Abstract
Background
Pregnane X receptor (PXR), which is a member of the nuclear receptor protein family (nuclear receptor subfamily 1 group I member 2 [NR 1I2]), mediates the drug-resistance in the hepatocellular carcinoma (HCC) via enhancing the expression of drug-resistance-related genes which accelerate the clearance of antitumor drugs, eg, sorafenib. However, there are few reports on miRNA targeting PXR participating in the epigenetic regulation of PXR in HCC cells.
Materials and methods
TargetScan 7.2, an online method, was used to predict the miRNAs potentially targeting PXR. The expression of PXR and PXR downstream genes was detected by quantitative real-time PCR (qPCR) and Western blot. The clearance of sorafenib in HCC cells was monitored by liquid chromatograph-mass spectrometer/mass spectrometer (LC-MS/MS). The effects of miRNA on sorafenib’s efficacy were examined by in vitro methods, eg, MTT, and in vivo methods, eg, subcutaneous or intrahepatic tumor model.
Results
By virtual screening, we identified that miR-140-3p possibly targets PXR and then confirmed that the overexpression of miR-140-3p via lentiviral particles inhibited the expression of PXR in HCC cells. The downregulation of PXR’s expression by miR-140-3p led to the reduction of PXR downstream genes’ expression, which finally resulted in the decelerating clearance of sorafenib in HCC cells and enhanced the sensitivity of HCC cells to sorafenib. The effect of miR-140-3p could not modulate the expression of mutated PXR and the effect of miR-140-3p could also be inhibited by miR-140-3p’s inhibitor. Moreover, miR-140-3p enhanced the anti-tumor effect of sorafenib in both the subcutaneous and intrahepatic HCC tumor models.
Conclusion
Our study suggests that targeting PXR by miR-140-3p is a promising strategy for enhancing sorafenib’s efficacy during HCC treatment.
Introduction
Pregnane X receptor (PXR) is a member of the nuclear receptors (NRs) protein family (classified as NR1I2).Citation1,Citation2 In mammal cells, PXR could function as a transcription factor that could be activated by endogenous or exogenous ligands, in turn leading to the transcription of downstream genes, eg, CYP/CYP450 (CYP450) or ATP-binding cassette (ABC), which often participate in drug-resistance or drug-metaboloisms.Citation3,Citation4 Previously, PXR was considered as one of the key regulators mediating the expression of phase I, II or III drug metabolism enzymes (DMEs) to accelerate the clearance of exogenous drugs to protect the human body and liver organ from drug-induced injuries.Citation5–Citation7 Recently, PXR has also been considered to play an important role in the resistance of antitumor drugs.Citation8,Citation9 Our recent study indicated that sorafenib, an oral molecular-targeted antitumor agent, enhances its own clearance in hepatocellular carcinoma (HCC) cells via enhancing the activation of PXR during treatment.Citation11 Although current researches are focusing on PXR as a potential target to overcome drug-resistance of HCC treatment, the approaches targeting PXR have been poorly developed.
MicroRNAs (miRNAs or miRs) are a series of RNAs which repress the targeted messenger RNAs (mRNAs) via recognizing and interacting their 3′-untranslated regions (3′-UTR), and miRs have critical roles in regulating proliferation, metastasis or apoptosis of human cancer cells.Citation12,Citation13 Many miRs have been applied for regulating cancers’ progress via targeting cancer-related signaling pathways.Citation14,Citation15 In the present work, we identified miRNAs potentially targeting PXR. By screening a series of miRNAs via online tools, PXR would be a target of miR-140-3p. Transfection of miR-140-3p decreased the expression of PXR or its downstream genes and enhanced the sensitivity of HCC cells to sorafenib.
Materials and methods
Plasmids and reagents
The expression vector of PXR with a mutation of miR-140-3p target sequences in 3′-UTR was constructed and purchased from Vigene Corporation, Jinan City, Shandong Province, People’s Republic of China. For cell-based experiments, the mimic miR-140-3p (Cat. no 4464070) and miR-140-3p inhibitor (Cat. no 4464088) was purchased from Thermo Fisher Scientific (Waltham, MA, USA). MHCC97-H (a highly metastatic cell line of HCC) and HepG2 (a cell line of HCC) cell lines were purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, People’s Republic of China), the culture collection center of the Chinese government. Cells were preserved in our laboratory; maintaining conditions are described in our previous work.Citation16,Citation17 Control miRNA (random molecular agent), was a gift from Dr Mingyang Li in the General Hospital of Chinese PLA. For animal experiments, a vector with miR-140-3p was constructed and purchased from Vigene Corporation. Sequences of miR-140-3p were prepared into lentivirus particles. The antitumor agent sorafenib (Cat. no S7397) was purchased from Selleck Corporation, Houston, TX, USA. For cell culture experiments, sorafenib were carefully dissolved in dimethyl sulfoxide (DMSO) and the concentrations of sorafenib in DMSO were 10, 3, 1, 0.3, 0.1, 0.03 or 0.01 mmol/L. For subcutaneous tumor experiments, sorafenib (4 mg) was dissolved by a mixture of DMSO (15 µL), PEG400 (60 µL) and Tween80 (40 µL). Then, the solution was carefully added with physiological saline to a total volume 2 mL. This sorafenib-containing solution was named as Sor-Sol. Rifampicin (Cat. no HY-B0272), which is a typical agonist of PXR, was purchased from MedChem Express (MCE) (Monmouth Junction, NJ, USA). Rifampicin was dissolved in DMSO and the indicated concentrations of rifampicin in DMSO were 10, 3, 1, 0.3, 0.1, 0.03 or 0.01 mmol/L.
Luciferase reporter gene assay
MHCC97-H or HepG2 cells, which were transfected with control miRNA or miR-140-3p, were seeded in 24-well plates (Corning Incorporated, Corning, NY, USA), and were co-transfected with luciferase reporters (DR3-Luc, ER6-Luc, XREM-Luc or PXRE-Luc).Citation18 After being cultured for 24 hours, cells were harvested and analyzed for luciferase activities, as described by Ma et al.Citation19 For luciferase experiments, the concentrations of rifampicin on HCC cells were 10, 3, 1, 0.3, 0.1, 0.03 or 0.01 µmol/L diluted from rifampicin solution (1:1,000).
RNA extraction and qPCR experiments
MHCC97-H or HepG2 cells were treated with the indicated concentrations of rifampicin (10, 3, 1, 0.3, 0.1, 0.03 or 0.01 µmol/L) for 24 hours. The cells were then harvested and the total RNA samples were extracted and qPCR experiments were performed following protocols provided by the manufacturer (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA, USA). The expression of drug-resistance-related genes within MHCC97-H cells was examined by qPCR with primers listed in .
Table 1 Primers used in qPCR experiments
Antibodies and Western blot
Antibodies against PXR (Cat. no ab85451), CYP3A4 (Cat. no ab155029), P-glycoprotein (Cat. no ab235954) and β-actin (Cat. no ab205) were obtained from Abcam (Cambridge, MA, USA). Cells, which were transfected with control miRNA, miR-140-3p mimic, PXRMut or miR-140-3p inhibitor, were harvested to be extracted for Western blot experiments, which were performed following the standard protocol of Western blot experiments.
Cell culture and proliferation analysis
HCC cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (Thermo Fisher Scientific, Waltham, MA, USA) with 0.5%–1% fetal bovine serum (FBS) (Thermo Fisher Scientific) in a sterile incubator maintained at 37°C under 5% CO2 condition. For drug antitumor, cells were seeded in 24-well plate (Corning Incorporated) and incubated by the indicated concentrations (10, 3, 1, 0.3, 0.1, 0.03 or 0.01 µmol/L) of sorafenib diluted by sorafenib DMSO solution (final DMSO concentration was 1‰). Then, cells were incubated by sorafenib for 48 hours and harvested for MTT experiments. The inhibition rates of sorafenib on HCC cells were calculated by the methods described by Feng et al.Citation20
Subcutaneous HCC tumor model
Animal experiments were permitted by the Institutional Animal Care and Use Committee of the 302nd Hospital, People’s Liberation Army of China. All animal studies were carried out in accordance with the UK Animals (Scientific Procedures) Act, 1986 and associated guidelines. For subcutaneous tumor experiments,Citation21,Citation22 nude mice (severe combined immune deficiency mice) aged 4–6 weeks were purchased from Si-Bei-Fu Biotechnology Corporation, Beijing, People’s Republic of China. MHCC97-H or HepG2 cells which were cultured in DMEM adding 10% FBS, were injected into nude mice (5×106 cells per inoculation point). After 2–4 weeks’ growth, the tumoral volume reached almost 1,000–1,200 mm3.
Clearance of sorafenib in HCC cells
To examine the clearance of sorafenib in HCC cells, MHCC97-H or HepG2 cells were transfected with miR-140-3p or PXR expression plasmids, and were treated with 1 µmol/L (the IC50 concentration of sorafenib on MHCC97-H cells) for 12 hours. For cultured MHCC97-H cells, cells were harvested after sorafenib treatment at the indicated time points and then harvested for liquid chromatograph-mass spectrometer/mass spectrometer (LC-MS/MS) examination.Citation23 For the subcutaneous tumor model, MHCC97-H or HepG2 cells which were infected with miR-140-3p or PXR, were seeded into nude mice to form subcutaneous tumors. Sor-Sol was injected into subcutaneous tumors (20 µL per tumor) in the subcutaneous tumors of the nude mice formed by HCC cells. Cells or tumor tissues were harvested and the sorafenib was extracted by acetonitrile (ACN) at the indicated time points. The amount of sorafenib in tumor tissues was measured by LC-MS/MS.Citation11,Citation23
In vivo tumor growth
MHCC97-H cells, which were transfected with plasmids, were injected into the nude mice to form subcutaneous tumors or into the liver organs via hepatic portal vein injection.Citation24 Five days after injection, the nude mice were orally administrated 2 mg/kg sorafenib treatment every 2 days. After 10 × treatments for 21 days, the mice were collected. For the subcutaneous tumor model, tumors were harvested and the tumor weights or volumes were examined following the methods described in the references.Citation21,Citation22 For the intrahepatic growth model, mice were harvested and the liver organs with nodules formed by MHCC97-H were collected for taking photographs. Photographs were quantitatively analyzed to determine the total amount of nodules by ImageJ software following the methods described by Xie et al.Citation24 The inhibition rate of each group was calculated as [control group relative nodule area (percentages of nodules to total area of liver organs, %) - administration group relative nodule area]/control group relative nodule area × 100%.
Statistical analysis
Statistical analysis was performed by Bonferroni’s correction with or without two-way ANOVA, SPSS Software (IBM Corporation, Armonk, NY, USA). The IC50 values half-life (t1/2 value) values were calculated by an Origin software (Version No 6.1, OriginLab Corporation, Northampton, MA, USA). A P-value of <0.05 was considered statistically significant.
Results
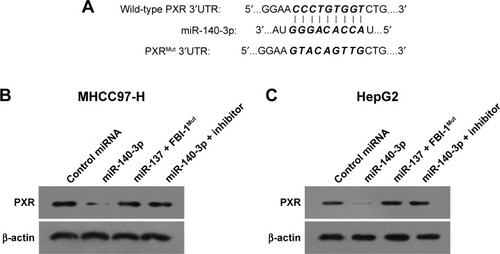
Transfection of miR-140-3p decreased PXR expression by targeting PXR mRNA’s 3′-UTR
In order to predict the miR potentially targeting PXR, online tools TargetScan and MiRanda database were used, and it was found that PXR was a potential target of miR-140-3p: the italicized font in indicates the miR-140-3p targeted sequence within PXR mRNA’s 3′-UTR (). Next, MHCC97-H cells and HepG2 cells were transfected with control miRNA, miR-140-3p mimic, miR-140-3p mimic+ PXRMut or miR-140-3p mimic+ inhibitor for 48 hours and harvested for Western blot. We found that compared with control miRNA, miR-140-3p significantly decreased the expression of PXR () but not PXRMut which had a mutation in the miR-140-3p-binding site (shown in ); whereas transfection of miR-140-3p’s inhibitor could block the effect of miR-140-3p on PXR’s expression in MHCC97-H cells () or HepG2 cells (). Therefore, miR-140-3p could repress the expression of PXR in HCC cells by directly targeting PXR mRNA’s 3′-UTR.
Figure 1 miR-140-3p targets PXR’s 3′-UTR.
Abbreviations: miR, microRNA; PXR, pregnane X receptor; Mut, mutation; 3′-UTR, 3′-untranslated regions.
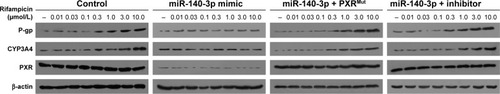
MiR-140-3p inhibits PXR pathway’s activation
To further identify the effect of miR-140-3p on PXR, luciferase experiments were performed. As shown in , PXR’s agonist rifampicin could induce the transcription factor activity of PXR, whereas miR-140-3p blocked the activity of PXR induced by rifampicin. Moreover, transfection of PXRMut or miR-140-3p’s inhibitor decreased the effect of miR-140-3p on PXR’s transcription factor activity. Accordingly, miR-140-3p inhibited the effect of rifampicin on the mRNA or protein expression of PXR downstream genes, CYP3A4 and MDR-1. Transfection of PXRMut or miR-140-3p’s inhibitor inhibited the effect of miR-140-3p ( and ). Therefore, miR-140-3p inhibits PXR pathway activation.
Table 2 The EC50 values of rifampicin inducing luciferase reporters’ activation in each group
Table 3 The EC50 values of rifampicin inducing the expression of PXR downstream gene CYP3A4 or MDR-1 in each group
Figure 2 Transfection of miR-140-3p inhibited the expression of PXR downstream genes.
Abbreviations: miR, microRNA; PXR, pregnane X receptor; CYP3A4, CYP/CYP450 3A4; P-gp, P-glycoprotein; Mut, mutation.
MiR-140-3p decelerates the clearance of sorafenib in HCC cells
Since PXR, CYP3A4 and MDR-1 are related to the clearance of sorafenib in HCC cells, we then aimed to study the correlation of miR-140-3p and the clearance of sorafenib in HCC cells. We performed sorafenib treatment on HCC cells in vitro or injected Sor-Sol in vivo in HCC tumors and detected the clearance of sorafenib under different conditions. As shown in , miR-140-3p decelerated the clearance of sorafenib in cultured HCC cells or subcutaneous HCC tumors, the half-life (t1/2 value) of sorafenib in cultured HCC cells correspondingly increased. PXRMut or miR-140-3p’s inhibitor inhibited the effect of miR-140-3p on the clearance of sorafenib. Therefore, our in vitro and in vivo data suggest that miR-140-3p decelerated the clearance of sorafenib in HCC cells.
Table 4 The clearance (half-life/t1/2 value) of Sor in HCC cells
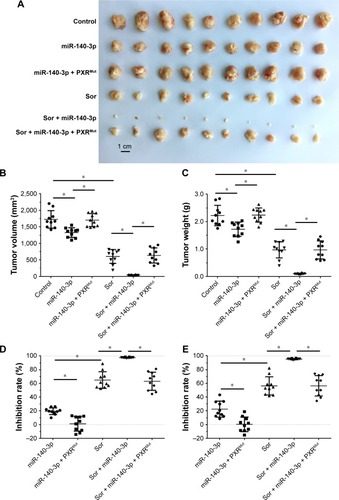
MiR-140-3p enhances the sensitivity of HCC cells to sorafenib
To determine whether miR-140-3p enhances the in vitro antitumor effect of sorafenib on HCC cells, MTT assays were performed. As shown in , miR-140-3p enhanced the sensitivity of MHCC97-H and HepG2 cells to sorafenib, and the IC50 value of sorafenib on MHCC97-H or Hepg2 cells were decreased.
Table 5 The antitumor effect (IC50 values) of Sor on HCC cells in each group
Next, the effect of miR-140-3p and sorafenib co-administration on subcutaneous HCC tumors was determined. As shown in , treatment of sorafenib could significantly inhibit the subcutaneous growth of HCC cells in nude mice. Infection of miR-140-3p could enhance the antitumor effect of sorafenib on MHCC97-H cells’ subcutaneous growth and PXRMut expressing cells/tumors could not be affected by miR-140-3p ( and ).
Table 6 The antitumor effect (inhibition rate) of Sor on HCC cells in each group
Figure 3 Transfection of miR-140-3p enhanced the antitumor effect of Sor on MHCC97-H cells’ subcutaneous growth.
Abbreviations: miR, microRNA; PXR, pregnane X receptor; Mut, mutation; Sor, sorafenib.
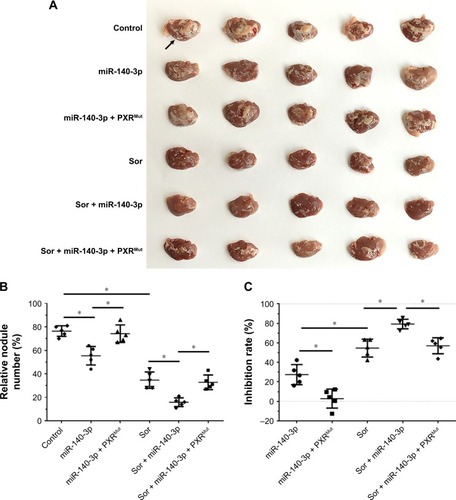
To mimic the in vivo growth of MHCC97-H cells in mouse liver, MHCC97-H cells were injected into nude mice via the hepatic portal vein to form tumor nodules on their livers (). We firstly established an oral sorafenib administration method for this intrahepatic HCC tumor model ( and ). Then, we performed miR-140-3p and sorafenib co-administration. As shown in , MHCC97-H cells could form multi-nodules in the livers via hepatic portal vein injection (). The results showed that treatment of sorafenib could significantly inhibit the intrahepatic growth of MHCC97-H cells in the livers of nude mice ( and ). Infection of miR-140-3p in MHCC97-H cells could enhance the antitumor effect of sorafenib on MHCC97-H cells’ intrahepatic growth ( and ), whereas transfection of PXRMut in MHCC97-H cells could almost block the effect of to miR-140-3p on sorafenib’s antitumor activation. Therefore, miR-140-3p could enhance the sensitivity of HCC cells to sorafenib by targeting PXR.
Figure 4 Transfection of miR-140-3p enhanced the antitumor effect of Sor on MHCC97-H cells’ intrahepatic growth.
Abbreviations: miR, microRNA; PXR, pregnane X receptor; Mut, mutation; Sor, sorafenib.
Discussion
Advanced HCC is one of the foremost cancers in People’s Republic of China nowadays.Citation25–Citation27 Due to the limitation of current diagnostic and therapeutic strategies, most patients could not be treated by the radical ablation.Citation28,Citation29 Due to the fact that advanced HCC is insensitive to chemotherapyCitation30 and radiotherapy,Citation31 the prognosis or clinical outcome of patients with advanced HCC is poor. Although some kinds of local ablation therapies, eg, radiofrequency ablation (RFA)Citation32 or transarterial chemoembolization (TACE),Citation33 could decelerate the progress of HCC to some extent, the rapid and aggressive recurrence of HCC after RFA or TACE treatment affect the application of these kinds of local ablation therapies. Therefore, molecular-targeted agents play a central role in advanced HCC treatment. Sorafenib is the only choice for advanced HCC treatment.Citation34–Citation36 Although the application of sorafenib enhances the survival of HCC patients, sorafenib-resistance has become one of the major obstacles in improving its efficacy.Citation37–Citation39 Thus, it is urgent to develop new strategies to overcome sorafenib-resistance.
Previous work has often focused on the antitumor drug-resistance by regulating multi-drug resistance genes, such as ABCs, eg, MDR-1/ABCB1 (ABC Subfamily B Member 1).Citation40–Citation49 However, the role of PXR, which is the transcription factor of ABCs and mediates the transcription of ABCs in antitumor agents-resistance, is not very clear. PXR expressed in the liver organ or intestine organ could mediate drug clearance in general; however, the tumor-specific expression of PXR could inhibit the therapeutic efficacy of anticancer agents by accelerating these drugs during treatment.Citation9–Citation11 As a central regulator of drug metabolism or clearance, PXR alters the absorption and clearance of xenobiotics (eg, antitumor drugs), leading to the reduced efficacy of the treatments.Citation9–Citation11 In our previous work, we showed that the endogenous PXR is a master regulator of sorafenib-resistance in HCC cells.Citation11 Sorafenib can bind to and activate PXR and facilitate the expression of PXR downstream genes’ expression.Citation11 By examining the clinical specimens, we found that a high level of PXR is correlated with poor prognosis of advanced HCC patients receiving sorafenib treatment. However, the regulatory mechanism of endogenous PXR mRNA level is not clear.
MiR is an important component of gene expression regulation.Citation50–Citation56 MiRs are important regulators of HCC proliferation and metastasis.Citation57–Citation63 In the present work, we firstly identified miR-140-3p as a potential miRNA target for PXR. Then, the regulatory effect of miR-140-3p on PXR was identified. Results showed that overexpression of miR-140-3p via lentiviral vector decreased the expression of PXR and PXR downstream genes in HCC cells; whereas overexpression of PXR with mutated miR-140-3p target sequence or transfection of miR-140-3p’s inhibitor blocked the effect of miR-140-3p on PXR’s expression. Moreover, miR-140-3p also enhanced the sensitivity of HCC cells to sorafenib. The effect of sorafenib on HCC cells were examined by in vitro methods or in vivo experiments. Our data illustrated that miR-140-3p administration was a promising strategy to enhance the antitumor effect of sorafenib on HCC cells. Interestingly, miR-140-3p could decelerate the clearance of sorafenib in cultured HCC cells or HCC subcutaneous tumor by decreased PXR’s expression. This result could extend our knowledge of miRs regulating sorafenib clearance and provide useful methods in metabolism-related drug-resistance researches.
Moreover, this work reported on miR-140-3p targeting PXR and such reports of miRs targeting PXR are rare. Beside PXR, researchers often focus on the miRs targeting ABCs.Citation64,Citation65 Awortwe et al and Rao et al reported that miR-655-3p or miR-148a could modulate the drug-resistance by targeting ABCs, eg, MRP3 or ABCG2.Citation65,Citation66 Following on from the paper by Sharma et al that concluded that miR-18a-5p is a negative regulator and research by Vachirayonstien et al that stated that miR-30c-1-3p could be a suppressor of PXR, our results extend the knowledge of miRs on the PXR signaling pathway.Citation67,Citation68
Conclusion
This work for the first time reported that PXR is a target of miR-140-3p. MiR-140-3p suppresses the PXR pathway and enhances the sensitivity of HCC cells to sorafenib. Downregulation of PXR by miR-140-3p may be a promising strategy for enhancing sorafenib-based HCC treatment.
Author contributions
All authors made substantial contributions to the design and conception; acquisition, analysis or interpretation of data. All authors took part in either drafting or revising the manuscript. At the same time, all authors gave final approval of the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Disclosure
The author reports no conflicts of interest in this work.
References
- GotohSMiyauchiYMooreRNegishiMGlucose elicits serine/threonine kinase VRK1 to phosphorylate nuclear pregnane X receptor as a novel hepatic gluconeogenic signalCell Signal20174020020928911860
- SeowCLLauAJDifferential activation of pregnane X receptor by carnosic acid, carnosol, ursolic acid, and rosmarinic acidPharmacol Res2017120233328288941
- KliewerSANuclear receptor PXR: discovery of a pharmaceutical anti-targetJ Clin Invest201512541388138925831443
- CoppleBLLiTPharmacology of bile acid receptors: Evolution of bile acids from simple detergents to complex signaling moleculesPharmacol Res201610492126706784
- WatkinsREWiselyGBMooreLBThe human nuclear xenobiotic receptor PXR: structural determinants of directed promiscuityScience200129255252329233311408620
- ChenTChenQXuYSRC-3 is required for CAR-regulated hepatocyte proliferation and drug metabolismJ Hepatol201256121021721827731
- BuchmanCDChaiSCChenTA current structural perspective on PXR and CAR in drug metabolismExpert Opin Drug Metab Toxicol201814663564729757018
- ThakurJKArthanariHYangFA nuclear receptor-like pathway regulating multidrug resistance in fungiNature2008452718760460918385733
- OladimejiPOChenTPXR: more than just a master xenobiotic receptorMol Pharmacol201893211912729113993
- MackowiakBHodgeJSternSWangHThe roles of xenobiotic receptors: beyond chemical dispositionDrug Metab Dispos20184691361137129759961
- FengFJiangQCaoSPregnane X receptor mediates sorafenib resistance in advanced hepatocellular carcinomaBiochim Biophys Acta20181862410171030
- HinesPJMicroRNAs in functional and dysfunctional painScience2017356634311341136
- LiLLiangYKangLTranscriptional regulation of the Warburg effect in cancer by SIX1Cancer Cell2018333368.e7385.e729455928
- JiQXuXLiLmiR-216a inhibits osteosarcoma cell proliferation, invasion and metastasis by targeting CDK14Cell Death Dis2017810e310329022909
- LiangYXuXWangTThe EGFR/miR-338-3p/EYA2 axis controls breast tumor growth and lung metastasisCell Death Dis201787e292828703807
- ChenYFengFGaoXMiRNA153 reduces effects of chemotherapeutic agents or small molecular kinase inhibitor in HCC cellsCurr Cancer Drug Targets201515317618725714700
- HouJHongZFengFA novel chemotherapeutic sensitivity-testing system based on collagen gel droplet embedded 3D-culture methods for hepatocellular carcinomaBMC Cancer201717172929117859
- ZhaoJBaiZFengFCross-talk between EPAS-1/HIF-2α and PXR signaling pathway regulates multi-drug resistance of stomach cancer cellInt J Biochem Cell Biol201672738826783937
- MaHYaoYWangCTranscription factor activity of estrogen receptor α activation upon nonylphenol or bisphenol A treatment enhances the in vitro proliferation, invasion, and migration of neuroblastoma cellsOnco Targets Ther201693451346327366082
- FengFLuYYZhangFLong interspersed nuclear element ORF-1 protein promotes proliferation and resistance to chemotherapy in hepatocellular carcinomaWorld J Gastroenterol20131971068107823466962
- JiaHYangQWangTRhamnetin induces sensitization of hepatocellular carcinoma cells to a small molecular kinase inhibitor or chemotherapeutic agentsBiochim Biophys Acta2016186071417143027091611
- AnLLiDDChuHXTerfenadine combined with epirubicin impedes the chemo-resistant human non-small cell lung cancer both in vitro and in vivo through EMT and Notch reversalPharmacol Res201712410511528754458
- WuMZhaoGZhuangXTriclosan treatment decreased the antitumor effect of sorafenib on hepatocellular carcinoma cellsOnco Targets Ther2018112945295429849464
- XieHTianSYuHA new apatinib microcrystal formulation enhances the effect of radiofrequency ablation treatment on hepatocellular carcinomaOnco Targets Ther2018113257326529910621
- KimGALimYSHanSHigh risk of hepatocellular carcinoma and death in patients with immune-tolerant-phase chronic hepatitis BGut201867594595229055908
- RajyaguruDJBorgertAJSmithALRadiofrequency ablation versus stereotactic body radiotherapy for localized hepatocellular carcinoma in nonsurgically managed patients: analysis of the National Cancer DatabaseJ Clin Oncol201836660060829328861
- GomaaAWakedIManagement of advanced hepatocellular carcinoma: review of current and potential therapiesHepatoma Res201736112122
- ZhangHLiXXYangYZhangYWangHYZhengXFSSignificance and mechanism of androgen receptor overexpression and androgen receptor/mechanistic target of rapamycin cross-talk in hepatocellular carcinomaHepatology20186762271228629220539
- CastAValanejadLWrightMC/EBPα-dependent preneoplastic tumor foci are the origin of hepatocellular carcinoma and aggressive pediatric liver cancerHepatology20186751857187129159818
- KimDWTalatiCKimRHepatocellular carcinoma (HCC): beyond sorafenib-chemotherapyJ Gastrointest Oncol20178225626528480065
- LiuRZhaoDZhangXA20 enhances the radiosensitivity of hepatocellular carcinoma cells to 60Co-γ ionizing radiationOncotarget2017854931039311629190981
- XieHYuHTianSMEIS-1 level in unresectable hepatocellular carcinoma can predict the post-treatment outcomes of radiofrequency ablationOncotarget2018920152521526529632641
- XieHYuHTianSWhat is the best combination treatment with transarterial chemoembolization of unresectable hepatocellular carcinoma? A systematic review and network meta-analysisOncotarget201785910050810052329245997
- RimCHYoonWSLeaflet manual of external beam radiation therapy for hepatocellular carcinoma: a review of the indications, evidences, and clinical trialsOnco Targets Ther2018112865287429844684
- RaufiATironaMTProspect of the use of checkpoint inhibitors in hepatocellular cancer treatmentsCancer Manag Res20179192728223846
- RoskoskiRThe role of small molecule platelet-derived growth factor receptor (PDGFR) inhibitors in the treatment of neoplastic disordersPharmacol Res2018129658329408302
- ZhuYJZhengBWangHYChenLNew knowledge of the mechanisms of sorafenib resistance in liver cancerActa Pharmacol Sin201738561462228344323
- ZhangYLiDJiangQNovel ADAM-17 inhibitor ZLDI-8 enhances the in vitro and in vivo chemotherapeutic effects of Sorafenib on hepatocellular carcinoma cellsCell Death Dis20189774329970890
- FengFJiangQJiaHWhich is the best combination of TACE and sorafenib for advanced hepatocellular carcinoma treatment? A systematic review and network meta-analysisPharmacol Res20181358910129959032
- LuXZhouDHouBDichloroacetate enhances the antitumor efficacy of chemotherapeutic agents via inhibiting autophagy in non-small-cell lung cancerCancer Manag Res2018101231124129844702
- TangLZhangCHeHAssociations between ABCG2 gene polymorphisms and gefitinib toxicity in non-small cell lung cancer: a meta-analysisOnco Targets Ther20181166567529440914
- FangYSunJZhongXES2 enhances the efficacy of chemotherapeutic agents in ABCB1-overexpressing cancer cells in vitro and in vivoPharmacol Res201812938839929122696
- PatelALiTWAnreddyNSuppression of ABCG2 mediated MDR in vitro and in vivo by a novel inhibitor of ABCG2 drug transportPharmacol Res201712118419328455266
- JinWLiaoXLvYMUC1 induces acquired chemoresistance by upregulating ABCB1 in EGFR-dependent mannerCell Death Dis201788e298028796259
- ShubbarMHPennyJIEffect of amyloid beta on ATP-binding cassette transporter expression and activity in porcine brain microvascular endothelial cellsBiochim Biophys Acta201818621023142322
- DartierJLemaitreEChourpaIATP-dependent activity and mitochondrial localization of drug efflux pumps in doxorubicin-resistant breast cancer cellsBiochim Biophys Acta201718615 Pt A10751084
- KimSWHasanuzzamanMDChoMRole of 14-3-3 sigma in over-expression of P-gp by rifampin and paclitaxel stimulation through interaction with PXRCell Signal20173112413428077325
- KhabouBDurand-SchneiderAMDelaunayJLComparison of in silico prediction and experimental assessment of ABCB4 variants identified in patients with biliary diseasesInt J Biochem Cell Biol20178910110928587926
- XiaoJEggerMEMcMastersKMHaoHDifferential expression of ABCB5 in BRAF inhibitor-resistant melanoma cell linesBMC Cancer201818167529929490
- RuhlRRanaSKelleyKmicroRNA-451a regulates colorectal cancer proliferation in response to radiationBMC Cancer201818151729720118
- GaoSZhaoZYWuRZhangYZhangZYPrognostic value of micro RNAs in colorectal cancer: a meta-analysisCancer Manag Res20181090792929750053
- ShahidSKimGJohnsonNRMicroRNAs from the parasitic plant Cuscuta campestris target host messenger RNAsNature20185537686828529300014
- HydbringPWangYFasslACell-cycle-targeting MicroRNAs as therapeutic tools against refractory cancersCancer Cell2017314576.e8590.e828399412
- CasselsLBardeYAScaling pain threshold with microRNAsScience201735663431124112528619901
- FernandesJVieiraASKramer-SoaresJCHippocampal microRNA-mRNA regulatory network is affected by physical exerciseBiochim Biophys Acta20181862817111720
- SunTLong noncoding RNAs act as regulators of autophagy in cancerPharmacol Res201812915115529133213
- WangRYuZChenFmiR-300 regulates the epithelial-mesenchymal transition and invasion of hepatocellular carcinoma by targeting the FAK/PI3K/AKT signaling pathwayBiomed Pharmacother20181031632164229864952
- ZhangXLuoPJingWZhouHLiangCTuJcircSMAD2 inhibits the epithelial-mesenchymal transition by targeting miR-629 in hepatocellular carcinomaOnco Targets Ther2018112853286329844683
- LuoCYinDZhanHmicroRNA-501-3p suppresses metastasis and progression of hepatocellular carcinoma through targeting LIN7ACell Death Dis20189553529749382
- ChenZZuoXZhangYMiR-3662 suppresses hepatocellular carcinoma growth through inhibition of HIF-1α-mediated Warburg effectCell Death Dis20189554929748591
- LiaoXZhuGHuangRIdentification of potential prognostic microRNA biomarkers for predicting survival in patients with hepatocellular carcinomaCancer Manag Res20181078780329713196
- VasuriFFittipaldiSde PaceVTissue miRNA 483-3p expression predicts tumor recurrence after surgical resection in histologically advanced hepatocellular carcinomasOncotarget2018925178951790529707155
- O’BrienKPKhanSGilliganKEEmploying mesenchymal stem cells to support tumor-targeted delivery of extracellular vesicle (EV)-encapsulated microRNA-379Oncogene201837162137214929367765
- BensonEAEadonMTDestaZRifampin regulation of drug transporters gene expression and the association of microRNAs in human hepatocytesFront Pharmacol2016711127199754
- AwortweCKaehlerMRosenkranzBCascorbiIBruckmuellerHMicroRNA-655-3p regulates Echinacea purpurea mediated activation of ABCG2Xenobiotica201848101050105828990842
- RaoZZZhangXWDingYLYangMYmiR-148a-mediated estrogen-induced cholestasis in intrahepatic cholestasis of pregnancy: Role of PXR/MRP3PLoS One2017126e017870228575098
- SharmaDTurkistaniAAChangWHuCXuZChangTKHNegative regulation of human pregnane X receptor by microRNA-18a-5p: evidence for suppression of microRNA-18a-5p expression by rifampin and rilpivirineMol Pharmacol2017921485628408657
- VachirayonstienTYanBMicroRNA-30c-1-3p is a silencer of the pregnane X receptor by targeting the 3′-untranslated region and alters the expression of its target gene cytochrome P450 3A4Biochim Biophys Acta2016185991238124427085140
