2020. During the flowering period of A. altissima, the following other bioaerosols were enumerated in similar or higher quantities: fungal spores (Alternaria and Epicoccum), as well as pollen of pine (Pinus), plantains (Plantago), grasses (Poaceae), dock (Rumex), lime (Tilia), and stinging nettle (Urtica). Potentially allergenic Poaceae pollen could be detected from mid-May to the end of July and co-occurred in the sample with the peaks of A. altissima (). The assessment of local pollen concentrations showed that they were highest near the trees (0–20 m) and decreased at a distance of 10–50 m ().
Figure 3 Ailanthus altissima (black line, right y-axis), grasses (Poaceae) (gray line), Alternaria (red dashed line), Urtica (orange dashed line) (all on left y-axis) pollen and spore concentrations (pollen m−3) in 2019 (A) and 2020 (B).
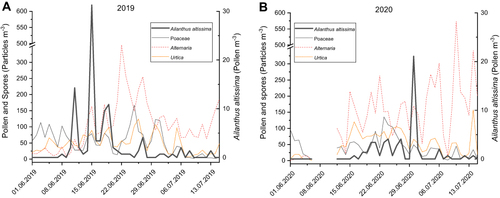
Figure 4 Local pollen counts for grass (Poaceae) and Alternaria altissima pollen near a young (A) and an old (B) solitary tree collected for one week (two/ three consecutive weeks) on a Vaseline-covered object slide (Supplemental Figure 2B and 2C).
Patients and IgE Values
In 59/138 (42%) of patient’s sera we detected specific IgE against A. altissima (). Proportions of sensitization was similar in children and adults (46% and 39%, respectively; n.s.). The mean specific IgE concentration against A. altissima was 4.14 kU (± 8.32 kU/L) with ImmunoCAP classes ranging from 1 to 4. Within the group of the A. altissima-sensitized patients, 59% had neither IgE antibodies against the combination of the profilin Bet v 2 and the polcalcin Bet v 4 nor against the cross-reactive carbohydrate determinants (CCD). The coefficients of variation of the determination of specific IgE against the research allergen SAS 535 tree of heaven were intra-day and inter-day 1.9 and 5.1, respectively. Sensitization to other aeroallergens was very common in both children and adults (). Geomapping showed that individuals from the city and neighboring communities were sensitized to A. altissima at about the same proportion (data not shown).
Table 1 Demographics and Proportions of Sensitization Against Ailanthus altissmia (A. altissima) and Crossreactive Panallergens
Table 2 Concomitant Sensitization to Aeroallergens in Patients with Specific IgE to A. Altissima
Discussion
After several decades of active tree planting, a remarkable population of planted and feral A. altissima trees has developed in Leipzig. As we were able to detect A. altissima pollen in air samples, airborne allergic sensitization is possible. Accordingly, we found sensitization to A. altissima in 42% of pediatric and adult patients with seasonal exacerbations of their atopic diseases.
So far, there are no population-based studies evaluating sensitization to A. altissima. However, allergies to Ailanthus have been described repeatedly and as far back as 80 years ago.Citation23–25 Sensitizations may have been overlooked so far since the allergen is not included in European standards and flowering coincides with other aeroallergens such as grass pollen or Alternaria spores.Citation29,Citation30 It is also possible that sensitization is only detectable in certain local regions with high tree abundance, because – as shown in our investigation - A. altissima pollen are distributed mainly locally.
However, the annual pollen load across the northern hemisphere has increased in recent decades.Citation31 A. altissima has already spread as an invasive neophyte in many parts of the world. Climate change and urban conditions promote further rapid spread and may lead to increased sensitization rates and ultimately enhanced healthcare utilization due to climate change-induced disruption of antigen-specific tolerance.Citation32–37 While 20 years ago the main pollen season for A. altissima was reported in July,Citation15,Citation38 we now detected the highest pollen concentrations in June. This may be a result of very high temperature in early summertime in the years 2019 and 2020. In line, the importance of changing climate conditions for a change in pollination periods along with changes in sensitization rates has been previously documented.Citation39 Simultaneously with the Ailanthus pollen count, we also detected other pollen and spores so that inhalative symptoms at this time might falsely be attributed to these allergens and a relevant sensitization to A. altissima might be missed.
The allergen content of birch pollen was reported to be higher at sites with air pollution.Citation40 As A. altissima mainly spreads in urban areas, any impact of traffic pollution on its allergenicity should be an issue in further investigations. In our study, individuals from the city and neighboring communities were sensitized to A. altissima at about the same proportion. This can be explained by similar exposure due to visits to the workplace, school, daycare center, or city center, as well as a high degree of tree distribution throughout the city.
While the proportion of sensitization among the patients in our study is comparable to that in northeastern Iran, it is much higher than proportions previously reported from European and Chinese patient cohort without considering possible cross-sensitization.Citation20–22,Citation41 At the same time, there is evidence that A. altissima has been present in the landscape of Leipzig city center for at least a hundred years. Sensitization may have occurred during this period. In the trajectories of atopic diseases, allergic sensitization can develop in early childhood and increases with age.Citation42 Therefore, due to the high, increasing and long-lasting population of A. altissima in Leipzig, the high proportion of sensitization appears reasonable. This also corresponds to the assessment that the tree has a high allergenic potential.Citation43 Sensitization in the aforementioned smaller cohorts from Italy was reported almost 20 years ago and may have increased in the meantime. Prediction models show a further spread of A. altissima in the future along with an increasing risk of allergic sensitization worldwide.Citation14 Our pilot study in Leipzig could therefore be a blueprint for many cities and regions worldwide with increasing risk of allergic sensitization worldwide.
While in our study the proportion of sensitization was approximately the same in the different age groups, Werchan et al reported higher proportions in patients younger than 20 years of age.Citation20 The exposure in Leipzig has obviously existed for many decades, explaining sensitizations in all age groups. In China, industrialization and habitat changes have taken a different course in time, which could explain the age dependency.Citation44 Differences may also result from the limited agreement between prick tests in the Chinese cohort and the IgE measurement used in our investigation.Citation45
We included patients with worsening of their atopic diseases during the flowering period of A. altissima. Therefore, the A. altissima pollen may suspected to be a triggering allergen. Of course, other factors, including temperature, and other allergens may as well have contributed to the clinical symptoms. In particular, this includes grass and Urtica pollen or Alternaria spores, which appeared at the same time and were also detected in our pollen trap.Citation11,Citation46
We measured specific IgE directed against A. altissima using the established ImmunoCAP system with a research allergen that has been extracted from pollen in the manufacturer’s standard procedure. Variation coefficients indicate high precision.Citation47,Citation48 In half of the group, mean specific IgE levels against A. altissima reached levels above CAP class 1. Some of the patients have a rather low specific IgE value. These subjects may still be at the beginning of sensitization, as pollen is spread more locally. It is also possible that allergenic structures are underrepresented in the assay. However, since all our patients had seasonal exacerbations of their atopic diseases, IgE sensitization to A. altissima pollen could be clinically relevant.Citation49
In the search for allergenic structures in A. altissima pollen, a number of proteins of 20–55 kDa and <10 kDa were found and identified as enolases, calreticulin, pectate lyases, conserved hypothetical protein, and ras-related protein RHN1-like.Citation23,Citation50–52 It has been shown that A. altissima pollen contains cross-reactive calcium binding proteins and cross-reactive CCDs,Citation23,Citation53,Citation54 but 59% of our patients had no sensitization to CCDs, profilins, or polcalcins which suggests primary sensitization to specific allergens of A. altissima.
Limitations of our clinical study are the ongoing allergen identification of both the pollen itself and the CAP allergen used, and the non-inclusion of other possible panallergens, ie cyclophilin. Therefore, we cannot exclude a certain overestimation of the proportion of sensitization. No commercial and approved diagnostic allergen was available for skin prick tests. Therefore, sensitization could not be confirmed by this in vivo method.
Conclusions
A. altissima plantings in the city of Leipzig led to an expansive spread of this invasive species. The pollen could be detected both locally and remotely from trees in the flowering months of June and July. In a hospital-based, high-risk group of patients with atopic diseases, we found sensitization to A. altissima in 42% with no significant differences between age groups and in the majority without cross-sensitization to panallergens.
Future international population-based cross-sectional and prospective studies including mapping of tree populations are needed to clarify epidemiology of A. altissima pollen allergy. Standardized testing procedures (IgE, skin prick test, provocation tests) need to be developed and validated. Our data suggest that planting of A. altissima should be avoided. Development of control strategies seems mandatory, due to the invasive growth and the allergenic potential of this alien species.
Data Sharing Statement
The pollen data relevant for the study will be available from Dr. Susanne Dunker ([email protected]) on reasonable request.
Ethics Approval and Informed Consent
The study was approved by the Medical Ethics Committee of the Medical Faculty of the University of Leipzig (reference number 082-10-19042010). All patients gave informed consent. For the patients under 18 years of age, parents or legal guardians of provided informed consent. The patients’ data were processed in compliance with the Hospital Law of the Federal State of Saxony (§ 34 Abs.1 SächsKHG).
Consent for Publication
The authors give consent to publish the details of any images, videos, recordings, etc and that the person(s) providing consent have been shown the article contents to be published.
Authors` Contributions
SD, FP, JS, JG, MH and RT designed the study. SD performed the pollen distribution assessments. FP, MH, TL, JZ, MB, VZ, PK and RT were involved in clinical investigations, SK and TK performed IgE diagnostics, TH conducted the plotting of Figure S1 and was involved in managing the pollen traps, MB counted the pollen of the Burkard (Hirst-type) pollen traps. SK provided local knowledge of A. altissima. FP, SD, RT, and JS prepared the draft.
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. Freerk Prenzel and Regina Treudler should be considered joint first author.
Disclosure
Dr Freerk Prenzel reports personal fees from Sanofi Genzyme, personal fees from Novartis, personal fees from Takeda, personal fees from Nutricia Milupa, outside the submitted work. Dr Tobias Lipek reports personal fees from Novartis, personal fees from Aimmune, personal fees from Sanofi Genzyme, outside the submitted work. Prof. Dr. Jon Genuneit reports grants from Danone Nutricia Research, grants from German Federal Ministry of Health, grants from German Federal Ministry of Education and Research, personal fees from Wiley Publishing Company & European Academy of Allergy and Clinical Immunology, outside the submitted work; In addition, Prof. Dr. Jon Genuneit has 2 patents on breast milk composition submitted by Danone Nutricia Research pending . Drs Susanne Dunker reports non-financial support from Luminex, during the conduct of the study; In addition, Drs Susanne Dunker has a patent PCT/EP2017/075553 pending to Susanne Dunker; and the work was partially funded by the German Research Foundation for support via the iDiv Flexpool Funding projects Grant Number: 34600865-16 (“Kick-off-Meeting PolDiv”), Grant Number: 34600830-13, (“PolDiv”-Project) and Grant Number: RA-373/20 (iCyt - Support Unit). The authors report no other conflicts of interest in this work.
Acknowledgments
In addition, we want to thank Paul Remmler for the technical support during the installation and maintenance of the measuring station, as well as Marcus Karsten for pollen sampling in spring 2020, Prof. Dr. Karl-Christian Bergmann, Dr. Matthias & Dr. Barbora Werchan from the German Pollen Information Service (PID) at the Charité Berlin for renting the pollen trap, Vladislav Svyechkin for performing the A. altissima tree inventory during his BELL work under the supervision of Prof. Dr. Ingolf Kühn. Dr. Peter Gutte supported us with a lot of local literature about A. altissima.
Additional information
Funding
References
- Akdis CA. Does the epithelial barrier hypothesis explain the increase in allergy, autoimmunity and other chronic conditions? Nat Rev Immunol. 2021;21(11):739–751. doi:10.1038/s41577-021-00538-7
- Asher MI, Montefort S, Björkstén B, et al. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC phases one and three repeat multicountry cross-sectional surveys. Lancet. 2006;368(9537):733–743. doi:10.1016/S0140-6736(06)69283-0
- Eder W, Ege MJ, Mutius von E. The asthma epidemic. N Engl J Med. 2006;355(21):2226–2235. doi:10.1056/NEJMra054308
- Platts-Mills TAE. The allergy epidemics: 1870–2010. J Allergy Clin Immunol. 2015;136(1):3–13. doi:10.1016/j.jaci.2015.03.048
- Strachan DP, Rutter CE, Asher MI, et al. Worldwide time trends in prevalence of symptoms of rhinoconjunctivitis in children: global asthma network phase I. Pediatr Allergy Immunol. 2022;33(1):e13656. doi:10.1111/pai.13656
- Pawankar R. Allergic diseases and asthma: a global public health concern and a call to action. World Allergy Organ J. 2014;7(1):12. doi:10.1186/1939-4551-7-12
- Damialis A, Traidl-Hoffmann C, Treudler R. Climate change and pollen allergies. In: Marselle MR, Stadler J, Korn H, Irvine KN, Bonn A, editors. Biodiversity and Health in the Face of Climate Change. Cham: Springer International Publishing; 2019:47–66.
- Hornick T, Richter A, Harpole WS, et al. An integrative environmental pollen diversity assessment and its importance for the Sustainable Development Goals. Plants People Planet. 2022;4:110–121. doi:10.1002/ppp3.10234
- Prescott SL. Allergy as a sentinel measure of planetary health and biodiversity loss. Allergy. 2020;75(9):2358–2360. doi:10.1111/all.14255
- Agache I, Sampath V, Aguilera J, et al. Climate change and global health: a call to more research and more action. Allergy. 2022;00:1–19. doi:10.1111/all.15229
- D’Amato G, Chong-Neto HJ, Monge Ortega OP, et al. The effects of climate change on respiratory allergy and asthma induced by pollen and mold allergens. Allergy. 2020;75(9):2219–2228. doi:10.1111/all.14476
- Lake IR, Jones NR, Agnew M, et al. Climate change and future pollen allergy in Europe. Environ Health Perspect. 2017;125(3):385–391. doi:10.1289/EHP173
- Arianoutsou M, Bazos I, Christopoulou A, et al. Alien plants of Europe: introduction pathways, gateways and time trends. Peer J. 2021;9:e11270. doi:10.7717/peerj.11270
- Walker GA, Gaertner M, Robertson MP, Richardson DM. The prognosis for Ailanthus altissima (Simaroubaceae; tree of heaven) as an invasive species in South Africa; insights from its performance elsewhere in the world. S Afr J Bot. 2017;112:283–289. doi:10.1016/j.sajb.2017.06.007
- Kowarik I, Säumel I. Biological flora of Central Europe: Ailanthus altissima (Mill.) Swingle. Perspect Plant Ecol Evol. 2007;8(4):207–237. doi:10.1016/j.ppees.2007.03.002
- Sladonja B, Sušek M, Guillermic J. Review on invasive tree of heaven (Ailanthus altissima (Mill.) Swingle) conflicting values: assessment of its ecosystem services and potential biological threat. Environ Manage. 2015;56(4):1009–1034. doi:10.1007/s00267-015-0546-5
- Davies PA. Floral glands in Ailanthus altissima. Trans Kentucky Acad Sci. 1943;11:12–16.
- Swingle WT. The early European history and the botanical name of the tree of heaven, Ailanthus altissima. J Wash Acad Sci. 1916;6(14):490–498.
- Heisey RM. Allelopathic and herbicidal effects of extracts from tree of heaven (Ailanthus altissima). Am J Bot. 1990;77(5):662–670. doi:10.1002/j.1537-2197.1990.tb14451.x
- Werchan M, Zhi Y, Gu J, Werchan B, Bergmann K-C. Sensitization to the native tree-of-heaven (Ailanthus altissima (Mill.) Swingle) in Beijing, China. ICA 2018-11th Intern. Congress of Aerobiology, Parma; 2018. Available from: https://www.researchgate.net/publication/327558554_Sensitization_to_the_native_Tree-of-Heaven_Ailanthus_altissima_Mill_Swingle_in_Beijing_China. Accessed September 21, 2021.
- Ballero M, Ariu A, Falagiani P. Allergy to Ailanthus altissima (tree of heaven) pollen. Allergy. 2003;58(6):532–533. doi:10.1034/j.1398-9995.2003.00172.x
- Maxia A, Maxia L. Ailanthus altissima (Miller) Swingle as a cause of immunoallergic respiratory manifestations. Rendiconti Seminario Facoltà Scienze Università Cagliari. 2003;73(1):27–32.
- Martí-Garrido J, Corominas M, Castillo-Fernández M, Belmonte J, Pineda F, Lleonart R. Allergy to Ailanthus altissima pollen: a local allergen to consider. J Investig Allergol Clin Immunol. 2020;30(6):452–454. doi:10.18176/jiaci.0577
- Mousavi F, Majd A, Shahali Y, Ghahremaninejad F, Kardar G, Pourpak Z. Pollinosis to tree of heaven (Ailanthus altissima) and detection of allergenic proteins: a case report. Ann Allergy Asthma Immunol. 2016;116(4):374–375. doi:10.1016/j.anai.2016.01.019
- Blumstein GI. Sensitivity to ailanthus pollen. J Allergy. 1943;14(4):329–334. doi:10.1016/S0021-8707(43)90668-9
- Bergmann K-C, Werchan M, Werchan B. Allergy to tree-of-heaven pollen in Germany: detection by positive nasal provocation. Allergo J Int. 2020;29(4):126–128. doi:10.1007/s40629-020-00122-w
- Gutte P, Klotz S, Lahr C, Trefflich A. Ailanthus altissima (Mill.) Swingle—eine vergleichende pflanzengeographische Studie. Folia Geobot Phytotax. 1987;22(3):241–262. doi:10.1007/BF02854625
- Breiter CA. Hortus Breiterianus. Verzeichnis aller derjenigen Gewächse, welche im Breiterschen botanischen Garten zu Leipzig gezogen und unterhalten werden. [List of all the plants that are grown and maintained in Breiter's Botanic Garden in Leipzig]. Leipzig: C. F. Franz; 1817.
- Heinzerling L, Mari A, Bergmann K-C, et al. The skin prick test - European standards. Clin Transl Allergy. 2013;3(1):3. doi:10.1186/2045-7022-3-3
- Matricardi PM, Kleine-Tebbe J, Hoffmann HJ, et al. EAACI molecular allergology user’s guide. Pediatr Allergy Immunol. 2016;27(Suppl 23):1–250. doi:10.1111/pai.12563
- Ziska LH, Makra L, Harry SK, et al. Temperature-related changes in airborne allergenic pollen abundance and seasonality across the northern hemisphere: a retrospective data analysis. Lancet Planet Health. 2019;3(3):e124–e131. doi:10.1016/S2542-5196(19)30015-4
- Wang A, Melton AE, Soltis DE, Soltis PS. Potential distributional shifts in North America of allelopathic invasive plant species under climate change models. Plant Divers. 2022;44(1):11–19. doi:10.1016/j.pld.2021.06.010
- Motti R, Zotti M, Bonanomi G, Cozzolino A, Stinca A, Migliozzi A. Climatic and anthropogenic factors affect Ailanthus altissima invasion in a Mediterranean region. Plant Ecol. 2021;222:1347–1359. doi:10.1007/s11258-021-01183-9
- Walker GA, Robertson MP, Gaertner M, Gallien L, Richardson DM. The potential range of Ailanthus altissima (tree of heaven) in South Africa: the roles of climate, land use and disturbance. Biol Invasions. 2017;19(12):3675–3690. doi:10.1007/s10530-017-1597-8
- Haahtela T, Alenius H, Lehtimäki J, et al. Immunological resilience and biodiversity for prevention of allergic diseases and asthma. Allergy. 2021;76(12):3613–3626. doi:10.1111/all.14895
- Ray C, Ming X. Climate change and human health: a review of allergies, autoimmunity and the microbiome. Int J Environ Res Public Health. 2020;17(13):4814. doi:10.3390/ijerph17134814
- Roblin DW, Jones JW, Fuller CH. Pollen exposure and associated healthcare utilization: a population-based study using health maintenance organization data in the Washington, DC, area. Ann Am Thorac Soc. 2021;18(10):1642–1649. doi:10.1513/AnnalsATS.202008-976OC
- Klotz S, Kühn I, Durka W. BIOFLOR - Eine Datenbank mit biologisch-ökologischen Merkmalen zur Flora von Deutschland. [A database with biological-ecological features on the flora of Germany]. Schriftenreihe für Vegetationskunde. 2002;38:1–334.
- Lee KS, Kim K, Choi Y-J, et al. Increased sensitization rates to tree pollens in allergic children and adolescents and a change in the pollen season in the metropolitan area of Seoul, Korea. Pediatr Allergy Immunol. 2021;32(5):872–879. doi:10.1111/pai.13472
- Ziemianin M, Waga J, Czarnobilska E, Myszkowska D. Changes in qualitative and quantitative traits of birch (Betula pendula) pollen allergenic proteins in relation to the pollution contamination. Environ Sci Pollut Res Int. 2021;28(29):39952–39965. doi:10.1007/s11356-021-13483-8
- Fereidouni M, Farid Hossini R, Jabbari Azad F, Ali Assarezadegan M, Varasteh A. Skin prick test reactivity to common aeroallergens among allergic rhinitis patients in Iran. Allergol Immunopathol. 2009;37(2):73–79. doi:10.1016/S0301-0546(09)71108-5
- Ödling M, Wang G, Andersson N, et al. Characterization of asthma trajectories from infancy to young adulthood. J Allergy Clin Immunol Pract. 2021;9(6):2368–2376.e3. doi:10.1016/j.jaip.2021.02.007
- Cariñanos P, Marinangeli F. An updated proposal of the potential allergenicity of 150 ornamental trees and shrubs in mediterranean cities. Urban for Urban Green. 2021;63:127218. doi:10.1016/j.ufug.2021.127218
- Wang Q, Su M, Li R. Toward to economic growth without emission growth: the role of urbanization and industrialization in China and India. J Clean Prod. 2018;205:499–511. doi:10.1016/j.jclepro.2018.09.034
- Chauveau A, Dalphin M-L, Mauny F, et al. Skin prick tests and specific IgE in 10-year-old children: agreement and association with allergic diseases. Allergy. 2017;72(9):1365–1373. doi:10.1111/all.13148
- Erbas B, Jazayeri M, Lambert KA, et al. Outdoor pollen is a trigger of child and adolescent asthma emergency department presentations: a systematic review and meta-analysis. Allergy. 2018;73(8):1632–1641. doi:10.1111/all.13407
- van Hage M, Hamsten C, Valenta R. ImmunoCAP assays: pros and cons in allergology. J Allergy Clin Immunol. 2017;140(4):974–977. doi:10.1016/j.jaci.2017.05.008
- Gore A, Evans G, Rilvën M. Phadia Laboratory Systems. In: The Immunoassay Handbook. Amsterdam: Elsevier; 2013:617–619.
- Roberts G, Ollert M, Aalberse R, et al. A new framework for the interpretation of IgE sensitization tests. Allergy. 2016;71(11):1540–1551. doi:10.1111/all.12939
- Mousavi F, Shahali Y, Pourpak Z, Majd A, Ghahremaninejad F. Year-to-year variation of the elemental and allergenic contents of Ailanthus altissima pollen grains: an allergomic study. Environ Monit Assess. 2019;191(6):362. doi:10.1007/s10661-019-7458-4
- Mousavi F, Majd A, Shahali Y, Ghahremaninejad F, Shokouhi Shoormasti R, Pourpak Z. Immunoproteomics of tree of heaven (Ailanthus atltissima) pollen allergens. J Proteomics. 2017;154:94–101. doi:10.1016/j.jprot.2016.12.013
- Morales-Amparano MB, Huerta-Ocampo JÁ, Pastor-Palacios G, Teran LM. The role of enolases in allergic disease. J Allergy Clin Immunol Pract. 2021;9(8):3026–3032. doi:10.1016/j.jaip.2021.04.005
- Asam C, Hofer H, Wolf M, Aglas L, Wallner M. Tree pollen allergens-an update from a molecular perspective. Allergy. 2015;70(10):1201–1211. doi:10.1111/all.12696
- Samei A, Fallahpour M, Bolouri MR, et al. Evaluating the Immunoreactivity of Ailanthus Altissima (the tree of heaven) pollen extract in atopic patients. Iran J Allergy Asthma Immunol. 2020;19(2):132–138. doi:10.18502/ijaai.v19i2.2763
