Abstract
Background
Recent clinical studies have shown that initial therapy with combined BRAF and mitogen-activated extracellular signal-regulated kinase (MEK) inhibition is more effective in metastatic melanoma than single-agent BRAF inhibitors. However, the response rates with single-agent BRAF are low. Thus, the objective of this study was to conduct a meta-analysis of randomized controlled trials to compare the efficacy and adverse events risk between mono-therapy and combination therapy.
Materials and methods
Searches were made in PubMed and EMBASE electronic databases and conference abstracts published by the American Society of Clinical Oncology from 2000 to 2017. Outcomes included overall response, progression-free survival, and overall survival, as well as the incidence rate of adverse events.
Results
Eight trials comprising 2,664 patients were included in the meta-analysis. Patients with combined therapies showed superior results compared to those with BRAF inhibitors alone for the following: overall response rate (combined relative risk [RR] =1.34, 95% confidence interval [95% CI]: 1.24–1.45, P<0.00001), progression-free survival (combined hazards ratio [HR] =0.58, 95% CI: 0.52–0.64, P<0.00001), and overall survival rate (combined HR =0.70, 95% CI: 0.62–0.80, P<0.00001). Patients with combination therapies had higher incidence of adverse events including pyrexia (combined RR =2.00, 95% CI: 1.40–2.84), nausea (combined RR =1.41, 95% CI: 1.03–1.94), diarrhea (combined RR =1.50, 95% CI: 1.08–2.06), and vomiting (combined RR =1.87, 95% CI: 01.52–2.31) compared to those with BRAF inhibitors alone.
Conclusion
These data suggested that the combined BRAF and MEK inhibition was associated with a significant improvement in overall response, progression-free survival, and overall survival, but increased the incidence of adverse events among the patients with BRAF V600-mutated metastatic melanoma. Further large-scale, high-quality, placebo-controlled, double-blind trials are needed to confirm this conclusion.
Introduction
The global incidence of melanoma is increasing, with about 200,000 new cases and about 65,000 melanoma-associated deaths every year.Citation1 Although metastatic melanoma typically has been associated with poor prognosis, with low survival averaged by 15%–60% in patients, the therapeutic options recently changed substanti-ally.Citation2 After the approval of BRAF inhibitor such as vemurafenib or dabrafenib by the United States Food and Drug Administration (US FDA) in 2011, there has been a significant improvement in the response rates, progression-free survival (PFS), and overall survival (OS), in patients with metastatic melanoma with BRAF V600E or V600K mutations, compared with chemotherapy.Citation3 However, the mitogen-activated protein kinase (MAPK) pathway was frequently reactivated, thereby causing acquired resistance to BRAF inhibitors. In addition, the use of BRAF inhibitors may result in the development of secondary skin tumors, originating from a paradoxical activation of the MAPK pathway in cells without a BRAF mutation.Citation4 In recent years, clinical benefits of combining a BRAF inhibitor with a mitogen-activated extracellular signal-regulated kinase (MEK) inhibitor were initially shown in several randomized trials, which found that it can address not only the limitations of single-agent BRAF inhibitors, but also improve the PFS and OS than with BRAF inhibitors alone.Citation5,Citation6
Although combined use of BRAF and MEK inhibitors is well tolerated by many patients, it is not devoid of side effects. Several clinical trials reported that diarrhea, anorexia, nausea, and vomiting are common adverse events frequently associated with the use of a combination of BRAF and MEK inhibitors in daily clinical practice, thus requiring early and appropriate managements to avoid unnecessary dose reductions and transitory or definitive treatment discontinuations.Citation7 Therefore, there is a need to master the characteristic features, incidence, and relative risk (RR) of significant adverse events to take adequate prevention and intervention as early as possible.Citation8 Hence, in this study, we have performed a meta-analysis to assess the therapeutic efficacy and safety of combined BRAF and MEK inhibition in patients with malignant melanoma and to provide treatment recommendations for these symptoms.
Materials and methods
Search strategy
The PubMed, EMBASE, and Cochrane Library electronic databases were searched for articles published from January 2000 to January 2017 using keywords “BRAF inhibition”, “MEK inhibition”, “Dabrafenib”, “Trametinib”, “Vemurafenib”, “Cobimetinib”, “Melanoma”, “Val600”, and “BRAF-mutant”. Additionally, the abstracts presented at major meetings of the American Society of Clinical Oncology (ASCO), the European Society for Medical Oncology, and the World Lung Cancer Conference were manually searched. Finally, full publications (not abstracts) from the Web of Science database were also searched to ensure that there were no additional studies.
Study selection
Studies that met the following criteria were included in the study: 1) studies of prospective Phase II and Phase III clinical trials and expanded-access (ie, outside clinical trials) programs, 2) studies that clinically investigated patients with melanoma or metastatic melanoma and those that studied the combinatorial effect of the participants assigned to BRAF and MEK inhibitors, and 3) studies that analyzed efficiency measures including overall response rate (ORR), PFS, and OS, as well as adverse events. The exclusion criteria were as follows: 1) investigations in patients of original studies that were unrelated to the study drug and 2) original studies that met the inclusion criterion (1) but required information such as overall response, PFS, and OS, as well as adverse events, etc, were not available.
Data extraction and quality assessment
We extracted information such as the first author, number of patients enrolled in the study, treatment information and characteristics of the participants to understand the baseline of all the included studies. To determine the validity of the selected studies, a modified Jadad scale was used to assess the quality of the included randomized studies. The scores of high-quality studies ranged from 4 to 8, whereas that of low-quality studies from 0 to 3. For non-randomized studies, the Newcastle-Ottawa Quality Assessment Scale was used. Each study was graded as either of low quality (0–5) or high quality (6–9). Any disagreements were resolved by discussion among our investigators or by referring to the original publication.
Definition of main outcomes
Complete response was defined as disappearance of all symptoms and signs of all measurable disease for at least 4 weeks and no appearance of any new lesions during that time. Partial response was defined as a 50% reduction in the sum of the products of the perpendicular diameters of all measurable lesions for at least 4 weeks and no new lesions or no enlargement in existing lesions during that time. Overall response included complete response and partial response. PFS was defined as one type of measurement that can be used in a clinical study or trial to help determine whether a new treatment is effective or not. It refers to the probability that a patient will remain alive, without the disease getting worse. We defined the OS as the time from the date of randomization to the date of death from any cause, or the date of last follow-up of a living patient. As for the safety outcomes, we referred to the trial authors’ definitions. We collected data about five frequent toxicity events (pyrexia, nausea, diarrhea, vomiting, and arthralgia) to enter into the meta-analysis.
Statistical analysis
Meta-analyses were carried out using software Review Manager Version 5.3 and R 2.13.2 Meta package. The pooled RR (risk ratio) and HR (hazard ratio) estimate and 95% confidence intervals (CIs), stratified by study setting and gender, were conducted using a random- or fixed-effect model, with between-study heterogeneity assessed using I2 statistic. If I2 was ≥50%, then the random-effect model was used, and if not, then the fixed-effect model was used. Statistical significance was defined as a P-value <0.05. All 95% CIs were two-sided. Finally, the publication bias were quantitatively tested by conducting Egger’s test.
Results
Search results
Study selection
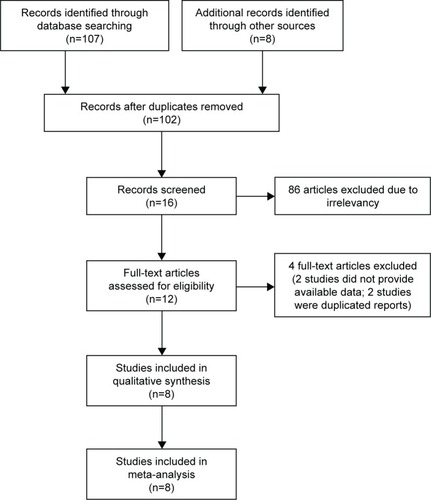
Of the studies initially identified, we excluded reports that did not meet the inclusion criteria after first screening the study titles and abstracts. Finally, eight studiesCitation9–Citation16 were ultimately included in the meta-analysis. illustrates how the eight studies were obtained from the literature search. The eight selected studies (including six randomized controlled trials), comprising a total of 2,664 participants, were published between 2012 and 2017. In all studies (including three Phase I/II studiesCitation10,Citation12 and five Phase III studiesCitation9,Citation11,Citation13,Citation14), the starting dose and schedule of vemurafenib (dabrafenib) and cobimetinib (trametinib) was based on US FDA guidelines (960 mg vemurafenib, bid; 60 or 80 mg cobimetinib, qd; 150 mg dabrafenib, bid; 2 mg trametinib qd). The main characteristics and qualities of included trials are listed in . Jadad scores of each of the eight studies included in the meta-analysis are also listed in ; the mean score was 4.25 (range, 3–6), which indicates that the overall quality of the study was fair.
Table 1 Summary of the characteristics of the studies included in the meta-analysis
Effectiveness
In our meta-analysis, four comparisons were used to analyze ORR and six comparisons to analyze PFS and OS.
Overall response rate
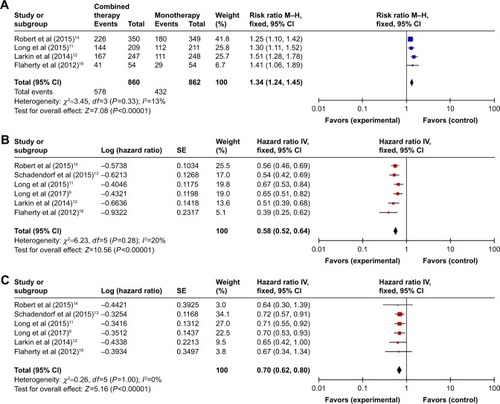
For the overall response, testing for interstudy heterogeneity gave significant results (ChiCitation2 =3.45, df=3 [P=0.33], I2=13%); therefore, RR and 95% CI were calculated by a fixed-effect model. Our fixed-effect model analysis revealed that combination therapy significantly improved the ORR in comparison to monotherapy (RR: 1.34 [95% CI: 1.24–1.45], P<0.00001) as shown in .
Figure 2 Forest plot analysis of the efficiency outcomes of combined BRAF and MEK inhibition versus BRAF inhibition alone. (A) Overall response rate, (B) progression-free survival, and (C) overall survival.
Progression-free survival
For the progression-free survival, testing for interstudy heterogeneity gave significant results (P=0.28, I2=20%), so we used a fixed-effect model for meta-analysis. Our fixed-effects model analysis indicated that PFS in combination therapy was significantly longer than that in monotherapy (HR: 0.58 [95% CI: 0.52–0.64], P<0.00001; ).
Overall survival
Information about HRs for OS was available from six trials,Citation9,Citation12–Citation14,Citation16 which reported that combination therapy showed an improved OS over monotherapy. The pooled HR obtained in our study demonstrated that combination therapy was associated with a significant enhancement of OS compared to monotherapy (HR: 0.70 [95% CI: 0.62–0.80], P<0.00001; heterogeneity: ChiCitation2 =0.26, df=5 [P<0.00001], I2=0%) ().
Safety
RR of all-grade adverse events
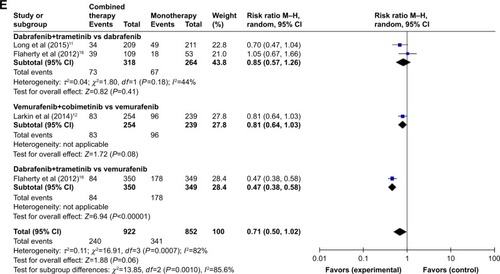
The pooled RRs of developing all-grade adverse events (pyrexia, nausea, diarrhea, vomiting, and arthralgia) with combination therapy versus controls were 2.00 (95% CI: 1.40–2.84), 1.41 (95% CI: 1.03–1.94), 1.50 (95% CI: 1.08–2.06), 1.87 (95% CI: 1.52–2.31), and 0.71 (95% CI: 0.50–1.02), respectively (). The results showed a statistically significant increased risk of developing pyrexia, nausea, vomiting, and arthralgia (all P<0.05) associated with combination therapy compared with monotherapy.
Table 2 RR of adverse events between combined targeted therapy and monotherapy
Of the six RCTsCitation9,Citation11–Citation14,Citation16 included in the RR analyses, four trialsCitation9,Citation11,Citation13,Citation16 examined the combination of dabrafenib and trametinib versus dabrafenib alone, one trialCitation12 the combination of vemurafenib and cobimetinib versus vemurafenib alone, and one trial the combination of dabrafenib and trametinib versus vemurafenib alone. Subgroup analysis of the RR of all-grade adverse events for combined BRAF and MEK inhibition versus BRAF inhibition alone showed different RRs for pyrexia: combination of dabrafenib and trametinib versus dabrafenib alone (Z=6.70; P<0.00001, RR =2.22, 95% CI: 1.76–2.81); combination of vemurafenib and cobimetinib versus vemurafenib alone (Z=0.98; P=0.32, RR =1.17, 95% CI: 0.85–1.61); combination of dabrafenib and trametinib versus vemurafenib alone (Z=7.96; P<0.00001, RR =2.51, 95% CI: 2.00–3.15) ().
Figure 3 Subgroup analysis of the relative risk (RR) of all-grade adverse events for combined BRAF and MEK inhibition versus BRAF inhibition alone. (A) Pyrexia, (B) nausea, (C) diarrhea, (D) vomiting, and (E) arthralgia.
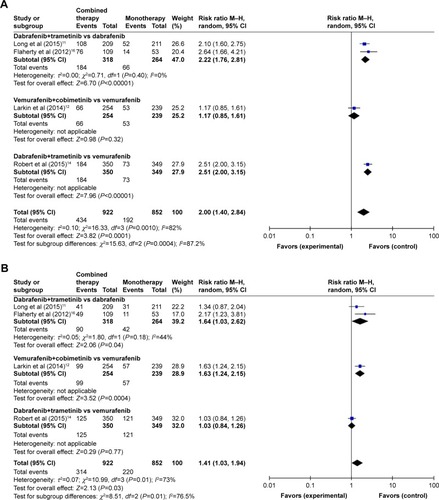
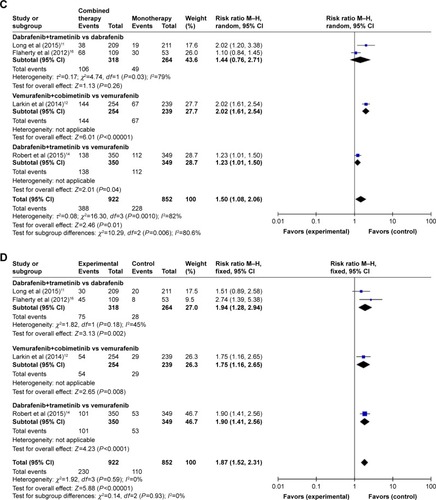
Regarding the RR of nausea events, combination of dabrafenib and trametinib versus dabrafenib (Z=2.06; P=0.04, RR =1.64, 95% CI: 1.03–2.62) and combination of vemurafenib and cobimetinib versus vemurafenib (Z=3.52; P=0.004, RR =1.63, 95% CI: 1.24–2.15) showed a significant difference, while combination of dabrafenib and trametinib versus vemurafenib showed no significant difference (Z=0.29; P=0.77, RR =1.03, 95% CI: 0.84–1.26) (). Regarding the RR of diarrhea, combination of vemurafenib and cobimetinib versus vemurafenib alone and combination of dabrafenib and trametinib versus vemurafenib alone showed significant results (Z=6.01; P<0.00001, RR =2.02, 95% CI: 1.61–2.54; Z=2.01; P=0.04, RR =1.23, 95% CI: 1.01–1.50), whereas combination of dabrafenib and trametinib versus dabrafenib alone showed no significant results (Z=1.13; P=0.26, RR =1.44, 95% CI: 0.76–2.71) ().
Regarding vomiting, significant differences were found between combination of dabrafenib and trametinib versus dabrafenib, combination of vemurafenib and cobimetinib versus vemurafenib, and combination of dabrafenib with trametinib versus vemurafenib (Z=3.13; P=0.002, RR =1.94, 95% CI: 1.28–2.94; Z=2.65; P=0.008, RR =1.75, 95% CI: 1.16–2.25; Z=4.23; P<0.0001, RR =1.90, 95% CI: 1.41–2.56, respectively) ().
Regarding RR of arthralgia, combination of dabrafenib and trametinib versus vemurafenib showed a significant difference (Z=6.94; P<0.0001, RR =0.47, 95% CI: 0.55–1.00). No significant differences were observed between combination of dabrafenib and trametinib versus dabrafenib and combination of vemurafenib and cobimetinib versus vemurafenib (Z=0.82; P=0.41, RR =0.85, 95% CI: 0.57–1.26; Z=1.72; P=0.08, RR =0.81, 95% CI: 0.64–1.03) ().
Incidence of all-grade adverse events associated with combined BRAF and MEK inhibition
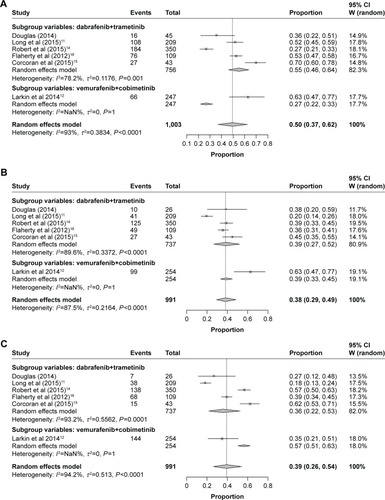
Results of the random-effect model showed that the pooled incidence of all-grade adverse events associated with combined BRAF and MEK inhibition of all the included trials was 50% (95% CI: 37%–62%), 38% (95% CI: 29%–49%), 39% (95% CI: 26%–54%), 29% (95% CI: 21%–39%), and 24% (95% CI: 18%–32%), respectively ().
Table 3 Overall incidence of all-grade adverse events in patients receiving combined targeted therapy
As meta-analysis results about adverse events revealed heterogeneity, we further performed subgroup analysis according to the treatment regimens. All the studies were classified into two subgroups: 1) combined dabrafenib and trametinib and 2) combined vemurafenib and cobimetinib. By subgroup analysis of the incidences of combined BRAF and MEK inhibition-associated all-grade adverse events stratified by treatment regimen, we found higher overall incidences of all-grade pyrexia were observed in patients on combined dabrafenib and trametinib (55%, 95% CI: 46%–64%) and patients on combined vemurafenib and cobimetinib (27%, 95% CI: 22%–33%) ().
Figure 4 Subgroup analysis of the incidence of all-grade adverse events for combined BRAF and MEK inhibition. (A) Pyrexia, (B) nausea, (C) diarrhea, (D) vomiting, and (E) arthralgia.
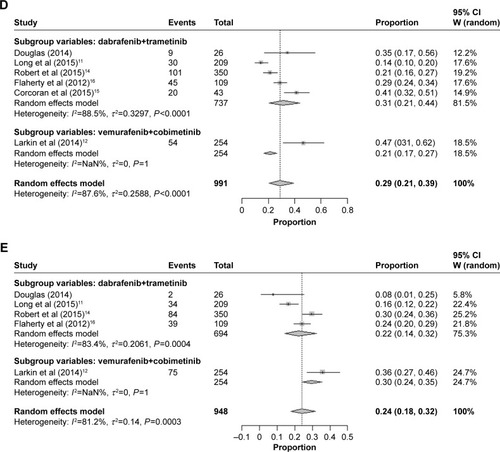
Different overall incidences of all-grade nausea were observed in patients treated with combination of dabrafenib and trametinib (39%, 95% CI: 27%–52%) and combination of vemurafenib and cobimetinib (39%, 95% CI: 33%–45%) (). Furthermore, higher incidences of all-grade diarrhea were observed in patients treated with combination of vemurafenib and cobimetinib (57%, 95% CI: 51%–63%) followed by those treated with combination of dabrafenib and trametinib (36%, 95% CI: 22%–53%) ().
The incidence of all-grade vomiting events was more higher in patients treated with combination of dabrafenib and trametinib (31%, 95% CI: 21%–41%) followed by combination of vemurafenib and cobimetinib (21%, 95% CI: 17%–27%) (). Finally, the overall incidences of all-grade arthralgia were more higher in patients treated with combination of vemurafenib and cobimetinib (30%, 95% CI: 24%–35%) followed by combination of dabrafenib and trametinib (22%, 95% CI: 14%–32%) ().
Publication bias
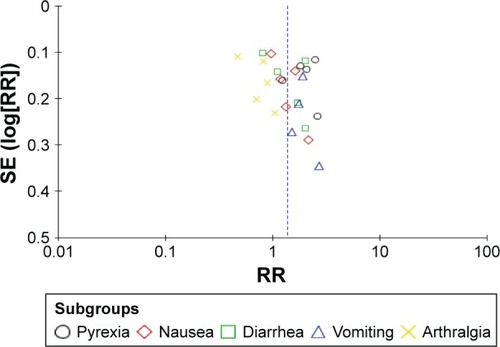
Publication bias was assessed using funnel plot produced by Review Manager 5.3 software. There was an obvious asymmetry of the funnel plot (), which suggests that there was some level of publication bias. However, because the number of included studies was only eight, the funnel plots may not be significant. Egger’s test revealed that publication bias was not significant for both the incidence and RR of adverse events (incidence: P=0.29; RR: P=0.51).
Discussion
For metastatic melanoma patients with activating BRAF mutations, the treatment standard is BRAF-targeted therapy, which is based on improvement in the rate of survival, compared with conventional chemotherapy.Citation17 However, clinical evidence shows that resistance occurs in 6–7 months after the initiation of treatment. Several mechanisms of MAPK-dependent resistance to BRAF inhibitors have been described in vitro and corroborated in tumor specimens obtained from patients. It was hypothesized that inhibition of the MAPK pathway downstream of BRAF could suppress mechanisms of resistance.Citation18 In recent years, lots of clinical data suggest that combined BRAF and MEK inhibition can block paradoxical activation of the MAPK pathway induced by single-agent BRAF inhibitors, and an opportunity to investigate a regimen combining a BRAF inhibitor with an MEK inhibitor was provided by the fact of MEK inhibition having been validated as a therapeutic approach in the same patient population.Citation19 However, it has been found that sequential use of an MEK inhibitor after progression on a BRAF inhibitor does not result in clinically meaningful antitumor activity either in vitro or in patients. Concurrent administration of MEK and BRAF inhibitors is effective on cell lines with acquired resistance to BRAF inhibitors and can achieve responses in patients with BRAF V600-mutant melanoma progressing on single-agent BRAF inhibitors.Citation20 More importantly, many studies have confirmed that for patients who have not previously received a BRAF inhibitor, treatment with a combination of a BRAF inhibitor and an MEK inhibitor could lead to greater initial antitumor activity and could prevent or delay MAPK-driven acquired resistance mechanisms.Citation21 Consistent with this, the results of our meta-analysis also demonstrated that combined BRAF and MEK inhibition provided significant advantage in ORR, PFS, and OS over MEK inhibition alone. However, the reported success of these agents comes at the cost of a set of adverse events, which significantly can affect therapeutic effect and quality of life of patients, and can lead to infection, discomfort, and bring some mental burden for patients to a certain degree.Citation22 Thus, a comprehensive analysis of all these reported adverse events has been conducted in our study.
The results of our meta-analysis showed that the group treated with combination of BRAF and MEK inhibitors exhibited superior results compared to that treated with BRAF alone: ORR – combined RR =1.34, 95% CI: 1.24–1.45, P<0.00001; PFS – combined HR =0.58, 95% CI: 0.52–0.64, P<0.00001); and OS rate – combined HR =0.70, 95% CI: 0.62–0.80, P<0.00001. Despite these advantages, combined therapy with BRAF and MEK inhibitors revealed some additional toxicities. To our knowledge, this is the first meta-analysis focusing specifically on the adverse events associated with the combination therapy. Our meta-analysis was able to demonstrate that combination therapy was associated with a significant increase in the risk of adverse events such as pyrexia, nausea, diarrhea, and vomiting in patients with melanoma. The most frequent treatment-related all-grade adverse event was pyrexia, while diarrhea was the less common.Citation23 Based on our further subgroup analysis, it was not difficult to find that combined vemurafenib and cobimetinib therapy may pose a higher risk of developing nausea and diarrhea compared to the combined dabrafenib and trametinib therapy.Citation24 Moreover, subgroup analysis also showed that combined vemurafenib and cobimetinib therapy may carry lower risks of developing pyrexia and vomiting than the combined dabrafenib and trametinib therapy.
Based on our results, we also found that patients receiving combination therapy had more frequent gastrointestinal toxic effects (eg, nausea, vomiting, and diarrhea) in comparison with patients receiving BRAF inhibitor alone.Citation25 We believe that it is very import to use nonpharmacological and pharmacological management to adjust treatment and therapeutic regimens according to the severity and types of gastrointestinal events, because pain and discomfort of patients can be alleviated by adequate prevention and treatment.Citation26 Firstly, it is advised that patients should realize the importance of managing these adverse events at the early treatment phase and strictly avoiding combined BRAF and MEK inhibition discontinuation. Secondly, the health care team of cancer patients should be informed to be ready for nutritional advice so to avoid hyponatremia or hypokalemia when gastrointestinal events occur.Citation27 Thirdly, gastrointestinal protective agents such as loperamide, omeprazole, and ondansetron should be well prepared before patients receive combination therapy; these agents should be immediately started at the onset of gastrointestinal symptoms.Citation28
Our meta-analysis had several limitations. Firstly, only eight studies met our inclusion criteria, and hence the small number of trials and low quality of most of the work could make the conclusion less convincing; also publication bias could not be completely excluded based on funnel plot.Citation29 Secondly, because most researchers applied their personal experience in diagnosing the toxicities in the clinical trials, there were different judgements based on the same signs. Thirdly, the treatment regimens and doses of drugs are different among the studies included in the meta-analysis, which led to significant heterogeneity of the data. Therefore, large-scale and well-designed studies are needed to summarize and analyze the data to draw a more convincing conclusion.Citation30
Conclusion
In summary, this study shows that combined therapy of BRAF and MEK inhibitors may moderately improve the overall response, PFS, and OS, although it may increase the incidence of some adverse events. In addition, prompt and effective management of these adverse events might allow for the safer use of combination therapy. We believe that our results could provide a reference point for physicians in clinical practice and ensure the safety and efficacy of treatment regimen for melanoma patients.
Disclosure
The authors report no conflicts of interest in this work.
References
- MartinliberalJLarkinJVemurafenib for the treatment of BRAF mutant metastatic melanomaFuture Oncol201511457958925686114
- ShiHHugoWKongXAcquired resistance and clonal evolution in melanoma during BRAF inhibitor therapyCancer Discov201441809324265155
- ChapmanPBHauschildARobertCImproved survival with vemurafenib in melanoma with BRAF V600E mutationN Engl J Med2011364262507251621639808
- AranceAMBerrocalALopez-MartinJASafety of vemurafenib in patients with BRAF, V600, mutated metastatic melanoma: the Spanish experienceClin Transl Oncol20161811114726983408
- LiuLMayesPAEastmanSThe BRAF and MEK inhibitors dabrafenib and trametinib: effects on immune function and in combination with immunomodulatory antibodies targeting PD-1, PD-L1, and CTLA-4Clin Cancer Res20152171639165125589619
- PuzanovICombining targeted and immunotherapy: BRAF inhibitor dabrafenib (D) ± the MEK inhibitor trametinib (T) in combination with ipilimumab (Ipi) for V600E/K mutation-positive unresectable or metastatic melanoma (MM)J Transl Med20151311225591711
- CebolleroAPuértolasTPajaresIComparative safety of BRAF and MEK inhibitors (vemurafenib, dabrafenib and trametinib) in first-line therapy for BRAF-mutated metastatic melanomaMol Clin Oncol20165445846227699043
- QueiroloPPicassoVSpagnoloFCombined BRAF and MEK inhibition for the treatment of BRAF-mutated metastatic melanomaCancer Treat Rev201541651952625944484
- LongGVFlahertyKTStroyakovskiyDDabrafenib plus trametinib versus dabrafenib monotherapy in patients with metastatic BRAF V600E/K-mutant melanoma: long-term survival and safety analysis of a phase 3 studyAnn Oncol20172871631163928475671
- JohnsonDBFlahertyKTWeberJSCombined BRAF (dabrafenib) and MEK inhibition (trametinib) in patients with BRAFV600-mutant melanoma experiencing progression with single-agent BRAF inhibitorJ Clin Oncol201432333697370425287827
- LongGVStroyakovskiyDGogasHDabrafenib and trametinib versus dabrafenib and placebo for Val600 BRAF-mutant melanoma: a multicentre, double-blind, phase 3 randomised controlled trialLancet2015386999244445126037941
- LarkinJAsciertoPADrenoBCombined vemurafenib and cobimetinib in BRAF-mutated melanomaN Engl J Med2014371201867187625265494
- SchadendorfDAmonkarMMStroyakovskiyDHealth-related quality of life impact in a randomised phase III study of the combination of dabrafenib and trametinib versus dabrafenib monotherapy in patients with BRAF V600 metastatic melanomaEur J Cancer201551783384025794603
- RobertCKaraszewskaBSchachterJImproved overall survival in melanoma with combined dabrafenib and trametinibN Engl J Med20153721303925399551
- CorcoranRBAtreyaCEFalchookGSCombined BRAF and MEK inhibition with dabrafenib and trametinib in BRAF V600-mutant colorectal cancerJ Clin Oncol201533344023403126392102
- FlahertyKTInfanteJRDaudACombined BRAF and MEK inhibition in melanoma with BRAF V600 MutationsN Engl J Med2012367181694170323020132
- PatelBGAhmedKAJohnstonePAInitial experience with combined BRAF and MEK inhibition with stereotactic radiosurgery for BRAF mutant melanoma brain metastasesMelanoma Res201626438238826926151
- SchmittRJKreidlerSMGlueckDHCorrelation between early FDG PET/CT response to BRAF and MEK inhibition and survival in patients with BRAF-mutant metastatic melanomaNucl Med Commun201637212212826440571
- KimACohenMSThe discovery of vemurafenib for the treatment of BRAF-mutated metastatic melanomaExpert Opin Drug Discov201611990791627327499
- DossettLAKudchadkarRRZagerJSBRAF and MEK inhibition in melanomaExpert Opin Drug Saf201514455957025648338
- RichmanJMartinliberalJDiemSBRAF and MEK inhibition for the treatment of advanced BRAF mutant melanomaExpert Opin Pharmacother20151691285129726001180
- WelshSJCorriePGManagement of BRAF and MEK inhibitor toxicities in patients with metastatic melanomaTher Adv in Med Oncol20157212213625755684
- PintoRStrippoliSSummaSDMicroRNA expression in BRAF-mutated and wild-type metastatic melanoma and its correlation with response duration to BRAF inhibitorsExpert Opin Ther Targets20151981027103526156293
- JohnsonASCrandallHDahlmanKPreliminary results from a prospective trial of preoperative combined BRAF, and MEK-targeted therapy in advanced BRAF, mutation-positive melanomaJ Am Coll Surg2015220458159325797743
- GallikerNAMurerCKamarashevJClinical observation of panniculitis in two patients with BRAF-mutated metastatic melanoma treated with a combination of a BRAF inhibitor and a MEK inhibitorEur J Dermatol201525217718025788221
- KhojaLHoggDDabrafenib in the treatment of metastatic or unresectable melanomaExpert Rev Anticancer Ther201515326527625711514
- DhillonSDabrafenib plus trametinib: a review in advanced melanoma with a BRAF, V600, mutationTarget Oncol201611341742827246822
- TietzeMJKHepptMGrafSAAktuelle und neue Standards in der therapieInfo Onkologie20151884046
- HermsFKramkimelNRegnierrosencherEAge and clear eyes are associated with an increased risk of cutaneous squamous cell carcinomas in vemurafenib-treated melanoma patientsMelanoma Res201626548749127261949
- SchmidSSianoMJoergerMResponse to dabrafenib after progression on vemurafenib in a patient with advanced BRAF V600E-mutant bronchial adenocarcinomaLung Cancer2015871858725466451