Abstract
Much of human interaction is predicated upon our innate capacity to infer the thoughts, beliefs, emotions, and perspectives of others, in short, to possess a “theory of mind” (ToM). While the term has evolved considerably since its inception, ToM encompasses our unique ability to apprehend the mental states of others, enabling us to anticipate and predict subsequent behavior. From a developmental perspective, ToM has been a topic of keen research interest, with numerous studies seeking to explicate the origins of this fundamental capacity and its disruption in developmental disorders such as autism. The study of ToM at the opposite end of the lifespan, however, is paradoxically new born, emerging as a topic of interest in its own right comparatively recently. Here, we consider the unique insights afforded by studying ToM capacity in neurodegenerative disorders. Arguing from a novel, transdiagnostic perspective, we consider how ToM vulnerability reflects the progressive degradation of neural circuits specialized for an array of higher-order cognitive processes. This mechanistic approach enables us to consider the common and unique neurocognitive mechanisms that underpin ToM dysfunction across neurodegenerative disorders and for the first time examine its relation to behavioral disturbances across social, intimate, legal, and criminal settings. As such, we aim to provide a comprehensive overview of ToM research in neurodegeneration, the resultant challenges for family members, clinicians, and the legal profession, and future directions worthy of exploration.
Introduction
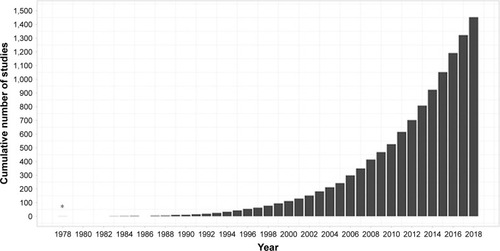
The term “theory of mind” (ToM) was first coined in 1978 to refer to an individual’s capacity to attribute mental states to oneself and others.Citation1 Since then, over 1,450 studies have been published on this topic, exploring the cognitive and neural mechanisms that support this capacity and its disruption in clinical populations (). The definition has since been updated to incorporate a broader range of abilities, including understanding and inferring the thoughts, feelings, beliefs, and intentions of ourselves (ie, first-person ToM) and others (ie, third-person ToM).Citation2 Intuitively, the conceptualization of ToM overlaps with the related construct of empathy, which involves the basic recognition and understanding of another person’s affective state, in addition to the sharing of this emotional experience.Citation3 The precise manner in which these two processes are related, however, remains a matter of debate.Citation3,Citation4 One argument holds that ToM is a “domain-specific” ability, distinct from other cognitive functions,Citation5 providing the foundational mechanism upon which other complex social processes, such as empathy,Citation6 are built. By contrast, the “domain-general” theory proposes that ToM involves the understanding of representations in general, invoking various component cognitive processes, some of which are shared with empathyCitation3 and other social functions.Citation7 Accordingly, affective ToM (ie, feelings and emotions) is separable from its cognitive counterpart (ie, thoughts, beliefs, and intentions).Citation8
Figure 1 The exponential increase in number of published articles on theory of mind since its inception.
As shall be demonstrated, this fractionation between cognitive and affective ToM has proven particularly useful in characterizing the nature of social cognitive dysfunction in neurodegeneration,Citation9 as well as in differentiating between dementia syndromes.Citation10 Nonetheless, parcellating dynamic and multifaceted social interactions into exclusive categories remains challenging,Citation11 as cognitive and affective ToM are themselves multifaceted constructs, which are not always uniformly affected in neurodegeneration.Citation12,Citation13 Moreover, according to the domain-general account, multiple cognitive abilities support ToM capacity, including executive function,Citation14,Citation15 memory,Citation16 language,Citation17 and visuospatial skills,Citation18 a pertinent issue to consider in the context of neurodegenerative disorders characterized by widespread cognitive impairment. Here, we provide an update on ToM research in neurodegeneration, considering how large-scale brain network dysfunction, and resultant cognitive impairment, impacts cognitive and affective expressions of ToM. In doing so, we aim to explicate the common and divergent neurocognitive mechanisms that subtend ToM dysfunction across neurodegenerative disorders and adjudicate between domain-specific vs domain-general accounts of ToM impairments in these syndromes.
Why study ToM in neurodegenerative disorders?
Functional neuroimaging studies in healthy individuals, complemented by lesion evidence from clinical populations, have accelerated our understanding of the neural circuitry supporting ToM performance. In the last decade, however, we have witnessed a shift from understanding the roles of localized brain regions to considering interactions between large-scale neural networks that support complex cognitive endeavors such as ToM (). The study of neurodegenerative disorders offers compelling insights into the neurocognitive architecture of ToM, as these syndromes target large-scale brain networks implicated in ToM and many of its associated processes ().Citation19 Moreover, neurodegenerative disorders frequently present with co-occurring social cognitive, memory, and executive impairments, offering an opportunity to explore the intersection between ToM and cognitive function more broadly. In this review, we focus on the most common neurodegenerative syndromes that present with early impairments in social cognition, as well as emerging evidence of ToM dysfunction in syndromes not traditionally classified as disorders of social cognition.
Figure 2 A distributed brain network supporting ToM reasoning, highlighting the corresponding putative general cognitive function and ToM-specific roles of each region.
Abbreviations: ACC, anterior cingulate cortex; ATL, anterior temporal lobe; fIN, frontoinsular; mPFC, medial prefrontal cortex; PCC, posterior cingulate cortex; TPJ, temporoparietal junction; ToM, theory of mind.
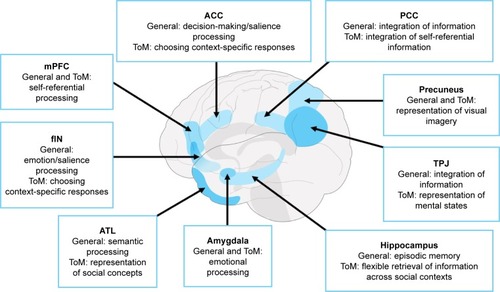
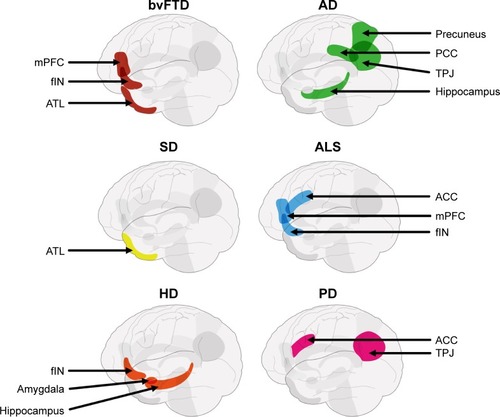
Figure 3 Schematic displaying differential vulnerability of key nodes of the ToM network across neurodegenerative disorders.
Abbreviations: ACC, anterior cingulate cortex; AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; ATL, anterior temporal lobe; bvFTD, behavioral variant of frontotemporal dementia; fIN, frontoinsular; HD, Huntington’s disease; mPFC, medial prefrontal cortex; PCC, posterior cingulate cortex; PD, Parkinson’s disease; SD, semantic dementia; TPJ, temporoparietal junction; ToM, theory of mind.
Behavioral-variant of frontotemporal dementia (bvFTD)
BvFTD represents the prototypical disorder of social cognition. Characterized by insidious changes in personality and behavior, patients with bvFTD display flagrant violation of social norms, lack of social comportment, and apparent loss of empathy for others.Citation20 The considerable parallels in social cognitive difficulties between bvFTD and developmental disorders, such as autism, provided an early clue that ToM might be altered in this dementia syndrome (John R Hodges, personal communication). From a neuroanatomical perspective, this observation prior to the advent of modern day neuroimaging techniques was particularly astute, given the now well-established vulnerability of large-scale networks specialized for socioemotional processing in bvFTD.Citation21
The emergence of prominent ToM impairments as manifested in socially disruptive and inappropriate behaviors typically heralds the onset of bvFTD.Citation22 Family members frequently report the affected individual to lack warmth, have an apparent disregard for others, and to display increasingly rigid and egocentric behavior. Collectively, these disturbances are posited to reflect a core, syndrome-specific difficulty in ToM,Citation23–Citation30 which in turn profoundly increases carer burden.Citation31 The observation of significant cognitive and affective ToM disruption irrespective of modality of testing (ie, verbal or nonverbal) points to a primary ToM impairment in bvFTD. The evidence to date corroborates this position, with bvFTD patients presenting marked deficits across the broad spectrum of ToM tasks including first-order (ie, understanding others’ false beliefs about the world)Citation32 and second-order false belief (ie, understanding others’ false beliefs about a third party’s mental state),Citation22,Citation33–Citation35 detection of social faux pas,Citation30,Citation31 intention and emotion attribution on short video vignettes,Citation36 and decoding emotion from visual cues (Reading the Mind in the Eyes Task; RMET).Citation22
Mechanisms of ToM disruption in bvFTD
Arriving at a precise understanding of the cognitive origins of ToM disruption in bvFTD has proven challenging, in part due to the widespread nature of cognitive impairment in this syndrome. As such, ToM impairments may arise simply as a consequence of global cognitive and executive dysfunction, rather than a primary socioemotional impairment per se. By this view, bvFTD patients would be predicted to perform poorly on ToM tasks that demand greater executive resources. Paradoxically, however, bvFTD patients demonstrate deficits on even simple first-order false belief tasksCitation32 as well as nonverbal ToM tasks with limited cognitive and executive loading,Citation16 suggesting a fundamental, domain-specific impairment. Crucially, ToM impairments in bvFTD emerge statistically independent of overall cognitive and executive status when data-driven clustering and prediction models are employed,Citation14,Citation37 suggesting that social cognitive deficits are dissociable from general cognitive decline in this syndrome. The observation of significant difficulties on ToM, but not on related control tasks,Citation38 reinforces the presence of a core ToM impairment, independent of co-occurring executive, memory, language, and other general cognitive deficits in this syndrome.Citation14,Citation25,Citation37
Could ToM impairments arise simply as a function of widespread socioemotional dysfunction characteristic of the bvFTD syndrome? A recent meta-analysis suggests that ToM and emotion processing impairments are present to a commensurate degree.Citation25 It is possible that the deterioration of affective ToM abilities co-occurs with a general decline in emotion processing in bvFTD,Citation27,Citation39 reflecting the shared neural structures implicated in these abilities. Importantly, however, the presence of early deficits on cognitive, as well as affective, ToM tasks in bvFTD suggests that emotion processing is unlikely to entirely account for disrupted ToM in this syndrome. Other mechanisms, such as an inability to integrate social and contextual cues, likely also contribute to ToM dysfunction in bvFTD.Citation40 Furthermore, it remains unclear whether the ToM impairment in bvFTD reflects a general incapacity to inhibit one’s own mental perspectiveCitation35 or adopt any perspective beyond the “here and now”,Citation41,Citation42 resonating with recent reports of egocentric, rigid behavior, and environmental dependency in this syndrome.Citation43,Citation44 As such, while ToM impairments in bvFTD appear to manifest independent of memory and executive functions, the precise contribution of socioemotional processes remains to be fully elucidated.
Neuroanatomy of ToM impairments in bvFTD
A common assumption is that ToM dysfunction in bvFTD reflects early atrophy to the medial prefrontal cortex (mPFC), one of the core regions of the ToM brain network ().Citation23,Citation45 Indeed, the magnitude of visually rated mPFC atrophy in bvFTD patients directly correlates with objective impairments in false belief and faux-pas judgments.Citation32 Reduced gray matter intensity of mPFC and neighboring prefrontal regions has been linked to performance impairments on the ToM component of the Frith–Happé animation task.Citation16 Similarly, a reduction in coherent, resting state mPFC activity has been found to correlate with poorer performance on emotion attribution tasks employing short story vignettes.Citation46 Finally, modulative neurostimulation via transcranial direct current stimulation over the mPFC has been demonstrated to improve accuracy on cognitive ToM (measured using intention attribution) tasks in bvFTD,Citation36 suggesting a causal role for this region in ToM performance.
Neurodegenerative disorders, however, rarely target discrete regions in isolation,Citation47 and progressive neural degradation in bvFTD reveals the importance of regions beyond the mPFC in modulating ToM performance. Emerging evidence suggests that in parallel with early atrophy of the mPFC, anterior cingulate cortices/frontoinsular (ACC/fIN) regions are particularly vulnerable to functional disruption and volumetric reduction in this syndrome.Citation48 The importance of ACC/fIN regions in the context of ToM impairments in bvFTD deserves particular attention (). A prominent hypothesis contends that accumulation of pathology around the ACC/fIN regions in bvFTD directly targets a specific population of neurons called von Economo neurons (VENs). VENs are an evolutionarily specialized set of cortical neurons suggested to play a critical role in ToM capacity, possibly through supporting the selection of quick and intuitive responses during uncertain social situations.Citation49 Furthermore, the ACC/fIN cortices, along with the mPFC, are implicated in regulating and selecting context-specific emotional responses,Citation50 and their degradation, in part, may contribute to co-occurring emotion dysregulation and ToM impairments in bvFTD. Finally, the ACC/fIN regions anchor the Salience Network of the brain, proposed to tag incoming salient information for further processing, thereby guiding appropriate behavioral and social responses.Citation51 Degeneration of ACC/fIN regions reduces VEN counts,Citation52 disrupts Salience Network functioning,Citation53 and is directly implicated in ToM dysfunction in bvFTD.Citation16 Atrophy of the ACC/fIN cortices in bvFTD is associated with compromised cognitive ToM on the Frith–Happé animationsCitation16 and affective ToM difficulties on emotion attribution tasks.Citation54 Furthermore, lower emotion attribution scores on affective ToM tasks in bvFTD parametrically relate to reduced connectivity between ACC/fIN, PFC, and temporal cortices, as demonstrated using resting-state functional connectivity,Citation46 suggesting that alterations in ACC/fIN circuitry disrupt ToM performance in bvFTD.
Looking beyond the prefrontal cortex
We next consider how damage to regions beyond the pre-frontal cortex gives rise to ToM dysfunction in bvFTD. With disease progression, atrophy encroaches into the anterior and medial temporal lobes most noticeably on the right-hand side,Citation55 including the hippocampus and amyg-dala,Citation56 parietal cortices including temporoparietal junction (TPJ), precuneus,Citation57 and posterior cingulate cortices (PCC)Citation58 as well as subcortical regions including the thalamus and caudate nuclei.Citation58 Importantly, atrophy to regions beyond the prefrontal cortex is associated with ToM dysfunction in bvFTD. For example, impairments on emotion attribution and faux-pas tasks in bvFTD have been shown to correlate with lateral temporal cortex integrity, including the right anterior temporal lobes (ATLs)Citation59,Citation60 and superior temporal cortices,Citation31,Citation61 as well as medial and lateral parietal regions such as the TPJ, precuneus,Citation54 and PCC.Citation61 Emergent evidence has further implicated cerebellar degeneration in modulating aspects of cognitive dysfunctionCitation62 as well as ToM difficulties on the Frith–Happé animations task.Citation16 Longitudinal studies tracking how the evolution of ToM dysfunction in bvFTD relates to encroachment of pathology in temporoparietal cortices and the cerebellum are clearly warranted.
Alzheimer’s disease (AD)
At first glance, it seems somewhat counterintuitive to discuss AD in the context of ToM impairment, given its conception primarily as an amnestic disorder. Clinically, AD patients are noted to retain their warmth, affability, and social graces,Citation63–Citation65 with social cognitive impairments typically manifesting in later stages of the disease course.Citation66 As a result, AD patients were historically included in ToM studies as disease control groups rather than syndromes of interest. With mounting evidence of episodic memory impairments in bvFTD,Citation67,Citation68 ToM performance was further suggested to hold high diagnostic utility in the discrimination of bvFTD from AD.Citation69 The emerging picture, however, is less clear, with mixed reports of moderate impairments on social cognitive tasks in AD, modulated largely by the cognitive demands of the task itself.
Mechanisms of ToM disruption in AD
Two competing hypotheses have been advanced in relation to ToM disturbances in AD. One position holds that AD patients display a fundamental, domain-specific ToM impairment, in light of evidence for a degraded capacity to accurately infer and attribute beliefs, intentionality, and emotional states to characters on some studies of cognitive (eg, false belief tasksCitation70,Citation71) and affective (eg, RMETCitation70) ToM. Other findings reveal significant impairments in AD in inferring mental exchanges of interlocutors from simple conversational exchanges in vignettesCitation72 as well as in real life.Citation73 The observation that impairment in attributing physical causality to characters in first-order false belief tasks is independent of comprehension abilities and executive performanceCitation70 has been interpreted as reflecting fundamental cognitive and affective ToM impairments in AD even on tasks with relatively low cognitive demand.
On the other hand, a more prominent view holds that ToM impairments in AD reflect a global decline in cognitive processes including episodic and working memory and executive function.Citation24,Citation74 In support of this position, general cognitive, executive, and memory performances in AD have been shown to predict subsequent ToM capacity.Citation14,Citation74 For example, Ramanan et alCitation14 found that between 50% and 70% of cognitive ToM performance in AD (as measured by the faux-pas test) could be explained by patients’ overall attention and executive performance. Similarly, Synn et alCitation16 found significant associations between ToM performance on the Frith–Happé animations and episodic memory retrieval impairments in AD. Moreover, AD performance on ToM tasks with low cognitive demands, such as first-order false belief, is relatively spared in the majority of studies,Citation32,Citation34,Citation75,Citation76 whereas its more cognitively demanding counterpart (second-order false belief) is impaired.Citation72 Interestingly, AD patients are impaired even on control items of second-order false belief tasksCitation75 that rely on working memory and comprehension abilities, further suggesting that ToM deficits in the syndrome arise due to the cognitive complexity of the tasks.
Although the aforementioned studies suggest that a decline in executive and memory functions influences ToM performance in AD, the precise nature of their contributions remains unclear. These processes may support the integration and maintenance of relevant information on ToM tasks, as well as facilitating the inference of mental states in the presence or absence of situational cues.Citation77,Citation78 With disease progression, increasing executive, visuospatial, and general cognitive dysfunction likely impact ToM performance, especially on tasks that tax visuospatial abilities or lack explicit situational cues.Citation16,Citation79 In this regard, the study by Synn et alCitation16 is notable in demonstrating comparable difficulties on the ToM and “random movement” conditions of the Frith–Happé animations task, suggesting that ambiguity of stimuli and increasingly abstract task demands contribute to ToM impairments in AD.Citation16 By contrast, with the provision of situational and social contexts, ToM deficits in AD are mitigated,Citation79 presumably due to attenuation of the executive and mnemonic task demands. In summary, the bulk of evidence points to a domain-general impairment in ToM, attributable to a primary decline in memory, executive, and general cognitive abilities.
Neuroanatomy of ToM impairments in AD
Efforts to clarify the neuroanatomical signature of ToM disruption in AD have been limited as social cognitive impairments emerge relatively late in the disease course.Citation66 Moreover, the diffuse gray and white matter neural damage at these later stages limits our capacity to attribute emergent impairments exclusively to the degeneration of the ToM network. Two recent studies, however, warrant discussion. First, Le Bouc et alCitation35 found ToM performance impairments on a false belief task in a combined cohort of AD and bvFTD patients to correlate with hypometabolism of the left TPJ/inferior parietal cortex; however, the admixture of bvFTD patients in this analysis precludes any interpretation of these results as specific to AD. Second, Synn et alCitation16 revealed significant associations between impaired overall performance on the Frith–Happé animations task and reduced gray matter integrity of the right hippocampus and bilateral cerebellum in AD. In addition to their key role in the ToM network, the TPJ, hippocampus, and cerebellum have also been implicated in general cognitive processes such as memory and executive function (). As such, whether the involvement of these regions in ToM impairments in AD reflects a domain-specific or domain-general process remains unclear. For example, the TPJ and neighboring parietal regions play a well-established role in supporting the representation of mental states,Citation80 but are also involved in the integration of information pertinent to successful semanticCitation81 and episodic memory retrieval.Citation82 Although typically discussed in relation to episodic memory dysfunction in AD,Citation68 the hippocampus may facilitate ToM by allowing us to draw upon past experiences to anticipate social intentions and reactionsCitation83 and supporting cognitive flexibility by updating our knowledge of social structure through acquired information.Citation84 Finally, cerebellar involvement has been reported during ToM tasks requiring high levels of abstraction, such as thinking about other’s traits,Citation85 yet it is also implicated in executive control and working memory capacity.Citation86 As such, further research delineating the precise role of these regions in ToM will enable us to adjudicate between domain-specific and domain-general accounts of ToM disruption in AD.
Amyotrophic lateral sclerosis (ALS)
ALS is a neurodegenerative disease involving the degradation of motor neurons in the brainstem, spinal cord, and motor cortex, manifesting in a progressive loss of motor function. Until recently, ALS was considered primarily a motor disease, although cognitive and behavioral changes are now accepted as common features of the condition. While a proportion of ALS patients will also be diagnosed with frontotemporal dementia (FTD), up to 50% of the individuals without dementiaCitation87 develop impairments in executive functioning, language, memory, and behavior, leading to the conceptualization of ALS and FTD as lying on a disease continuum.Citation88 Accumulating evidence reveals ToM deficits in ALS, not unlike those found in bvFTD, leading to inclusion of deficits in “social cognition” in the revised diagnostic criteria for the ALS-FTD spectrum disorder.Citation89 The neuroanatomical profiles of ALS and FTD, however, are not identical, with ALS spreading from its origin in the brain stem or spinal cord into motor cortex and subsequently frontotemporal, anterior cingulate, and basal ganglia regions.Citation90 The cognitive and neurobiological processes underlying impaired ToM in ALS are thus unlikely to precisely mirror those implicated in FTD.
ToM impairments have been uncovered across the majority of tasks in ALS. This incorporates basic affective ToM tasks, involving the attribution of emotions to characters based on vignettes (emotional attribution tasks)Citation91,Citation92 or facial expressions (RMET).Citation91,Citation93 Performance on more complex measures of cognitive and affective ToM is also compromised, including false belief tasks,Citation13,Citation93 understanding thoughts and feelings of cartoon characters (Happé cartoons),Citation94,Citation95 detecting faux pas,Citation96 and attributing intentions to characters in storiesCitation92,Citation97 (but see Ref. Citation98). Findings are less consistent for making social inferences from dynamic videos (The Awareness of Social Inference Test; TASIT)Citation94,Citation99,Citation100 and judging others’ preferences based on eye-gaze (Judgment of Preference task),Citation12,Citation101–Citation103 with intact performance revealed by some, but not other, studies. On balance, though, ToM impairments appear relatively pervasive in ALS.
Mechanisms of ToM disruption in ALS
Unsurprisingly, the cognitive mechanisms underlying ToM impairments in ALS vary according to the task in question. The so-called “pure” affective tasks (eg, RMET, emotion attribution) typically do not correlate with executive function in ALS,Citation91,Citation92,Citation98 indicating that degraded affective ToM in ALS arises independently from executive dysfunction. Similarly, executive function is not associated with performance on more complex ToM tasks combining affective and cognitive components, such as faux pasCitation96 and intention attribution,Citation98 further suggesting a domain-specific impairment of ToM in ALS. By contrast, executive functions have been consistently implicated in performance on false beliefCitation13 and the Happé cartoonsCitation94,Citation95 in ALS. This suggests that these latter tasks impose greater executive demands and as such may be less sensitive in detecting “pure” ToM impairment independent of other cognitive confounds. Studies examining the contribution of cognitive processes beyond executive function are limited; however, preliminary findings hint at a role for verbal memory and nonabstract reasoning in emotional attributionCitation91 and semantic naming in Judgment of Preference.Citation102
Neuroanatomy of ToM impairments in ALS
The neural substrates of ToM disruption in ALS appear to be task dependent. Emotion attribution performance is linked to gray matter intensity decrease in the ACC/fIN regions,Citation98 as well as reduced resting state functional connectivity between PCC and mPFC and between left supramarginal gyrus and right frontoparietal regions.Citation104 As such, impairments in affective ToM in ALS may result from disruption to a distributed frontotemporo–parietal network. A separate network, however, has been implicated in the more complex process of detecting false beliefs. Poor performance on this task in ALS is associated with reduced PET glucose consumption in bilateral dorsomedial and dorsolateral prefrontal cortex and supplementary motor area,Citation13 potentially reflecting the executive demands of the test. In addition, in a task-based fMRI study, poor Judgment of Preference performance in ALS was related to reduced activation in the right precentral gyrus extending into the inferior frontal gyrus.Citation103 Intriguingly, the same study revealed that reduced ToM in ALS was associated with increased brain activity in the right postcentral gyrus.Citation103 Increased brain activity on PET imaging has also been documented in relation to the false belief task in ALS, with hypermetabolism in the left fusiform gyrus correlating with reduced performance.Citation93 Together, these findings potentially reflect compensatory activity in the face of widespread degeneration of the ToM network.
Huntington’s disease (HD)
HD is a dominantly inherited neurodegenerative disease affecting motor, psychiatric, and cognitive functioning. Although formal diagnosis of HD is based on the presence of an extrapyramidal movement disorder, the advent of genetic testing has permitted assessment of symptoms in premanifest individuals harboring the CAG-repeat expansion in the Huntingtin gene, with mounting evidence to suggest that cognitive and psychiatric deficits can actually predate motor symptom onset.Citation105 HD pathology primarily infiltrates subcortical regions including the striatum, putamen, and caudate, and related frontostriatal networks, though also affects amygdala, insula, and thalamus, with extension into wider cortical areas with disease progression.Citation106 Given this neuroanatomical signature, it is unsurprising that deficits in ToM are a cardinal feature of the manifest disease.Citation107
Mechanisms of ToM disruption in HD
In patients with manifest HD, cognitive and affective ToM impairments have been documented across the majority of studies, irrespective of task employed. This includes basic affective tasks of decoding emotions from visual cues (RMET),Citation108,Citation109 attributing emotions to story characters,Citation108 as well as low demand cognitive tasks such as ascribing mental states to animated shapes (Frith–Happé animations)Citation110 and spatial perspective taking.Citation111 More complex measures encompassing both cognitive and affective elements are also impaired in HD, including faux pas detection,Citation111,Citation112 making social inferences (TASIT),Citation108,Citation113 understanding the thoughts and feelings of cartoon characters (Happé cartoons),Citation29 and attributing intentions to others.Citation114,Citation115 Unlike other neurodegenerative disorders, ToM performance is closely linked to executive function in HD, with consistent correlations between ToM impairments and executive function emerging across all the aforementioned studies. More intriguingly, deficits in intention attribution were ameliorated when cognitive flexibility impairments were controlled for.Citation115 Taken together, these findings suggest that the pervasive ToM impairments in HD are largely attributable to executive dysfunction, and as such, domain-general in nature. One anomaly is the Judgment of Preference task, which does not correlate with executive function in HD.Citation29,Citation116 Performance on this measure is not consistently impaired in HD,Citation29 suggesting that it may capture slightly different aspects of ToM vs other tasks. Deficits in ToM have also been documented prior to motor symptom onset in premanifest HD (ie, individuals carrying the CAG-repeat expansion in the Huntingtin gene), specifically on tests of faux pas detectionCitation117 and mental state attribution (Frith–Happé task).Citation117 Other tasks, including Judgment of PreferenceCitation116 and RMET,Citation118 while intact at the whole-group level in premanifest HD, are positively associated with time to symptom onset, suggesting that alterations in ToM ability may be an early feature of the disease.
Neuroanatomy of ToM impairments in HD
To date, no neuroimaging studies to our knowledge have directly related ToM performance in manifest HD to underlying brain changes. Based on the progression of pathology, ToM impairments in manifest HD are speculated to reflect widespread cortical atrophy of frontoparietal regions supporting higher-level cognition; a proposal that meshes well with the executive contribution to these deficits. Evidence from premanifest cases, however, suggests that alterations in functional brain networks likely play a mediating role. For example, in premanifest HD, reduced task-based functional connectivity between the left amygdala and the right fusiform face area during viewing of facial expressions was correlated with poorer attribution of emotions on a separate measure of RMET.Citation118 By contrast, a task-based fMRI study of cognitive ToM, involving the attribution of intentions and beliefs to cartoon characters, failed to discriminate between premanifest and control cases on the behavioral level, with no significant differences in fMRI activation patterns emerging during task performance.Citation119 As such, early impairments in affective ToM in HD may arise due to dysfunction to regions supporting emotional processing, such as the amygdala, with more extensive ToM deficits developing across the course of the disease, as a result of progressive neurodegeneration into frontoparietal cortices.Citation107 The genetic nature of this condition offers a unique opportunity to test this hypothesis, by identifying individuals in advance of marked cognitive decline and tracking longitudinal structural and functional network changes that may give rise to ToM disruption.
Parkinson’s disease (PD)
PD is a progressive extrapyramidal movement disorder, involving the degeneration of dopaminergic neurons in substantia nigra, striatum, thalamus, and subthalamic nuclei, which secondarily affects frontostriatal loops. Although traditionally conceptualized as a motor disorder, nonmotor symptoms are common, with the majority (80%) of PD patients developing mild cognitive impairment followed by frank dementia.Citation120 The characteristic cognitive complaints in PD include executive, visuospatial, memory, and processing speed impairments,Citation121 with mounting evidence of significant social cognitive dysfunction.Citation122
Mechanisms of ToM disruption in PD
Impairments in ToM are well established in PD, evident across the majority of tests employed including basic affective tasks of ascribing emotions to faces on the RMET (eg, reference Citation123–Citation125 but see reference Citation18), determining the affective state of cartoon characters in stories (emotion attribution task),Citation126 as well as low cognitive demand tasks such as Judgment of Preference,Citation127–Citation129 and more complex cognitive and affective measures of false beliefCitation130,Citation131 (but see reference Citation125), faux pas detection,Citation18,Citation132,Citation133 understanding of nonliteral utterances (Advanced Test of Theory of Mind),Citation126,Citation134 attribution of intentions to characters in comic strips,Citation124 and other story-based tasks.Citation135 It has been suggested that ToM impairments in PD may be at least partly attributable to executive dysfunction. Significant associations between executive function and ToM performance in PD have been demonstrated on complex measures combining cognitive and affective elements, such as faux pas detection,Citation135,Citation136 Advanced Test of Theory of Mind,Citation126,Citation134 intention attribution,Citation124 and other story tasks.Citation137 Mixed findings, however, have been reported on less cognitively demanding tasks, such as Judgment of PreferenceCitation127,Citation128 and RMET,Citation123,Citation124,Citation128,Citation138 with ToM correlating with executive function only at later disease stages.Citation124,Citation127,Citation138 Collectively, this suggests the presence of an independent, domain-specific ToM deficit in early stages of PD, with executive dysfunction playing a modulating role in more complex tasks and with disease progression. The modality of testing also warrants consideration given prominent visuospatial impairments in PD. Although a recent meta-analysis found no evidence for a disproportionate impairment on visual vs verbal ToM tasks,Citation122 impaired RMET performance has been associated with poor visuospatial functionCitation139 and reduced nonverbal reasoningCitation140 in PD. Determining the precise role of visuospatial dysfunction in ToM impairment in PD is required, particularly given the heavy visual demands of many commonly used measures.
Neuroanatomy of ToM impairments in PD
The prefrontal cortex has long been implicated in ToM dysfunction in PD, given secondary disruption to frontostriatal loops as a consequence of dopaminergic degeneration.Citation141 Indeed, recent studies point to a distributed frontoparietal network mediating ToM impairments in PD, suggesting that these deficits reflect damage beyond dopaminergic pathways.Citation18 Specifically, understanding nonliteral utterances on the Advanced Test of Theory of Mind is associated with reduced gray matter volume in lateral frontal and parietal regions and reduced white matter connectivity in tracts adjacent to frontal and parietal regions (eg, superior longitudinal fasciculus).Citation134 Interestingly, correlations with these gray and frontal white matter areas are negated when executive function performance is controlled for, with integrity of the superior longitudinal fasciculus adjacent to the parietal lobe remaining significant. These findings further imply a domain-specific ToM impairment in PD, associated with the disruption of frontoparietal tracts.
Other neurodegenerative disorders
Comparatively less attention has been paid to ToM impairments in the primary progressive aphasias (eg, semantic dementia [SD], progressive nonfluent aphasia [PNFA], and logopenic aphasia [LPA]), Parkinson-related syndromes (eg, dementia with Lewy Bodies [DLB], progressive supra-nuclear palsy [PSP], and corticobasal syndrome [CBS]), and vascular dementia (VD). Emerging evidence, however, suggests significant ToM deficits across the majority of these syndromes, reflecting disruption of key nodes of the canonical ToM network. While current consensus criteria emphasize aphasia as the most prominent clinical feature,Citation142 mounting evidence of social cognitive dysfunction in primary progressive aphasia suggests that the cognitive profile extends beyond the domain of language. For example, ToM impairments have been characterized in SD on cartoon tasks designed to circumvent the semantic deficits of this syndrome.Citation60,Citation143 Importantly, a recent study revealed that impaired ToM capacity in SD persisted despite controlling for overall semantic comprehension, reflecting atrophy in right anterior temporal cortices, as well as the bilateral amygdala and temporal poles, left orbitofrontal, and insular cortices.Citation60 Similarly, ToM impairments on RMET in PNFA have been shown to relate to atrophy in the insula, temporal pole, and amygdala,Citation144 bilaterally. These studies add to a growing body of evidence implicating the right temporal lobe in the origin of social cognitive dysfunction in neurodegenerative disorders.Citation145,Citation146 While ToM has not been directly examined in LPA, early atrophy to the TPJ, along with reports of empathy loss,Citation147 converges to suggest that ToM is likely to be affected.
With regard to Parkinson-related syndromes, deficits on false belief,Citation148 faux pas,Citation148,Citation149 and RMETCitation148,Citation149 tasks have been uncovered in DLB. Interestingly, only RMET performance was correlated with executive function, supported on the neural level by an association with bilateral superior and middle frontal gyri atrophy in DLB.Citation149 By contrast, poor performance on the faux-pas test in DLB correlated with atrophy to key nodes of the ToM network, including mPFC, TPJ, precuneus, and insula,Citation149 suggesting that this test may capture core aspects of ToM impairment in this syndrome. In PSP, impairments in social inference-making have been documented on the TASIT, reflecting atrophy in mPFC.Citation150 Finally, RMET impairments in a single case of CBS have been posited to reflect mPFC hypometabolism.Citation151 The status of ToM capacity in VD remains unclear, with intact comprehension of insincere speech (TASIT),Citation27 but impaired RMET, faux pas, and false belief performance in a single case study.Citation148 These equivocal findings likely reflect the diffuse neuroanatomical predilection of this disease, which can target subcortical and/or cortical regions.
Research on ToM remains in its infancy in these disorders, yet the findings to date converge to suggest transdiagnostic alterations in ToM capacity in neurodegeneration. Future work uncovering the nature and extent of these impairments in the lesser researched syndromes is essential not only to validate this hypothesis but also to assist in identifying and managing these diagnostically challenging syndromes and providing feedback to clinicians and carers about the expected deficits. The careful study of ToM disruption in these syndromes will further refine our understanding of the neurocognitive architecture of ToM more broadly.
Summary: Toward a refined understanding of ToM impairment in neurodegeneration
Taken together, the evidence suggests that ToM impairments, and their respective neurocognitive mechanisms, vary in a syndrome-specific manner. Domain-specific impairments in ToM are present in bvFTD, ALS, and PD, reflecting pathological insult targeting, but not exclusive to, multiple nodes of the ToM network (). These deficits, however, are exacerbated with encroachment of disease pathology into brain regions supporting ancillary cognitive processes implicated in ToM, such as executive function. By contrast, the bulk of the behavioral evidence in AD points to domain-general ToM impairments, attributable to disruption of brain regions supporting memory and executive processes. Finally, ToM impairments in manifest HD patients are strongly related to executive dysfunction, suggesting a domain-general impairment. Findings of amygdala involvement in ToM dysfunction in premanifest HD, however, raise the possibility of a domain-specific impairment of affective ToM in the earliest stages of the disease. Determining how this variability across syndromes manifests on the behavioral level is essential to arrive at a comprehensive understanding of ToM and its expression in everyday functioning. For example, it may be that neurodegenerative disorders with domain-specific ToM impairments (eg, bvFTD, ALS, PD, and HD) display significantly greater disruption to real-world social functioning than those with domain-general deficits (eg, AD), an issue we tackle in the next section.
Understanding the real-world impact of ToM disturbances in neurodegeneration
The literature reviewed thus far highlights ToM disruption as a potential transdiagnostic feature across neurodegenerative disorders, mediated by common and divergent neurocognitive mechanisms. The majority of these studies, however, have employed laboratory-based measures of ToM, which arguably lack many of the contextual and situational cues that inform social behavior in the real world. While more recently developed tasks involving dynamic videos of social interactions, such as the TASIT,Citation11 offer improved ecological validity, the participant nevertheless remains an observer, rather than an active agent in the social milieu. As such, it is unclear to what extent ToM performance in the laboratory reflects real-world social functioning in its many permutations and settings. A more thorough picture of ToM deficits, and its resultant impact on interpersonal function, may be gleaned from carer observations and reports of patients’ behavior in daily life. This approach reveals syndrome-specific patterns of social functioning that do not always neatly map onto ToM performance as assessed formally on experimental tasks. For example, although significant impairments on ToM tasks have been documented in AD, such patients are widely reported to uphold appropriate social functioning, at least in the mild-to-moderate stages of the disease. In particular, social graces, interpersonal warmth, and the ability to form and maintain interpersonal relationships remain intact in AD,Citation63–Citation65 as does empathic concern and behavior.Citation152 In fact, social–emotional sensitivity may be enhanced with AD onset, attributable to increased functional connectivity between the ACC/fIN and striatum.Citation53 We suggest that this discrepancy between compromised task performance and intact real-world behavior in AD reflects the often ambiguous and contextually devoid nature of ToM tasks. For example, many of the tasks in which significant ToM impairments in AD have been documented rely upon interpretation of decontextualized stimuli, such as cartoons or photographs. Social function, however, hinges upon multifaceted interpersonal and contextual cues enabling us to interpret and understand other’s mental states (see also Ref. Citation79). Individuals with AD may find abstract tasks of ToM function overly cognitively demanding,Citation16 yet if assessed within familiar and contextually rich social settings may display appropriate behavior. Our brief survey of the literature highlights the need for a new breed of ToM tasks to better capture this dimension of social cognitive functioning in AD.
Despite limited translation of laboratory-based ToM studies to daily functioning in AD, emerging evidence suggests that ToM impairments closely mirror real-world social dysfunction in other neurodegenerative disorders. This is exemplified by bvFTD, where carer reports of severe behavioral disruption in everyday life corroborate the stark ToM impairments displayed by patients on formal testing. For example, bvFTD patients are reported to display an array of inappropriate social behaviors such as loss of empathy,Citation152 reduced tact, and failure to acknowledge the presence of others.Citation153 Furthermore, ToM deficits on formal measures are strongly correlated with carer ratings of behavioral disturbance in bvFTD,Citation32 suggesting that performance on laboratory measures accurately captures functionally relevant impairments in this syndrome. We note further parallels between ToM impairments and carer reports of behavioral dysfunction in SD. Although typically viewed as a language disorder, patients with SD can present with a similar profile of social behavioral disturbances to bvFTD,Citation154 although to a milder degree. Such disturbances include reduced empathyCitation145,Citation155 and increased egocentric behavior manifesting in a failure to consider other’s viewpoints.Citation156 In line with their ToM impairments, reductions in empathy are also reported by carers across the primary progressive aphasias, namely LPA and PNFA.Citation147 Difficulties adopting or considering the mental state of others in daily life in neurodegenerative disorders result in profound carer stress and negatively impact the patient–carer relationship,Citation157 underscoring the importance of studying real-world manifestations of ToM in these patients.
Emerging evidence also suggests an association between ToM deficits and real-world social functioning in PD, ALS, and HD. In PD, carer-reported reductions in empathy occur in parallel with impairments on ToM tasks.Citation127 Furthermore, ToM impairments in PD are associated with disturbances in social behaviourCitation127 and increased apathy.Citation126 This relationship between ToM and apathy has also been uncovered in ALS,Citation101,Citation102 which may reflect reduced motivation for social engagement in the face of difficulties adopting other’s perspectives. The directionality of this relationship, however, is yet to be explored. In addition, carer reports reveal altered empathy and impaired social functioning in a subset of ALS patients,Citation98,Citation158 although whether this subset overlaps with those demonstrating ToM deficits remains to be determined. While the direct link between ToM and social functioning has not been explored in HD, alterations in social behavior such as empathy are characteristic of the disease,Citation29 suggesting that impairments on ToM measures in HD extend to real-world dysfunction. By contrast, carers of PSP and CBS report intact empathy in these patients,Citation145 and no study, to our knowledge, has explored empathic behavior in DLB or VD. Further examination of the potential relationship between ToM impairment and social functioning across each condition is critical to establish the ecological validity of formal ToM measures.
ToM and moral reasoning
Alterations in moral behavior, or transgressions from societal values about how its members should behave, have been documented in neurodegenerative disorders. Specifically, patients with bvFTD and PD have been shown to make abnormal responses to moral dilemmas, which involve a choice between two morally conflicting actionsCitation159,Citation160 (but see Ref. Citation161). Such difficult moral decisions are proposed to require ToM, as the thoughts and feelings of other people in the scenario are often considered when choosing how to respond.Citation7 The overlap between ToM and moral reasoning is supported at the neural level, with mPFC, ACC/fIN, and PCC implicated across both processes.Citation162–Citation164 Altered moral reasoning in bvFTD correlates with impaired performance on cognitive, but not affective, ToM tasks,Citation165 and morality and ToM can also be simultaneously affected in these patients.Citation166 No such relationship between ToM and moral reasoning, however, is apparent in PD.Citation160,Citation161 Furthermore, despite impaired ToM performance in AD, moral reasoning appears intact in this syndrome.Citation159 As such, the evidence to date suggests that ToM dysfunction may contribute to altered moral behavior in some neurodegenerative disorders (ie, bvFTD), potentially attributable to atrophy in regions such as the mPFC; however, the extent to which this relationship is present across other syndromes requires further exploration.
ToM and criminality
Taken to the extreme, impairments in moral reasoning can translate into real-world transgressions, such as criminal behavior. Crimes such as theft, violence, hypersexuality, traffic violations, and even homicide have been reported in neurodegeneration.Citation167 In fact, for some individuals, lawbreaking represents the first-noticed symptom of the disease. Estimates of criminality range from 37% to 54% in bvFTD and 4.5% to 12% in AD, as well as 20%–40% in HD.Citation167,Citation168 Despite commonalities in the neural substrates of these processes (eg, mPFC, PCC, TPJ),Citation169 no study, to our knowledge, has explored the relationship between ToM dysfunction and criminal behavior in neurodegeneration. The nature of crimes committed in each syndrome, however, suggests potential syndrome-specific associations between ToM and criminality. In bvFTD, for example, crimes are often of an antisocial disinhibited nature, such as harassing strangers, sexual advances, theft from other’s purses, and urinating in public.Citation167 Similarly, in HD, antisocial behaviors of violence and reckless driving represent the most frequently committed crimes.Citation170 The socially disruptive nature of criminal behavior in bvFTD and HD may reflect an inability to consider or comprehend how behavior affects others, mediated, in part, by impaired ToM. In AD, however, criminal behavior is less likely to be of a violent or aggressive nature (eg, traffic violations, theft) and potentially attributable to general cognitive dysfunction, such as forgetting to pay for items, or failure to notice a stop sign, rather than a genuine ToM impairment. Given the significant burden criminality places on patients, families, care facilities, and society more broadly,Citation167 improved understanding of how ToM contributes to this behavior across specific syndromes is urgently required.
ToM and gullibility
Patients with neurodegenerative disorders are particularly vulnerable to financial exploitation and abuse, often falling victim to scams or being easily misled.Citation171,Citation172 Avoiding this kind of deception requires complex social processes,Citation173 including the advanced ToM function of correctly interpreting the speaker’s intention as insincere.Citation174 The intersecting network of brain regions implicated across ToM, lie comprehension,Citation175 and exploitation risk,Citation176 namely mPFC, TPJ, ATL, and cerebellum, suggests a common neural substrate mediating ToM and the avoidance of deception. bvFTD patients are widely known to fall victim to scams, make irrational financial decisions, and engage in excessive gambling. Their concomitant difficulty in interpreting the sincerity of speech, related to their broader ToM deficits,Citation27 combined with an inability to incorporate social contextual information to guide economic decisions,Citation177 may explain the high levels of gullibility displayed by bvFTD patients in daily life.Citation178,Citation179 Misinterpretation of insincere speech is also found in ADCitation180–Citation183 (but see Ref. Citation27), along with an increasing tendency to believe things that are untrue.Citation179 Financial exploitation in AD, however, likely reflects episodic memory dysfunction and overall cognitive decline.Citation179 Collectively, these findings suggest that ToM underlies gullibility in some neurodegenerative syndromes, although further research directly testing this hypothesis is required.
Concluding remarks: beyond the clinic
Translating converging findings of ToM disruption from the cognitive neuroscience literature into socially relevant behaviors such as empathy, morality, criminality, and gullibility is essential to understand the real-world implications of these impairments and their underlying neurocognitive mechanisms to inform targeted interventions. In turn, this enhanced understanding will better equip carers, clinicians, and the legal system to cope with aberrant socioemotional behavior, particularly when viewed in light of underlying neuropathology. Moreover, the study of neurodegenerative syndromes in which ToM difficulties do not manifest in disruptive behaviors may offer compelling insights regarding potential protective factors for behavioral change. These findings have important implications for legal responsibility in neurodegeneration. In summary, our review highlights how extensive neural network degeneration can impair understanding of the effect of one’s behaviors on other people, as well as detection of other’s intentions, raising important questions regarding culpability for criminal activity and accountability for financial loss in neurodegenerative disorders. Moving forward, increased awareness and further rigorous investigation of these issues are required to ensure appropriate ethical treatment, care, and quality of life for individuals living with neurodegenerative disorders and their families.
Acknowledgments
This work was supported in part by an Australian Research Council (ARC) Centre of Excellence in Cognition and its Disorders (CE110001021). CS-B is supported by the National Health and Medical Research Council (NHMRC) Postgraduate Scholarship (APP1132764). SR is supported by the Faculty of Science PhD Research Scholarship from the University of Sydney. MI is supported by an ARC Future Fellowship (FT160100096) and an ARC Discovery Project (DP180101584). The authors would like to thank Ms Heidi Cartwright for assistance with preparation of and .
Disclosure
The authors report no conflicts of interest in this work.
References
- PremackDWoodruffGDoes the chimpanzee have a theory of mind?Behav Brain Sci1978104515526
- Baron-CohenSMindblindness: An Essay On Autism and Theory of MindCambridgeMIT Press1995
- DecetyJJacksonPLThe functional architecture of human empathyBehav Cogn Neurosci Rev2004327110015537986
- HappéFCookJLBirdGThe Structure of Social Cognition: In(ter) dependence of Sociocognitive ProcessesAnnu Rev Psychol201768124326727687121
- LeslieAMThaissLDomain specificity in conceptual development: neuropsychological evidence from autismCognition19924332252511643814
- DvashJShamay-TsoorySGTheory of mind and empathy as multidimensional constructs: neurological foundationsTop Lang Disord2014344282295
- Shany-UrTRankinKPPersonality and social cognition in neurodegenerative diseaseCurr Opin Neurol201124655055522002077
- Shamay-TsoorySGHarariHAharon-PeretzJLevkovitzYThe role of the orbitofrontal cortex in affective theory of mind deficits in criminal offenders with psychopathic tendenciesCortex201046566867719501818
- PolettiMEnriciIAdenzatoMCognitive and affective theory of mind in neurodegenerative diseases: neuropsychological, neuroanatomical and neurochemical levelsNeurosci Biobehav Rev20123692147216422819986
- DodichACeramiCCrespiCDifferential impairment of cognitive and affective mentalizing abilities in neurodegenerative dementias: evidence from behavioral variant of frontotemporal dementia, Alzheimer’s disease, and mild cognitive impairmentJ Alzheimers Dis20165041011102226836153
- McdonaldSFlanaganSRollinsJKinchJTASIT: a new clinical tool for assessing social perception after traumatic brain injuryJ Head Trauma Rehabil200318321923812802165
- BurkeTElaminMBedePDiscordant performance on the “Reading the Mind in the Eyes” Test, based on disease onset in amyotrophic lateral sclerosisAmyotroph Lateral Scler Frontotemporal Degener2016177–846747227152765
- CarluerLMondouABuhourMSNeural substrate of cognitive theory of mind impairment in amyotrophic lateral sclerosisCortex201565193025618325
- RamananSde SouzaLCMoreauNDeterminants of theory of mind performance in Alzheimer’s disease: a data-mining studyCortex20178881828012370
- FriedmanOLeslieAMProcessing demands in belief-desire reasoning: inhibition or general difficulty?Dev Sci20058321822515819753
- SynnAMothakunnelAKumforFMental states in moving shapes: distinct cortical and subcortical contributions to theory of mind impairments in dementiaJ Alzheimers Dis201861252153529172002
- BoraEWalterfangMVelakoulisDTheory of mind in behavioural-variant frontotemporal dementia and Alzheimer’s disease: a meta-analysisJ Neurol Neurosurg Psychiatry201586771471925595152
- PéronJVicenteSLerayEAre dopaminergic pathways involved in theory of mind? A study in Parkinson’s diseaseNeuropsychologia200947240641418845171
- SeeleyWWCrawfordRKZhouJMillerBLGreiciusMDNeurodegenerative diseases target large-scale human brain networksNeuron2009621425219376066
- PiguetOHornbergerMMioshiEHodgesJRBehavioural-variant frontotemporal dementia: diagnosis, clinical staging, and managementLancet Neurol201110216217221147039
- SeeleyWWZhouJKimEJFrontotemporal dementia: what can the behavioral variant teach us about human brain organization?Neuroscientist201218437338521670424
- TorralvaTGleichgerrchtETorres ArdilaMJRocaMManesFFDifferential cognitive and affective theory of mind abilities at mild and moderate stages of behavioral variant frontotemporal dementiaCogn Behav Neurol2015282637026102996
- AdenzatoMCavalloMEnriciITheory of mind ability in the behavioural variant of frontotemporal dementia: an analysis of the neural, cognitive, and social levelsNeuropsychologia201048121219666039
- BoraEWalterfangMVelakoulisDTheory of mind in behavioural-variant frontotemporal dementia and Alzheimer’s disease: a meta-analysisJ Neurol Neurosurg Psychiatry201586771471925595152
- HenryJDPhillipsLHvon HippelCA meta-analytic review of theory of mind difficulties in behavioural-variant frontotemporal dementiaNeuropsychologia201456536224412701
- KippsCMHodgesJRTheory of mind in frontotemporal dementiaSoc Neurosci200613–423524418633790
- Shany-UrTPoorzandPGrossmanSNComprehension of insincere communication in neurodegenerative disease: lies, sarcasm, and theory of mindCortex201248101329134121978867
- LoughSHodgesJRMeasuring and modifying abnormal social cognition in frontal variant frontotemporal dementiaJ Psychosom Res200253263964612169338
- SnowdenJSGibbonsZCBlackshawASocial cognition in frontotemporal dementia and Huntington’s diseaseNeuropsychologia200341668870112591026
- FunkiewiezABertouxMde SouzaLCLévyRDuboisBThe sea (social cognition and emotional assessment): a clinical neuropsychological tool for early diagnosis of frontal variant of frontotemporal lobar degenerationNeuropsychology2012261819021895376
- Brioschi GuevaraAKnutsonKMWassermannEMPulaskiSGrafmanJKruegerFTheory of mind impairment in patients with behavioural variant fronto-temporal dementia (bv-FTD) increases caregiver burdenAge Ageing201544589189526025914
- GregoryCLoughSStoneVTheory of mind in patients with frontal variant frontotemporal dementia and Alzheimer’s disease: theoretical and practical implicationsBrain2002125475276411912109
- FreedmanMBinnsMASeBMurphyCDtSBlackSEStussDTTheory of mind and recognition of facial emotion in dementia: challenge to current conceptsAlzheimer Dis Assoc Disord2013271566122407224
- Fernandez-DuqueDBairdJABlackSEFalse-belief understanding in frontotemporal dementia and Alzheimer’s diseaseJ Clin Exp Neuropsychol200931448949718686116
- Le BoucRLenfantPDelbeuckXMy belief or yours? differential theory of mind deficits in frontotemporal dementia and Alzheimer’s diseaseBrain2012135Pt 103026303823065791
- CotelliMAdenzatoMCantoniVEnhancing theory of mind in behavioural variant frontotemporal dementia with transcranial direct current stimulationCogn Affect Behav Neurosci20181861065107529995274
- BertouxMO’CallaghanCDuboisBHornbergerMIn two minds: Executive functioning versus theory of mind in behavioural variant frontotemporal dementiaJ Neurol Neurosurg Psychiatry201687323123426492929
- LoughSGregoryCHodgesJRDissociation of social cognition and executive function in frontal variant frontotemporal dementiaNeurocase20017212313011320160
- KippsCMNestorPJAcosta-CabroneroJArnoldRHodgesJRUnderstanding social dysfunction in the behavioural variant of fronto-temporal dementia: the role of emotion and sarcasm processingBrain2009132Pt 359260319126572
- IbañezAManesFContextual social cognition and the behavioral variant of frontotemporal dementiaNeurology201278171354136222529204
- IrishMPiguetOHodgesJRSelf-projection and the default network in frontotemporal dementiaNat Rev Neurol20128315216122331029
- IrishMHodgesJRPiguetOEpisodic future thinking is impaired in the behavioural variant of frontotemporal dementiaCortex20134992377238823582296
- O’CallaghanCShineJMAndrews-HannaJIrishMHodgesJRHippocampal atrophy and intrinsic brain network alterations relate to impaired capacity for mind wandering in neurodegenerationBioRxiv2018
- Strikwerda-BrownCGrilliMDAndrews-HannaJIrishM“All is not lost”: episodic and semantic contributions to continuity of self in dementia2019 preprint133
- AmodioDMFrithCDMeeting of minds: The medial frontal cortex and social cognitionNat Rev Neurosci20067426827716552413
- CaminitiSPCanessaNCeramiCAffective mentalizing and brain activity at rest in the behavioral variant of frontotemporal dementiaNeuroimage Clin2015948449726594631
- AhmedRMDevenneyEMIrishMNeuronal network disintegration: common pathways linking neurodegenerative diseasesJ Neurol Neurosurg Psychiatry201687111234124127172939
- SeeleyWWCrawfordRRascovskyKFrontal paralimbic network atrophy in very mild behavioral variant frontotemporal dementiaArch Neurol200865224925518268196
- AllmanJMWatsonKKTetreaultNAHakeemAYIntuition and autism: a possible role for von Economo neuronsTrends Cogn Sci20059836737316002323
- EtkinAEgnerTKalischREmotional processing in anterior cingulate and medial prefrontal cortexTrends Cogn Sci2011152859321167765
- MenonVUddinLQSaliencyULQSaliency, switching, attention and control: a network model of insula functionBrain Struct Funct20102145–665566720512370
- KimE-JSidhuMGausSESelective frontoinsular von Economo neuron and fork cell loss in early behavioral variant frontotemporal dementiaCereb Cortex201222225125921653702
- ZhouJGreiciusMDGennatasEDDivergent network connectivity changes in behavioural variant frontotemporal dementia and Alzheimer’s diseaseBrain201013351352136720410145
- CeramiCDodichACanessaNNeural correlates of empathic impairment in the behavioral variant of frontotemporal dementiaAlzheimers Dement201410682783424589435
- MöllerCHafkemeijerAPijnenburgYALDifferent patterns of cortical gray matter loss over time in behavioral variant frontotemporal dementia and Alzheimer’s diseaseNeurobiol Aging201638213126827640
- FringsLYewBFlanaganELongitudinal grey and white matter changes in frontotemporal dementia and Alzheimer’s diseasePLoS One201493e9081424595028
- WhitwellJLBoeveBFWeigandSDBrain atrophy over time in genetic and sporadic frontotemporal dementia: a study of 198 serial magnetic resonance imagesEur J Neurol201522574575225683866
- Landin-RomeroRKumforFLeytonCEIrishMHodgesJRPiguetODisease-specific patterns of cortical and subcortical degeneration in a longitudinal study of Alzheimer’s disease and behavioural-variant frontotemporal dementiaNeuroimage2017151728027012504
- ZahnRMollJIyengarVSocial conceptual impairments in frontotemporal lobar degeneration with right anterior temporal hypo-metabolismBrain2009132360461619153155
- IrishMHodgesJRPiguetORight anterior temporal lobe dysfunction underlies theory of mind impairments in semantic dementiaBrain201413741241125324523434
- EslingerPJMoorePTroianiVOops! resolving social dilemmas in frontotemporal dementiaJ Neurol Neurosurg Psychiatry200778545746017012339
- ChenYKumforFLandin-RomeroRIrishMHodgesJRPiguetOCerebellar atrophy and its contribution to cognition in frontotemporal dementiasAnn Neurol20188419810930014499
- RankinKPKramerJHMychackPMillerBLDouble dissociation of social functioning in frontotemporal dementiaNeurology200360226627112552042
- SabatSRGladstoneCMWhat intact social cognition and social behavior reveal about cognition in the moderate stage of Alzheimer’s diseaseDementia2010916178
- SabatSRLeeJMRelatedness among people diagnosed with dementia: social cognition and the possibility of friendshipDementia2012113315327
- KumforFIrishMLeytonCTracking the progression of social cognition in neurodegenerative disordersJ Neurol Neurosurg Psychiatry201485101076108324569686
- RamananSBertouxMFlanaganELongitudinal executive function and episodic memory profiles in behavioral-variant frontotemporal dementia and Alzheimer’s diseaseJ Int Neuropsychol Soc2017231344327751195
- IrishMHornbergerMEl WahshSGrey and white matter correlates of recent and remote autobiographical memory retrieval: insights from the dementiasPLoS One2014911e11308125396740
- BertouxMde SouzaLCO’CallaghanCSocial cognition deficits: the key to discriminate behavioral variant frontotemporal dementia from Alzheimer’s disease regardless of amnesia?JAD201549410651074
- LaisneyMBonLGuiziouCDaluzeauNEustacheFDesgrangesBCognitive and affective theory of mind in mild to moderate Alzheimer’s diseaseJ Neuropsychol20137110712023088554
- VerdonCMFossatiPVernyMDieudonnéBTeilletLNadelJSocial cognition: an early impairment in dementia of the Alzheimer typeAlzheimer Dis Assoc Disord2007211253017334269
- CuervaAGSabeLKuzisGTibertiCDorregoFStarksteinSETheory of mind and pragmatic abilities in dementiaNeuropsychiatry Neuropsychol Behav Neurol200114315315811513098
- MoreauNRauzySVialletFChampagne-LavauMTheory of mind in Alzheimer disease: evidence of authentic impairment during social interactionNeuropsychology201630331232126146852
- FlissRLe GallDEtcharry-BouyxFChauviréVDesgrangesBAllainPTheory of Mind and social reserve: alternative hypothesis of progressive theory of mind decay during different stages of Alzheimer’s diseaseSoc Neurosci201611440942326490734
- ZaitchikDKoffEBrownellHWinnerEAlbertMInference of beliefs and emotions in patients with Alzheimer’s diseaseNeuropsychology2006201112016460218
- ZaitchikDKoffEBrownellHWinnerEAlbertMInference of mental states in patients with Alzheimer’s diseaseCogn Neuropsychiatry20049430131316571588
- Aboulafia-BrakhaTChristeBMartoryMDAnnoniJMTheory of mind tasks and executive functions: a systematic review of group studies in neurologyJ Neuropsychol201151395521366886
- ChoongCSDoodyGACan theory of mind deficits be measured reliably in people with mild and moderate Alzheimer’s dementia?BMC Psychol2013112825566376
- DuclosHBejaninAEustacheFDesgrangesBLaisneyMRole of context in affective theory of mind in Alzheimer’s diseaseNeuropsychologia201811936337230172829
- SaxeRKanwisherNPeople thinking about thinking people. The role of the temporo-parietal junction in “theory of mind”NeuroImage20031941835184212948738
- BinderJRDesaiRHGravesWWConantLLWhere is the semantic system? A critical review and meta-analysis of 120 functional neuro-imaging studiesCereb Cortex200919122767279619329570
- RamananSPiguetOIrishMRethinking the role of the angular gyrus in remembering the past and imagining the future: the contextual integration modelNeuroscientist201824434235229283042
- MoreauNVialletFChampagne-LavauMUsing memories to understand others: the role of episodic memory in theory of mind impairment in Alzheimer diseaseAgeing Res Rev201312483383923838323
- MontagrinASaioteCSchillerDThe social hippocampusHippocampus201828967267928843041
- van OverwalleFBaetensKMariënPVandekerckhoveMSocial cognition and the cerebellum: a meta-analysis of over 350 fMRI studiesNeuroimage20148655457224076206
- BellebaumCDaumICerebellar involvement in executive controlCerebellum20076318419217786814
- RingholzGMAppelSHBradshawMCookeNAMosnikDMSchulzPEPrevalence and patterns of cognitive impairment in sporadic ALSNeurology200565458659016116120
- NearyDSnowdenJSMannDMNorthenBGouldingPJMacdermottNFrontal lobe dementia and motor neuron diseaseJ Neurol Neurosurg Psychiatry199053123322303828
- StrongMJAbrahamsSGoldsteinLHAmyotrophic lateral sclerosis – frontotemporal spectrum disorder (ALS-FTSD): revised diagnostic criteriaAmyotroph Lateral Scler Frontotemporal Degener2017183–415317428054827
- ShenDCuiLFangJCuiBLiDTaiHVoxel-wise meta-analysis of gray matter changes in amyotrophic lateral sclerosisFront Aging Neurosci20168136427065078
- TrojsiFSicilianoMRussoATheory of mind and its neuropsychological and quality of life correlates in the early stages of amyotrophic lateral sclerosisFront Psychol193420167
- ConsonniMCatricalàEDalla BellaEGessaVCLauriaGCappaSFBeyond the consensus criteria: multiple cognitive profiles in amyotrophic lateral sclerosis?Cortex20168116216727236371
- BuhourM-SDoidyFMondouAVoxel-based mapping of grey matter volume and glucose metabolism profiles in amyotrophic lateral sclerosisEJNMMI Res2017712128266002
- WatermeyerTJBrownRGSidleKCLExecutive dysfunction predicts social cognition impairment in amyotrophic lateral sclerosisJ Neurol201526271681169025957636
- GibbonsZCSnowdenJSThompsonJCHappéFRichardsonANearyDInferring thought and action in motor neurone diseaseNeuropsychologia20074561196120717118410
- MeierSLCharlestonAJTippettLJCognitive and behavioural deficits associated with the orbitomedial prefrontal cortex in amyotrophic lateral sclerosisBrain2010133113444345720889583
- CavalloMAdenzatoMMacphersonSEKarwigGEnriciIAbrahamsSEvidence of social understanding impairment in patients with amyotrophic lateral sclerosisPLoS One2011610e2594821998727
- CeramiCDodichACanessaNEmotional empathy in amyotrophic lateral sclerosis: a behavioural and voxel-based morphometry studyAmyotroph Lateral Scler Frontotemporal Degener2014151–2212923586919
- StaiosMFisherFLindellAKOngBHoweJReardonKExploring sarcasm detection in amyotrophic lateral sclerosis using ecologically valid measuresFront Hum Neurosci2013717823734113
- SavageSALilloPKumforFKiernanMCPiguetOHodgesJREmotion processing deficits distinguish pure amyotrophic lateral sclerosis from frontotemporal dementiaAmyotrop Lateral Scler Frontotemporal Degener2014151–23946
- GirardiAMacphersonSEAbrahamsSDeficits in emotional and social cognition in amyotrophic lateral sclerosisNeuropsychology2011251536520919762
- van der HulstEJBakTHAbrahamsSImpaired affective and cognitive theory of mind and behavioural change in amyotrophic lateral sclerosisJ Neurol Neurosurg Psychiatry201586111208121525476003
- KellerJBöhmSAho-ÖzhanHEAFunctional reorganization during cognitive function tasks in patients with amyotrophic lateral sclerosisBrain Imaging Behav201812377178428600740
- TrojsiFdi NardoFSantangeloGResting state fMRI correlates of theory of mind impairment in amyotrophic lateral sclerosisCortex20179711629073458
- Vinther-JensenTLarsenIUHjermindLEA clinical classification acknowledging neuropsychiatric and cognitive impairment in Huntington’s diseaseOrphanet J Rare Dis20149111425026978
- DoganIEickhoffSBSchulzJBConsistent neurodegeneration and its association with clinical progression in Huntington’s disease: a coordinate-based meta-analysisNeurodegener Dis2013121233522922585
- BoraEVelakoulisDWalterfangMSocial cognition in Huntington’s disease: a meta-analysisBehav Brain Res201629713114026455876
- LarsenIUVinther-JensenTGadeANielsenJEVogelADo I misconstrue? Sarcasm detection, emotion recognition, and theory of mind in Huntington diseaseNeuropsychology201630218118926301773
- LagravineseGAvanzinoLRaffo de FerrariATheory of mind is impaired in mild to moderate Huntington’s disease independently from global cognitive functioningFront Psychol20178318028184209
- EddyCMRickardsHEInteraction without intent: the shape of the social world in Huntington’s diseaseSoc Cogn Affect Neurosci20151091228123525680992
- EddyCMSira MahalingappaSRickardsHEPutting things into perspective: the nature and impact of theory of mind impairment in Huntington’s diseaseEur Arch Psychiatry Clin Neurosci2014264869770524647535
- EddyCMSira MahalingappaSRickardsHEIs Huntington’s disease associated with deficits in theory of mind?Acta Neurol Scand2012126637638322404634
- PhilpottALAndrewsSCStaiosMChurchyardAFisherFEmotion evaluation and social inference impairments in Huntington’s diseaseJHD20165217518327163947
- AllainPHavet-ThomassinVVernyCEvidence for deficits on different components of theory of mind in Huntington’s diseaseNeuropsychology201125674175121728429
- BrüneMBlankKWitthausHSaftC“Theory of mind” is impaired in Huntington’s diseaseMov Disord201126467167821384426
- AdjeroudNBesnardJMassiouiNETheory of mind and empathy in preclinical and clinical Huntington’s diseaseSoc Cogn Affect Neurosci2016111899926211015
- EddyCMRickardsHETheory of mind can be impaired prior to motor onset in Huntington’s diseaseNeuropsychology201529579279825664466
- MasonSLZhangJBegetiFThe role of the amygdala during emotional processing in Huntington’s disease: from pre-manifest to late stage diseaseNeuropsychologia201570808925700742
- SaftCLissekSHoffmannRMentalizing in preclinical Huntington’s disease: an fMRI study using Cartoon picture storiesBrain Imaging Behav20137215416223179063
- AarslandDBeyerMKKurzMWDementia in Parkinson’s diseaseCurr Opin Neurol200821667668218989112
- MuslimovićDPostBSpeelmanJDSchmandBCognitive profile of patients with newly diagnosed Parkinson diseaseNeurology20056581239124516247051
- BoraEWalterfangMVelakoulisDTheory of mind in Parkinson’s disease: a meta-analysisBehav Brain Res201529251552026166188
- XiCZhuYMuYTheory of mind and decision-making processes are impaired in Parkinson’s diseaseBehav Brain Res201527922623325435317
- EnriciIMitkovaACastelliLLanotteMLopianoLAdenzatoMDeep brain stimulation of the subthalamic nucleus does not negatively affect social cognitive abilities of patients with Parkinson’s diseaseSci Rep201771941328842656
- PalermoSLopianoLZibettiMRosatoRLeottaDAmanzioMA novel framework for understanding reduced awareness of dyskinesias in Parkinson’s diseaseParkinsonism Relat Disord201739586328325582
- SantangeloGVitaleCTrojanoLNeuropsychological correlates of theory of mind in patients with early Parkinson’s diseaseMov Disord20122719810521915910
- NarmePMourasHRousselMDuruCKrystkowiakPGodefroyOEmotional and cognitive social processes are impaired in Parkinson’s disease and are related to behavioral disordersNeuropsychology201327218219223527646
- BoddenMEMollenhauerBTrenkwalderCAffective and cognitive theory of mind in patients with Parkinson’s diseaseParkinsonism Relat Disord201016746647020538499
- BoddenMEDodelRKalbeETheory of mind in Parkinson’s disease and related basal ganglia disorders: a systematic reviewMov Disord2010251132719908307
- EddyCMBeckSRMitchellIJPraamstraPPallHSTheory of mind deficits in Parkinson’s disease: a product of executive dysfunction?Neuropsychology2013271374723356595
- SaltzmanJStraussEHunterMArchibaldSTheory of mind and executive functions in normal human aging and Parkinson’s diseaseJ Int Neuropsychol Soc20006778178811105468
- RocaMTorralvaTGleichgerrchtEImpairments in social cognition in early medicated and unmedicated Parkinson diseaseCogn Behav Neurol201023315215820829664
- NobisLSchindlbeckKEhlenFTheory of mind performance in Parkinson’s disease is associated with motor and cognitive functions, but not with symptom lateralizationJ Neural Transm201712491067107228584926
- Díez-CirardaMOjedaNPeñaJNeuroanatomical correlates of theory of mind deficit in parkinson’s disease: a multimodal imaging studyPLoS One20151011e014223426559669
- YuRLRmWChiuMJTaiCHLinCHHuaMSAdvanced theory of mind in patients at early stage of Parkinson’s diseaseParkinsonism Relat Disord2012181212421868278
- CostaABakTCaffarraPThe need for harmonisation and innovation of neuropsychological assessment in neurodegenerative dementias in Europe: consensus document of the joint program for neurodegenerative diseases Working GroupAlzheimers Res Ther2017912728412978
- MonettaLGrindrodCMPellMDIrony comprehension and theory of mind deficits in patients with Parkinson’s diseaseCortex200945897298119371867
- PolettiMVergalloAUliviMSonnoliABonuccelliUAffective theory of mind in patients with Parkinson’s diseasePsychiatry Clin Neurosci201367427327623683159
- MckinlayAAlbiciniMKavanaghPThe effect of cognitive status and visuospatial performance on affective theory of mind in Parkinson’s diseaseNDT2013910711076
- RocaMManesFChadeAThe relationship between executive functions and fluid intelligence in Parkinson’s diseasePsychol Med201242112445245222440401
- MengelbergASiegertRJIs theory-of-mind impaired in Parkinson’s disease?Cogn Neuropsychiatry20038319120916571560
- Gorno-TempiniMLHillisAEWeintraubSClassification of primary progressive aphasia and its variantsNeurology201176111006101421325651
- DuvalCBejaninAPiolinoPTheory of mind impairments in patients with semantic dementiaBrain2012135122824122232593
- CoutoBManesFMontañésPStructural neuroimaging of social cognition in progressive non-fluent aphasia and behavioral variant of fronttemporal dementiaFront Hum Neurosci2013746723966929
- RankinKPGorno-TempiniMLAllisonSCStructural anatomy of empathy in neurodegenerative diseaseBrain2006129112945295617008334
- IrishMKumforFHodgesJRPiguetOA tale of two hemispheres: contrasting socioemotional dysfunction in right-versus left-lateralised semantic dementiaDement Neuropsychol201371889529213824
- HazeltonJLIrishMHodgesJRPiguetOKumforFCognitive and affective empathy disruption in non-fluent primary progressive aphasia syndromesBrain Impairment20171801117129
- ModinosGObiolsJEPousaEVicensJTheory of mind in different dementia profilesJ Neuropsychiatry Clin Neurosci200921110010119359462
- HeitzCNobletVPhillippsCCognitive and affective theory of mind in dementia with Lewy bodies and Alzheimer’s diseaseAlzheimers Res Ther2016811026979460
- GhoshBCPCalderAJPeersPVSocial cognitive deficits and their neural correlates in progressive supranuclear palsyBrain201213572089210222637582
- PolettiMBonuccelliUImpairment of affective theory of mind in corticobasal degenerationJ Neuropsychiatry Clin Neurosci2012241E7
- DermodyNWongSAhmedRPiguetOHodgesJRIrishMUncovering the neural bases of cognitive and affective empathy deficits in Alzheimer’s disease and the behavioral-variant of frontotemporal dementiaJ Alzheimers Dis201653380181627258418
- MendezMFFongSSShapiraJSObservation of social behavior in frontotemporal dementiaAm J Alzheimers Dis Other Demen201429321522124370617
- HodgesJRPattersonKSemantic dementia: a unique clinicopathological syndromeLancet Neurol20076111004101417945154
- RankinKPKramerJHMillerBLPatterns of cognitive and emotional empathy in frontotemporal lobar degenerationCogn Behav Neurol2005181283615761274
- BonLBelliardSEustacheFDesgrangesBBehavioural egocentrism in semantic dementia: effect of deficits in theory of mind or cognitive egocentrism?Rev Neuropsychol200912133149
- HsiehSIrishMDavesonNHodgesJRPiguetOWhen one loses empathy: its effect on carers of patients with dementiaJ Geriatr Psychiatry Neurol201326317418423857888
- AbrahamsSNewtonJNivenEFoleyJBakTHScreening for cognition and behaviour changes in ALSAmyotroph Lateral Scler Frontotemporal Degener2014151–291423781974
- MendezMFShapiraJSAltered emotional morality in frontotemporal dementiaCogn Neuropsychiatry200914316517919499384
- RosenJBRottEEbersbachGKalbeEAltered moral decision-making in patients with idiopathic Parkinson’s diseaseParkinsonism Relat Disord201521101191119926318961
- RosenJBBrandMPolzerCEbersbachGKalbeEMoral decision-making and theory of mind in patients with idiopathic Parkinson’s diseaseNeuropsychology201327556257224040930
- ChiongWWilsonSMD’EspositoMThe salience network causally influences default mode network activity during moral reasoningBrain201313661929194123576128
- Andrews-HannaJRSmallwoodJSprengRNThe default network and self-generated thought: component processes, dynamic control, and clinical relevanceAnn NY Acad Sci201413161295224502540
- van den StockJStamDde WinterFLMoral processing deficit in behavioral variant frontotemporal dementia is associated with facial emotion recognition and brain changes in default mode and salience network areasBrain Behav2017712e0084329299378
- GleichgerrchtETorralvaTRocaMPoseMManesFThe role of social cognition in moral judgment in frontotemporal dementiaSoc Neurosci20116211312220706963
- LoughSKippsCMTreiseCEmotion and empathy in frontotemporal dementiaNeuropsychologia200644695095816198378
- LiljegrenMNaasanGTemlettJCriminal behavior in frontotemporal dementia and Alzheimer’s diseaseJAMA Neurol201572329530025559744
- DewhurstKOliverJEMcknightALSocio-psychiatric consequences of Huntington’s diseaseBr J Psychiatry19701165322552584244788
- DarbyRRHornACushmanFFoxMDLesion network localization of criminal behaviorProc Natl Acad Sci U S A2018115360160629255017
- JensenPFengerKBolwigTGSørensenSACrime in Huntington’s disease: a study of registered offences among patients, relatives, and controlsJ Neurol Neurosurg Psychiatry19986544674719771767
- LichtenbergPAFinancial exploitation, financial capacity, and Alzheimer’s diseaseAm Psychol201671431232027159438
- TronettiPEvaluating abuse in the patient with dementiaClin Geriatr Med201430482583825439644
- SprengRNKarlawishJMarsonDCCognitiveMDCCognitive, social, and neural determinants of diminished decision-making and financial exploitation risk in aging and dementia: a review and new modelJ Elder Abuse Negl2016284–532034427644698
- WinnerEBrownellHHappéFBlumAPincusDDistinguishing lies from jokes: theory of mind deficits and discourse interpretation in right hemisphere brain-damaged patientsBrain Lang1998621891069570881
- HaradaTItakuraSXuFNeural correlates of the judgment of lying: a functional magnetic resonance imaging studyNeurosci Res2009631243418992288
- AspEManzelKKoestnerBColeCADenburgNLTranelDA neuropsychological test of belief and doubt: damage to ventrome-dial prefrontal cortex increases credulity for misleading advertisingFront Neurosci2012610010022787439
- O’CallaghanCBertouxMIrishMFair play: social norm compliance failures in behavioural variant frontotemporal dementiaBrain2016139120421626503957
- PressmanPSMillerBLDiagnosis and management of behavioral variant frontotemporal dementiaBiol Psychiatry201475757458124315411
- WongSIrishMO’CallaghanCShould I trust you? learning and memory of social interactions in dementiaNeuropsychologia201710415716728811259
- AgCSabeLKuzisGTheory of mind and pragmatic abilities in dementiaCogn Behav Neurol2001143153158
- MakiYYamaguchiTKoedaTYamaguchiHCommunicative competence in Alzheimer’s disease: metaphor and sarcasm comprehensionAm J Alzheimers Dis Other Demen2013281697423221027
- YamaguchiTMakiYTakatamaMYamaguchiHGullibility may be a warning sign of Alzheimer’s disease dementiaInt Psychogeriatr2018118
- YamaguchiTMakiYYamaguchiHPitfall intention explanation task with clue questions (pitfall task): assessment of comprehending other people’s behavioral intentions in Alzheimer’s diseaseInt Psychogeriatr201224121919192622874819
