Abstract
Curcumin, a natural polyphenol compound, exhibits tumor suppressive activity in a wide spectrum of cancers, including nasopharyngeal carcinoma cells. However, the exact molecular mechanisms governing this tumor suppressive activity remain elusive. Multiple studies have revealed that miRNAs are critically involved in tumorigenesis, indicating that targeting miRNAs could be a therapeutic strategy for treating human cancer. In the current study, we set out to determine whether curcumin regulates miR-7 expression in nasopharyngeal carcinoma cells. We found that curcumin inhibited cell growth, induced apoptosis, retarded cell migration and invasion, and triggered cell cycle arrest in the human nasopharyngeal carcinoma cell lines CNE1 and CNE2. Importantly, we observed that curcumin upregulated the expression of miR-7 and subsequently inhibited Skp2, a direct miR-7 target. Our results identified that upregulation of miR-7 by curcumin could benefit nasopharyngeal carcinoma patients.
Introduction
Nasopharyngeal carcinoma (NPC) is a rare form of head and neck cancer with an estimated 86,700 new cases and 50,800 deaths globally in 2012.Citation1 However, NPC has relatively high incidence rates in Southeast Asia and South-Eastern China, which could be due to genetic factors, environment, and Epstein Barr virus infection.Citation2 Interestingly, consumption of certain foods has been associated with NPC incidence, such as salted fish, hot spices, and preserved foods. In addition, smoking and alcohol use may also contribute to NPC incidence.Citation1 Radiotherapy is the primary method of treatment for NPC. Radiotherapy in combination with chemotherapy is an effective treatment for locoregionally advanced NPC,Citation3 however, survival benefit failed to be significantly improved irrespective of the chemotherapeutic agent or dosing schedules.Citation4 The reasons leading to low survival rate for advanced stage NPC may be related to metastasis and drug resistance.
miRNAs are small non-coding RNA molecules that regulate various cellular processes by binding to complementary target sites in the 3′ untranslated regions of mRNAs, and regulating mRNA degradation or translational repression.Citation5 It has been demonstrated that miRNAs are critically involved in tumorigenesis,Citation6 where they may play either an oncogenic or tumor suppressive role. Consistent with this, high expression of oncogenic miRNAs and down-regulation of antitumor miRNAs are observed in many cancers.Citation7 For example, miR-7 has been considered as a tumor suppressive miRNA in a variety of human malignancies,Citation8,Citation9 and down-regulation of miR-7 has been observed in a variety of human cancers.Citation10–Citation13 Intriguingly, miR-7 was reported to inhibit cell growth, migration, and invasion via direct targeting of PAK1 in thyroid cancer cells.Citation14 Similarly, miR-7-5p inhibited melanoma cell proliferation and metastasis through suppressing RelA/NF-κB.Citation15 Consistently, miR-7 inhibited colon cancer invasion and proliferation through targeting the expression of FAK.Citation16 Zhao et al reported that miR-7 enhanced cytotoxicity of gefitinib through inhibition of EGFR and IGF1R pathways in non-small cell lung cancer (NSCLC).Citation17 Moreover, docetaxel inhibited cell proliferation via upregulation of miR-7 expression in NSCLC cells.Citation18 Furthermore, mutations within the miR-7 promoter are associated with poor prognosis of lung cancer,Citation10 and serum miR-7 levels are a predictive biomarker in esophageal squamous cell carcinoma.Citation19 Interestingly, miR-7 was identified as an oncogene in renal cell carcinoma.Citation20 Moreover, miR-7 was reported to promote epithelial cell transformation through targeting KLF4.Citation21 Therefore, it still remains unclear in which contexts miR-7 may carry out oncogenic or tumor suppressive functions in human cancers.
Curcumin, a natural compound derived from turmeric (Curcuma longa), has been reported to exhibit antitumor activity in both in vitro and in vivo studies.Citation22,Citation23 Mechanistically, curcumin has been shown to regulate multiple targets including NF-κB, Akt, Notch, mTOR, and Hh.Citation23–Citation25 Recently, a growing body of evidence has implicated curcumin in regulating the expression of multiple miRNAs.Citation26 Curcumin regulated miR-138 expression, leading to inhibition of cell proliferation and invasion in human osteosarcoma cells.Citation27 Moreover, inhibition of miR-21 was suggested to be important for curcumin-mediated anticancer effects.Citation28 Wu et al found that curcumin induced miR-146a expression, resulting in enhancement of temozolomide cytotoxicity against human glioblastoma.Citation29 In NPC, curcumin was shown to exert its anticancer effects through inhibition of miR-125a-5p expression.Citation30 In the current study, we explored the mechanism of curcumin-mediated anticancer activity in NPC cells.
Materials and methods
Cell culture and reagents
Human NPC cell lines CNE1 and CNE2 were purchased from American Type Culture Collection (ATCC), Manassas, VA, USA and maintained in Roswell Park Memorial Institute-1640 medium containing 10% fetal bovine serum with penicillin (100 U/mL) and streptomycin (100 U/mL) at 37°C with 5% CO2. Primary antibodies against Skp2, tubulin and the secondary antibodies were purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA). Anti-p21 and anti-p57 antibodies were obtained from Cell Signaling Technology (Danvers, MA, USA). Lipofectamine 2000 was purchased from Invitrogen (Thermo Fisher Scientific, Waltham, MA, USA). MTT (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide) and curcumin (CAS number 458-37-7, 99.5% purity) was obtained from Sigma-Aldrich Co. (St Louis, MO, USA). Cells were treated with 0.1% dimethyl sulfoxide (DMSO) for control conditions.
Cell viability assay
NPC cells (5×103) were seeded in a 96-well plate overnight. Cells were treated with indicated concentrations of curcumin for 48 h and 72 h. At the end of the treatment period, 10 μL of the MTT (5 mg/mL) solution was added to each well and cells were incubated for 4 h at 37°C. Then, the supernatant was absorbed and 100 μL DMSO was added to dissolve MTT-formazan crystals. The absorption was measured in the microplate at 490 nm.
Cell apoptosis analysis
The Annexin V-fluorescein isothiocyanate/propidium iodide (FITC/PI) apoptosis detection kit (Biouniqure, Shanghai, China) was used to detect the apoptotic cells. Briefly, NPC cells were incubated in 6-well plates overnight and treated with indicated concentrations of curcumin for 48 h. Cells were harvested, washed with phosphate-buffered saline (PBS), and resuspended in 500 μL of binding buffer with 5 μL PI and 5 μL FITC-conjugated anti-Annexin V antibody. The cells were kept in the dark for 15 min at room temperature. Apoptosis was measured using a FACScalibur flow cytometer (BD Biosciences, San Jose, CA, USA).
Cell cycle analysis
NPC cells were seeded in a 6-well plate and incubated overnight. The cells were treated with curcumin and cultured for 48 h, collected, and fixed with ice-cold 70% (v/v) ethanol and stored at 4°C overnight. Fixed cells were washed and resuspended at 1×106 cells/mL in PBS and incubated with 0.1 mg/mL RNase A and 50 mg/mL PI at 37°C for 30 min. PI staining was measured with a FACScalibur flow cytometer.
Wound healing assay
NPC cells were seeded in a 6-well plate and allowed to grow until confluent. Once confluent, cells were scraped off (scratched) in a straight line using a sterile pipette tip. The cells were washed with PBS and treated with curcumin for 15 h. The scratched area was photographed at 0 h and 15 h, respectively.
Transwell invasion assay
Cell invasive capacity of NPC cells was measured using transwell filter with Matrigel (BD Biosciences). Briefly, NPC cells treated with curcumin were transferred into the upper chamber in 200 μL of serum-free medium. An amount of 500 μL complete medium was added into each bottom chamber with the same concentration of curcumin. After incubation for 20 h, the cells in the upper chamber were removed, and the invaded cells in the membrane were stained with Wright-Giemsa. The cells were also stained with 4 μg/mL calcein AM in Hanks’ buffered saline at 37°C for 1 h. The stained cells were photographed and counted (at least six randomly-selected fields) under a microscope.
Western blotting analysis
NPC cells were harvested, washed with PBS, and lysed. The protein concentrations were determined using BCA Protein Assay kit (Thermo Fisher Scientific). Protein samples were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred onto a polyvinylidene difluoride membrane. The membranes were incubated with primary antibodies at 4°C overnight. After washing three times with tris-buffered saline with Tween® 20, the membranes were incubated with secondary antibody at room temperature for 1 h. The protein bands were subsequently detected by enhanced chemiluminescence assay.
MiRNA real-time reverse transcription polymerase chain reaction (RT-PCR)
RT-PCR was performed to measure the changes in miR-7 expression in NPC cells treated with curcumin. Briefly, 10 ng of total RNA was reverse transcribed into cDNA using TaqMan miRNA hsa-miR-7-specific primers (Applied Biosystems, Thermo Fisher Scientific). Then real-time PCR was conducted by a TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems, Thermo Fisher Scientific). RNA U6 was determined and used as endogenous control in each sample.
MiR-7 mimics transfection
NPC cells were transfected with miR-7 mimics (Genepharma, Shanghai, China) or a non-specific control using Lipofectamine RNAiMAX reagent (Invitrogen, Thermo Fisher Scientific) following the manufacturer’s protocol. MiR-7 mimics: sense 5′-UGG AAG ACU AGU GAU UUU GUU GU-3′; antisense 5′-AAC AAA AUC ACU AGU CUU CCA UU-3′. After the indicated periods of incubation, the cells were subjected to further analysis as described under the results section.
MiRNA-7 inhibitor transfection
Cells were seeded in 6-well plates and transfected with antisense miR-7 oligonucleotide (Genepharma) or the nonspecific control, using DharmaFect Transfection Reagent (Dharmacon, Lafayette, CO, USA). MiR-7 inhibitor: 5′-ACA ACA AAA AUC ACU AGU CUU CCA-3′.
Statistical analysis
All data analyses were conducted using GraphPad Prism 4.0 (GraphPad Software Inc., La Jolla, CA, USA). Statistical comparisons were performed using the Student’s t-test. Results are expressed as means ± standard deviation. P-values <0.05 were considered statistically significant.
Results
Curcumin inhibited cell proliferation
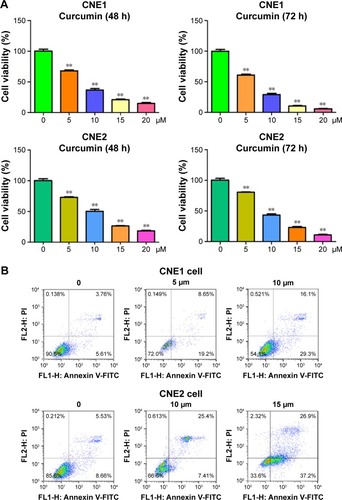
To explore whether curcumin could suppress cell proliferation in the NPC cell lines CNE1 and CNE2, cells were treated with increasing concentrations of curcumin, and subsequently cell proliferation was determined using an MTT assay. We found that curcumin significantly inhibited cell proliferation in both NPC cell lines (). Specifically, treatment with 5 μM and 10 μM of curcumin for 72 h led to 40% and 65% inhibition of cell proliferation in CNE1 cells, respectively (). In addition, treatment with 10 μM and 15 μM of curcumin for 72 h led to 55% and 70% inhibition of cell proliferation in CNE2 cells, respectively (). Therefore, curcumin inhibited cell proliferation in both NPC cell lines.
Figure 1 Effect of curcumin on NPC cell growth and apoptosis.
Abbreviations: NPC, nasopharyngeal carcinoma; MTT, (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide); DMSO, dimethyl sulfoxide; FITC/PI, fluorescein isothiocyanate/propidium iodide; FL1-H, fluorescence 1-H; FL2-H, fluorescence 2-H.
Curcumin triggered apoptosis
In order to determine whether curcumin triggered apoptosis, curcumin-treated NPC cells were stained with Annexin V-FITC/PI. We found that 5 μM and 10 μM curcumin treatments led to CNE1 cell apoptosis from 9.3% to 27.8% and 45.4%, respectively (). Similarly, the percentage of apoptotic cells increased from 14.1% in the control to 32.8% and 64.1% in 10 μM and 15 μM curcumin-treated CNE2 cells, respectively (). These findings demonstrated that curcumin induced cellular apoptosis, that contributed to the observed inhibition in cell proliferation.
Curcumin induced cell cycle arrest
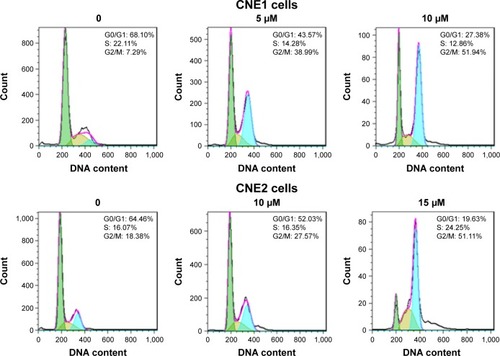
In addition to regulation of cellular proliferation by induction of apoptosis, we also assessed if cell cycle arrest was induced by curcumin to inhibit cellular proliferation of NPC cells. To determine whether curcumin regulates cell cycle progression, treated cells were stained with PI to measure DNA content, and assessed for cell cycle analysis. We observed that curcumin treatment induced a G2/M cell cycle arrest in both NPC cell lines. The G2/M phase fraction was increased from 7.29% in control cells to 38.99% and 51.94% in 5 μM and 10 μM curcumin-treated CNE1 cells, respectively (). Similar G2/M arrest was identified in curcumin-treated CNE2 cells (). These results revealed that curcumin induced cell cycle arrest in NPC cells.
Figure 2 Effect of curcumin on NPC cell cycle.
Abbreviations: NPC, nasopharyngeal carcinoma; PI, propidium iodide.
Curcumin retarded cell migration and invasion
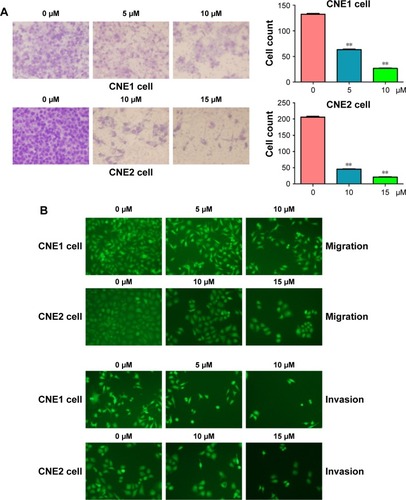
Several studies have reported that curcumin inhibited cell migration and invasion in various types of human cancer cell lines. In order to evaluate whether curcumin could govern cell motility in NPC cells, we utilized wound healing and transwell assays in NPC cells following curcumin treatment. Our wound healing assay results demonstrated that curcumin significantly inhibited cell migration (). Consistent with inhibition of cell motility by curcumin, transwell assay results demonstrated that curcumin suppressed the invasion of NPC cells into the Matrigel-coated membrane in a dose-dependent manner (). Our findings suggest that curcumin inhibited both cell migration and invasion in NPC cells.
Figure 3 Curcumin inhibited cell migration in NPC cells.
Abbreviations: NPC, nasopharyngeal carcinoma; DMSO, dimethyl sulfoxide.
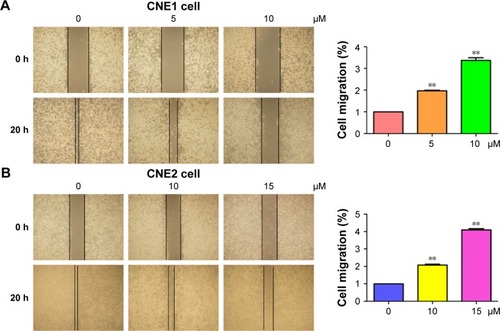
Figure 4 Curcumin inhibited cell invasion in NPC cells.
Abbreviation: NPC, nasopharyngeal carcinoma.
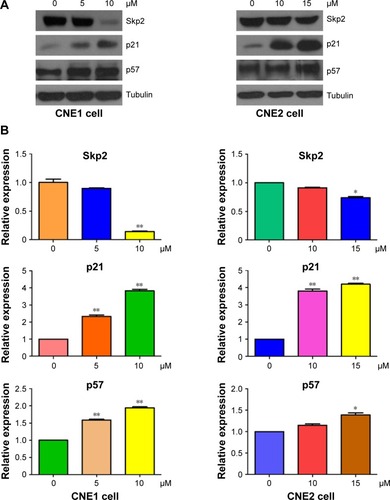
Curcumin inhibited Skp2 expression
Skp2, a substrate recognition component of an SCF E3 ubiquitin-protein ligase complex, has been characterized as an oncoprotein during tumorigenesis.Citation31 To further explore the molecular mechanism by which curcumin exerts its tumor suppressive function, the expression of Skp2 was measured by Western blotting in NPC cells after curcumin treatment. We found that curcumin down-regulated the expression of Skp2 in both NPC cell lines (). Furthermore, two targets of Skp2, p21, and p57, were upregulated in NPC cells following curcumin treatment (). Therefore, curcumin may exert its antitumor activity partly through inhibition of Skp2 and subsequent upregulation of p21 and p57 in NPC cells.
Figure 5 Curcumin inhibited the expression of Skp2 in NPC cells.
Abbreviation: NPC, nasopharyngeal carcinoma.
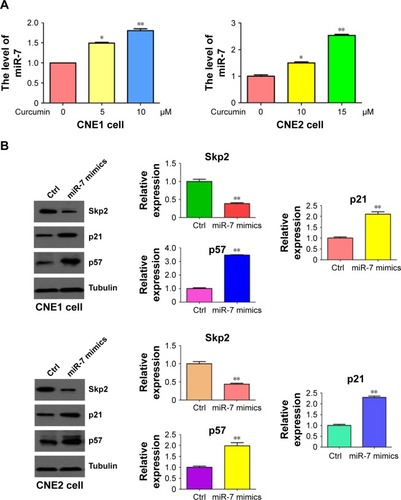
Curcumin increased miR-7 expression in NPC cells
Emerging evidence has revealed that miR-7 plays a key role in tumorigenesis. To explore whether curcumin could control the expression of miR-7 in NPC cells, miRNA RT-PCR was conducted on RNA isolated from NPC cells following curcumin treatment. As we expected, miR-7 expression was significantly upregulated in curcumin-treated NPC cells (). This observation indicated that curcumin could induce miR-7 expression in NPC cells. To further explore whether curcumin inhibited Skp2 via upregulation of miR-7, NPC cells were transfected with miR-7 mimics. Transfection of miR-7 mimics in NPC cells inhibited the expression of Skp2 and increased the abundance of p21 and p57 (). These results indicated that curcumin could inhibit Skp2 in part, through upregulation of miR-7 in NPC cells.
Figure 6 Curcumin upregulated miR-7 level in NPC cells.
Abbreviations: NPC, nasopharyngeal carcinoma; RT-PCR, reverse transcription polymerase chain reaction; Ctrl, control.
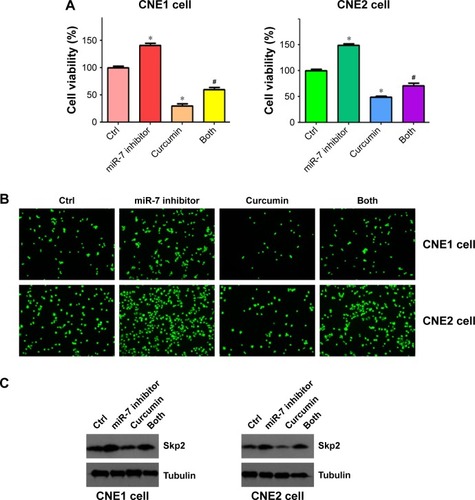
MiR-7 inhibitor enhanced cell growth in NPC cells
To further detect the role of miR-7 in cell proliferation, we transfected NPC cells with an miR-7 inhibitor and incubated them for 72 h prior to measuring cellular proliferation using an MTT assay. We observed that depletion of miR-7 promoted cell proliferation in both CNE1 and CNE2 cells (). Consistently, inhibition of miR-7 partly abrogated cell growth inhibition induced by curcumin treatment () in NPC cells. We also measured cell invasion in NPC cells following miR-7 inhibitor transfection and curcumin treatment. We observed that inhibition of miR-7 increased cell invasion in NPC cells, and partially rescued curcumin-induced inhibition of cell invasion (). Our protein analysis revealed that miR-7 inhibitor treatment led to elevated Skp2 levels (). These results indicated that curcumin exerted its antitumor effects in part, through upregulation of miR-7 and subsequent inhibition of Skp2 in NPC cells.
Figure 7 miR-7 inhibitor enhanced cell growth and invasion in NPC cells.
Abbreviations: NPC, nasopharyngeal carcinoma; MTT, (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide); Ctrl, control.
Discussion
Emerging evidence has demonstrated that curcumin exerts anticancer activity in human cancers including NPC. It has been reported that curcumin suppressed the growth of NPC cells through inducing apoptosis.Citation32 Moreover, curcumin altered the migratory phenotype of NPC cells via upregu-lation of E-cadherin.Citation33 Furthermore, curcumin induced apoptotic death via the ROS, mitochondrial depolarization, and CASP3-dependent signaling response in NPC cells.Citation34 Similarly, treating NPC cells with a new curcumin analog exhibited anticancer function and induced radiosensitivity through inactivation of Jab1.Citation35,Citation36 Notably, curcumin was also observed to enhance radiosensitivity through regulation of long non-coding RNAs in NPC.Citation37 Additionally, curcumin altered ERK1/2 signaling pathway and regulation of FOXO3a and p53, leading to inhibition of cell proliferation in NPC cells.Citation38,Citation39 In this study, we found that curcumin may also exert its antitumor activity in part, through suppression of Skp2 in NPC cells.
Recently, miR-7 has been considered as an important regulator in tumorigenesis. Regulation of EGFR and erlotinib sensitivity by miR-7 has been reported in head and neck cancer cells.Citation40 It has been shown that overexpression of miR-7 enhanced efficacy of green tea polyphenols due to induction of apoptosis in human malignant neuroblastoma cells.Citation40 Moreover, miR-7 inhibited brain metastasis of breast cancer stem-like cells via targeting of KLF4.Citation41 Ma et al found that down-regulation of miR-7 upregulated CUL5 to facilitate the G1/S transition.Citation42 Xu et al reported that miR-7 suppressed cell proliferation and induced apoptosis via inhibition of XRCC2 in colorectal cancer cells.Citation43 Similarly, miR-7 inhibited the invasion and metastasis in part, through suppressing EGFR expression in gastric cancer cells.Citation44 Restoration of miR-7 inhibited cell growth via modulation of EGFR signaling in Lewis lung cancer cells.Citation45 Furthermore, miR-7 inhibited cell growth via interference with PI3K/Akt and Raf/MEK/ERK pathways in glioblastoma.Citation46 Strikingly, miR-7 inhibited SETDB1 and reversed EMT of breast cancer stem cells through down-regulation of the STAT3 pathway.Citation47 It has been identified that Bcl-2 could be one target of miR-7 in serous ovarian carcinomas.Citation48 Recently, Liu et al identified that miR-7 modulated chemoresistance through repression of MRP1/ABCC1 in small cell lung cancer.Citation49 Notably, systemic miR-7 delivery inhibited tumor angiogenesis and growth in a murine xenograft model of glioblastoma.Citation50 Interestingly, miR-7 played a critical role in radioresistance of NPC cells to X-ray dose.Citation51 Chen et al reported that NPC cells significantly increased the expression of miR-7 after radiation treatment, indicating that suppression of miR-7 expression may enhance the radiosensitivity of NPC cells through upregulation of EGFR expression.Citation51 Strikingly, miR-7 triggered cell cycle arrest through inhibition of Skp2 expression in human cancer cells.Citation52 Indeed, in our study we also observed that miR-7 overexpression inhibited the expression of Skp2 in NPC cells. More importantly, our findings showed that curcumin increased miR-7 levels in NPC cells. Altogether, our results reveal that curcumin may exert its antitumor activity by increasing miR-7 expression, leading to a subsequent decrease in Skp2 expression in NPC cells.
Skp2 plays an oncogenic role by targeting and degrading its ubiquitination targets such as p21,Citation53 p27,Citation54 p57,Citation55 E-cadherin,Citation56 and FOXO1.Citation57 Higher expression of Skp2 is associated with poor prognosis in human cancers including pancreatic cancer,Citation58,Citation59 prostate cancer,Citation59 breast cancer,Citation60,Citation61 and glioma.Citation62 Wang et al reported that Skp2 expression predicted poor prognosis and maintained the cancer stem cell pool in NPC.Citation63–Citation65 Since Skp2 is a key oncoprotein, it is important to identify Skp2 inhibitors for treating human cancers. Several Skp2 inhibitors such as CpdA have been reported to block Skp2 E3 ligase activity.Citation66 Another Skp2 inhibitor, SZL-P1-41, was reported to trigger cellular senescence.Citation67 Due to the toxic nature of these chemical inhibitors, it is necessary to identify natural agents to target Skp2. In the present study, we validated that curcumin could be a safe agent for inactivation of Skp2 via upregulation of miR-7 in NPC.
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (NSFC 81572936) and a project funded by the priority academic program development of Jiangsu higher education institutions.
Disclosure
The authors report no conflicts of interest in this work.
References
- TorreLABrayFSiegelRLFerlayJLortet-TieulentJJemalAGlobal cancer statistics, 2012CA Cancer J Clin20156528710825651787
- DaiWZhengHCheungAKLungMLGenetic and epigenetic landscape of nasopharyngeal carcinomaChin Clin Oncol2016521627121876
- SzeHBlanchardPNgWTPignonJPLeeAWChemotherapy for nasopharyngeal carcinoma – current recommendation and controversiesHematol Oncol Clin North Am20152961107112226568551
- ChenQYWenYFGuoLConcurrent chemoradiotherapy vs radiotherapy alone in stage II nasopharyngeal carcinoma: phase III randomized trialJ Natl Cancer Inst2011103231761177022056739
- BartelDPMicroRNAs: target recognition and regulatory functionsCell2009136221523319167326
- AmbrosVMicroRNA pathways in flies and worms: growth, death, fat, stress, and timingCell2003113667367612809598
- LinSGregoryRIMicroRNA biogenesis pathways in cancerNat Rev Cancer201515632133325998712
- LiYJWangCHZhouYTLR9 signaling repressed tumor suppressor miR-7 expression through up-regulation of HuR in human lung cancer cellsCancer Cell Int20131319024004462
- HansenTBKjemsJDamgaardCKCircular RNA and miR-7 in cancerCancer Res201373185609561224014594
- ZhaoJWangKLiaoZPromoter mutation of tumor suppressor microRNA-7 is associated with poor prognosis of lung cancerMol Clin Oncol2015361329133626807243
- HorshamJLGandaCKalinowskiFCBrownRAEpisMRLeedmanPJMicroRNA-7: A miRNA with expanding roles in development and diseaseInt J Biochem Cell Biol20156921522426546742
- ZhaoJTaoYZhouYMicroRNA-7: a promising new target in cancer therapyCancer Cell Int20151510326516313
- GloverARZhaoJTGillAJMicroRNA-7 as a tumor suppressor and novel therapeutic for adrenocortical carcinomaOncotarget2015634366753668826452132
- YueKWangXWuYZhouXHeQDuanYmicroRNA-7 regulates cell growth, migration and invasion via direct targeting of PAK1 in thyroid cancerMol Med Rep20161432127213427430434
- GilesKMBrownRAGandaCmicroRNA-7–5p inhibits melanoma cell proliferation and metastasis by suppressing RelA/NF-kappaBOncotarget2016722316633168027203220
- ZengCYZhanYSHuangJChenYXMicroRNA-7 suppresses human colon cancer invasion and proliferation by targeting the expression of focal adhesion kinaseMol Med Rep20161321297130326648422
- ZhaoJGMenWFTangJMicroRNA-7 enhances cytotoxicity induced by gefitinib in non-small cell lung cancer via inhibiting the EGFR and IGF1R signalling pathwaysContemp Oncol (Pozn)201519320120626557760
- HeXLiCWuXYangGDocetaxel inhibits the proliferation of non-small-cell lung cancer cells via upregulation of microRNA-7 expressionInt J Clin Exp Pathol2015889072908026464649
- DongWLiBWangJDiagnostic and predictive significance of serum microRNA-7 in esophageal squamous cell carcinomaOncol Rep20163531449145626708917
- YuZNiLChenDIdentification of miR-7 as an oncogene in renal cell carcinomaJ Mol Histol201344666967723793934
- Meza-SosaKFPerez-GarciaEICamacho-ConchaNLopez-GutierrezOPedraza-AlvaGPerez-MartinezLMiR-7 promotes epithelial cell transformation by targeting the tumor suppressor KLF4PLoS One201499e10398725181544
- AggarwalBBDebLPrasadSCurcumin differs from tetrahydrocurcumin for molecular targets, signaling pathways and cellular responsesMolecules2015201185205
- KanaiMTherapeutic applications of curcumin for patients with pancreatic cancerWorld J Gastroenterol201420289384939125071333
- BeeversCSZhouHHuangSHitting the golden TORget: curcumin’s effects on mTOR signalingAnticancer Agents Med Chem201313798899423272912
- ShehzadALeeYSMolecular mechanisms of curcumin action: signal transductionBiofactors2013391273623303697
- MomtaziAAShahabipourFKhatibiSJohnstonTPPirroMSahebkarACurcumin as a MicroRNA regulator in cancer: a reviewRev Physiol Biochem Pharmacol201617113827457236
- YuDAnFHeXCaoXCurcumin inhibits the proliferation and invasion of human osteosarcoma cell line MG-63 by regulating miR-138Int J Clin Exp Pathol2015811149461495226823826
- ChenJXuTChenCThe critical roles of miR-21 in anti-cancer effects of curcuminAnn Transl Med201532133026734640
- WuHLiuQCaiTChenYDWangZFInduction of microRNA-146a is involved in curcumin-mediated enhancement of temozolomide cytotoxicity against human glioblastomaMol Med Rep20151245461546626239619
- GaoWChanJYWongTSCurcumin exerts inhibitory effects on undifferentiated nasopharyngeal carcinoma by inhibiting the expression of miR-125a-5pClin Sci (Lond)2014127957157924896104
- GaoJKWangLXLongBArsenic trioxide inhibits cell growth and invasion via down-regulation of Skp2 in pancreatic cancer cellsAsian Pac J Cancer Prev20151693805381025987041
- LinYTWangLFHsuYCCurcuminoids suppress the growth of pharynx and nasopharyngeal carcinoma cells through induced apoptosisJ Agric Food Chem20095793765377019317462
- WongTSChanWSLiCHCurcumin alters the migratory phenotype of nasopharyngeal carcinoma cells through up-regulation of E-cadherinAnticancer Res20103072851285620683022
- KuoCLWuSYIpSWApoptotic death in curcumin-treated NPC-TW 076 human nasopharyngeal carcinoma cells is mediated through the ROS, mitochondrial depolarization and caspase-3-dependent signaling responsesInt J Oncol201139231932821617861
- PanYXiaoJLiangGA new curcumin analogue exhibits enhanced antitumor activity in nasopharyngeal carcinomaOncol Rep201330123924523673810
- PanYWangMBuXCurcumin analogue T83 exhibits potent antitumor activity and induces radiosensitivity through inactivation of Jab1 in nasopharyngeal carcinomaBMC Cancer20131332323815987
- WangQFanHLiuYCurcumin enhances the radiosensitivity in nasopharyngeal carcinoma cells involving the reversal of differentially expressed long non-coding RNAsInt J Oncol201444385886424379026
- XieYQWuXBTangSQCurcumin treatment alters ERK-1/2 signaling in vitro and inhibits nasopharyngeal carcinoma proliferation in mouse xenograftsInt J Clin Exp Med20147110811424482695
- WuJTangQZhaoSExtracellular signal-regulated kinase signaling-mediated induction and interaction of FOXO3a and p53 contribute to the inhibition of nasopharyngeal carcinoma cell growth by curcuminInt J Oncol20144519510324806432
- ChakrabartiMAiWBanikNLRaySKOverexpression of miR-7-1 increases efficacy of green tea polyphenols for induction of apoptosis in human malignant neuroblastoma SH-SY5Y and SK-N-DZ cellsNeurochem Res201338242043223192662
- OkudaHXingFPandeyPRmiR-7 suppresses brain metastasis of breast cancer stem-like cells by modulating KLF4Cancer Res20137341434144423384942
- MaCQiYShaoLLiuMLiXTangHDownregulation of miR-7 upregulates Cullin 5 (CUL5) to facilitate G1/S transition in human hepatocellular carcinoma cellsIUBMB Life201365121026103424339204
- XuKChenZQinCSongXmiR-7 inhibits colorectal cancer cell proliferation and induces apoptosis by targeting XRCC2Onco Targets Ther2014732533224570594
- XieJChenMZhouJmiR-7 inhibits the invasion and metastasis of gastric cancer cells by suppressing epidermal growth factor receptor expressionOncol Rep20143141715172224573489
- LiJZhengYSunGXiongSRestoration of miR-7 expression suppresses the growth of Lewis lung cancer cells by modulating epidermal growth factor receptor signalingOncol Rep20143262511251625334070
- LiuZJiangZHuangJmiR-7 inhibits glioblastoma growth by simultaneously interfering with the PI3K/ATK and Raf/MEK/ERK pathwaysInt J Oncol20144451571158024603851
- ZhangHCaiKWangJMiR-7, inhibited indirectly by lincRNA HOTAIR, directly inhibits SETDB1 and reverses the EMT of breast cancer stem cells by downregulating the STAT3 pathwayStem Cells201432112858286825070049
- SwierczAChechlinskaMKupryjanczykJmiR-7 expression in serous ovarian carcinomasAnticancer Res20153542423242925862909
- LiuHWuXHuangJPengJGuoLmiR-7 modulates chemoresistance of small cell lung cancer by repressing MRP1/ABCC1Int J Exp Pathol201596424024726108539
- BabaeNBourajjajMLiuYSystemic miRNA-7 delivery inhibits tumor angiogenesis and growth in murine xenograft glioblastomaOncotarget20145166687670025149532
- ChenZXSunAMChenYEffects of radiosensitivity and X-ray dose on miR-7 expression in nasopharyngeal carcinomaNan Fang Yi Ke Da Xue Xue Bao2010308181018121816 Chinese20813671
- SanchezNGallagherMLaoNMiR-7 triggers cell cycle arrest at the G1/S transition by targeting multiple genes including Skp2 and Psme3PLoS One201386e6567123762407
- YuZKGervaisJLZhangHHuman CUL-1 associates with the SKP1/SKP2 complex and regulates p21(CIP1/WAF1) and cyclin D proteinsProc Natl Acad Sci U S A1998951911324113299736735
- TsvetkovLMYehKHLeeSJSunHZhangHp27(Kip1) ubiquitination and degradation is regulated by the SCF(Skp2) complex through phosphorylated Thr187 in p27Curr Biol199991266166410375532
- KamuraTHaraTKotoshibaSDegradation of p57Kip2 mediated by SCFSkp2-dependent ubiquitylationProc Natl Acad Sci U S A200310018102311023612925736
- InuzukaHGaoDFinleyLWAcetylation-dependent regulation of Skp2 functionCell2012150117919322770219
- HuangHReganKMWangFSkp2 inhibits FOXO1 in tumor suppression through ubiquitin-mediated degradationProc Natl Acad Sci U S A200510251649165415668399
- EinamaTKagataYTsudaHHigh-level Skp2 expression in pancreatic ductal adenocarcinoma: correlation with the extent of lymph node metastasis, higher histological grade, and poorer patient outcomePancreas200632437638116670630
- WangZGaoDFukushimaHSkp2: a novel potential therapeutic target for prostate cancerBiochim Biophys Acta201218251111721963805
- RadkeSPirkmaierAGermainDDifferential expression of the F-box proteins Skp2 and Skp2B in breast cancerOncogene200524213448345815782142
- FujitaTLiuWDoiharaHDateHWanYDissection of the APCCdh1-Skp2 cascade in breast cancerClin Cancer Res20081471966197518381934
- WangLYeXCaiXCurcumin suppresses cell growth and invasion and induces apoptosis by down-regulation of Skp2 pathway in glioma cellsOncotarget2015620180271803726046466
- WangJHuangYGuanZE3-ligase Skp2 predicts poor prognosis and maintains cancer stem cell pool in nasopharyngeal carcinomaOncotarget20145145591560125015320
- XuHMLiangYChenQCorrelation of Skp2 overexpression to prognosis of patients with nasopharyngeal carcinoma from South ChinaChin J Cancer201130320421221352698
- FangFMChienCYLiCFShiuWYChenCHHuangHYEffect of S-phase kinase-associated protein 2 expression on distant metastasis and survival in nasopharyngeal carcinoma patientsInt J Radiat Oncol Biol Phys200973120220718538504
- ChenQXieWKuhnDJTargeting the p27 E3 ligase SCF(Skp2) results in p27- and Skp2-mediated cell-cycle arrest and activation of autophagyBlood200811194690469918305219
- ChanCHMorrowJKLiCFPharmacological inactivation of Skp2 SCF ubiquitin ligase restricts cancer stem cell traits and cancer progressionCell2013154355656823911321
