Abstract
A retrospective study was performed to evaluate the outcome of alendronate (ALN) treatment for seven years in postmenopausal Japanese women with osteoporosis. Forty-seven postmenopausal women with osteoporosis (mean age at baseline 65.7 years) treated with ALN for over seven years in our outpatient clinic were analyzed. Lumbar spine bone mineral density (BMD) was measured using dual energy X-ray absorptiometry, and urinary levels of cross-linked N-terminal telopeptides of type I collagen (NTX) and serum alkaline phosphatase (ALP) were monitored during the seven-year treatment period. Urinary NTX and serum ALP levels decreased (−48.2% at three months and −15.7% at seven years, respectively) and lumbar spine BMD increased (+12.8% at seven years) compared with baseline values. No serious adverse events were observed, including osteonecrosis of jaw, atypical femoral diaphysis fractures, or atrial fibrillation. To our knowledge, this is the first report of the outcome of ALN treatment for seven years in Japanese patients with osteoporosis. ALN successfully suppressed bone turnover and increased lumbar spine BMD from the baseline value over the course of the seven-year treatment period without causing any severe adverse events.
Background
Osteoporosis most commonly affects postmenopausal women, placing them at increased risk of fractures. Alendronate (ALN) is widely used for the treatment of postmenopausal osteoporosis. The Fracture Intervention Trial demonstrated the antifracture efficacy of ALN for vertebral, nonvertebral, hip, and wrist fractures in postmenopausal women with osteoporosis.Citation1,Citation2 A recent systematic review analyzing 11 randomized controlled trials including 12,068 women has confirmed both clinically important and statistically significant reductions in vertebral, nonvertebral, hip, and wrist fractures for secondary prevention of osteoporosis (gold standard evidence).Citation3 ALN is regarded as a first-line drug for the treatment of osteoporosis in Japan.
The long-term efficacy of antifracture drugs needs to be established. Bone et alCitation4 reported their experience using ALN to treat postmenopausal Western women with osteoporosis for a period of 10 years. Ten years of ALN treatment produced an increase in bone mineral density (BMD) of 13.7% at the lumbar spine, 10.3% at the trochanter, 5.4% at the femoral neck, and 6.7% at the total proximal femur, compared with baseline values. Safety data, including fractures and stature, did not suggest that prolonged treatment resulted in any loss of benefit. Thus, the therapeutic effects of ALN were sustained, and the drug was well tolerated over a 10-year period.
In Japan, results of randomized controlled trials testing the effects of short-term (1–3 years) ALN treatment on BMD, bone turnover, and fracture incidence have been reported in postmenopausal women with osteoporosis.Citation5–Citation8 ALN reduces bone turnover, increases lumbar and hip BMD, and prevents vertebral fractures. Recently, we reported that ALN successfully reduced bone turnover and maintained metacarpal BMD for five years in older postmenopausal Japanese women with osteoporosis or osteopenia and clinical risk factors for fractures.Citation9 To our knowledge, however, the outcome of long-term (more than five years) ALN treatment has not been previously reported in Japanese patients. Therefore, a retrospective study was performed to evaluate the outcome of ALN treatment for seven years in postmenopausal Japanese women with osteoporosis. The primary endpoint was lumbar spine BMD, and the secondary endpoint was biochemical markers. Adverse events such as osteonecrosis of the jaw (ONJ), atypical femoral diaphysis fractures, and atrial fibrillation,Citation10–Citation12 as well as incident osteoporotic fractures were also assessed.
Subjects and methods
Subjects
Forty-seven postmenopausal Japanese women (age range 52–83 years, mean age 65.7 years at the beginning of treatment) who had been treated with ALN for over seven years were recruited from the outpatient clinic at Keiyu Orthopaedic Hospital (Gunma, Japan) during the period between July 1 and December 30, 2009. Exclusion criteria were a history of reflux esophagitis, gastric or duodenal ulcer, gastrectomy, or bone diseases secondary to primary hyperparathyroidism, hyperthyroidism, Cushing’s syndrome, multiple myeloma, rheumatoid arthritis, and osteogenesis imperfecta.
All the patients had been diagnosed as having osteoporosis according to the Japanese diagnostic criteria. Citation13,Citation14 Namely, patients with a BMD < 70% of the young adult mean (YAM) or a BMD 70%–80% of the YAM along with a history of osteoporotic fractures were diagnosed as having osteoporosis. Preliminary screening included a medical history, physical examination, plain X-rays of the thoracic and lumbar spine, lumbar spine BMD measurement, and blood and urinary biochemical tests, including serum calcium, phosphorus, alkaline phosphatase (ALP) and urinary cross-linked N-terminal telopeptides of type I collagen (NTX). Lumbar spine BMD was used for the diagnosis of osteoporosis.
The patients had been treated with ALN (5 mg daily or 35 mg weekly) over seven years. The doses indicated in parentheses above are the doses used in Japan for the treatment of postmenopausal women with osteoporosis and have been recognized as being safe and effective.Citation7 Daily ALN was available throughout the study period, but weekly ALN only became available in October 2006. All the patients had been treated with daily ALN and then switched to weekly ALN just after October 2006. The effects of daily and weekly ALN on BMD and bone turnover markers, as well as incidence of side effects, were reported to be similar in postmenopausal Japanese women with osteoporosis.Citation8 The subjects did not receive any elementary calcium or natural vitamin D supplementation.
Urinary levels of NTX were measured three months after the start of treatment, based on the results of our previous study.Citation15 Serum levels of calcium, phosphorus, and ALP, and lumbar spine BMD were measured every year after the start of treatment. Serum bone-specific ALP levels were also measured at seven years after the start of treatment. The outcome of seven years of ALN treatment was evaluated. The present study was approved by the Ethics Committee of Keiyu Orthopaedic Hospital.
Assessment of vertebral fractures
Plain lateral X-ray films of the thoracic and lumbar spine were obtained at baseline to detect evidence of morphometric vertebral fractures. According to the Japanese criteria, a vertebral fracture was defined according to the vertebral height on lateral X-ray films.Citation13,Citation14 Briefly, the vertebral height was measured at the anterior (A), central (C), and posterior (P) aspects of the vertebral body, and the presence of a vertebral fracture was confirmed when a reduction in vertebral height of more than 20% (A, C, and P) compared with the height of the adjacent vertebrae was observed, the C/A or C/P was less than 0.8, or the A/P was less than 0.5. The assessment for vertebral fractures was performed at the T4–L4 level.
Serum ALP, calcium, phosphorus, bone-specific ALP, and urinary NTX
Serum calcium, phosphorus, and ALP levels were measured by standard laboratory techniques (normal range 8.4–10.2 mg/dL, 2.5–4.5 mg/dL, and 135–310 IU/L, respectively). Serum bone-specific ALP levels were measured by enzyme immunoassay (EIA, normal range for Japanese women 7.9–29.0 U/L).Citation16 Urinary NTX levels were measured by enzyme-linked immunosorbent assay (ELISA, normal range 9.3–54.3 nM bone collagen equivalent [BCE]/mM Cr).Citation16
Measurement of lumbar spine BMD
BMD of the lumbar spine (L1–L4) in the anteroposterior (AP) view was measured using dual-energy X-ray absorptiometry (DXA) with Hologic QDR 1500W apparatus (Bedford, MA). The coefficient of variation (100 × standard deviation/mean) of five measurements, with repositioning within 72 hours each time, was less than 1.2% in three persons.
Statistical analysis
Data were expressed as the mean ± standard deviation (SD) in the table and the mean ± 95% confidence interval (CI) for calcium, phosphorus, and ALP, and the median ± 95% CI for urinary NTX in the figures. The significance of longitudinal changes in the BMD and biochemical markers was determined using a one-way analysis of variance (ANOVA) with repeated measurements. All statistical analyses were performed using the Stat View-J5.0 program on a Windows computer. A significance level of P < 0.05 was used for all the comparisons.
Results
Characteristics of subjects at start of treatment
shows the baseline characteristics of the study subjects at the start of ALN treatment. The mean age was 65.7 years. The mean lumbar spine BMD was 0.638 g/cm2 (62.7% of YAM). Twenty-three subjects (48.9%) had prevalent vertebral fractures, and the mean number of prevalent vertebral fractures per subject was 2.5. The mean level of urinary NTX was 73.3 nmol BCE/mmol Cr, indicating high turnover osteoporosis (normal range 9.3–54.3 nM BCE/mM Cr).Citation16
Table 1 Characteristics of study subjects
Changes in biochemical markers and lumbar spine BMD
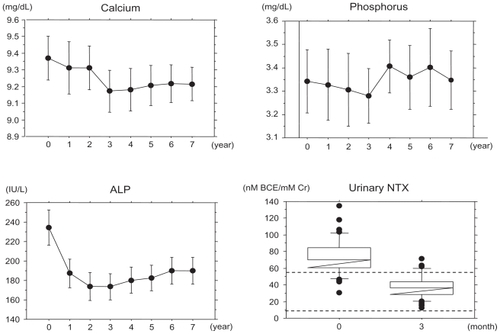
shows the changes in the biochemical markers. Urinary NTX levels decreased to the normal range (9.3–54.3 nmol BCE/mmol Cr)Citation16 after three months of treatment, and serum ALP levels decreased but remained within the normal range (135–310 IU/L) during the seven-year period. The mean change rate of the urinary NTX levels after three months of treatment was −48.2%. The mean change in serum ALP levels after two years of treatment was −24.1%, and this decrease continued gradually over the course of the seven-year treatment period (−23.9% at three years, −19.3% at five years and −15.7% at seven years). Serum calcium levels decreased after three years of treatment, and this effect was maintained. The mean change in serum calcium level after three months was −2.0%. However, the changes in the serum phosphorus levels were not significant during the course of the seven-year treatment period.
Figure 1 Changes in biochemical markers.
Data were expressed as the mean ± 95% confidence interval (CI) for calcium, phosphorus, and ALP and the median ± 95% CI for urinary NTX. One-way ANOVA with repeated measurements showed that changes in calcium and ALP, but not those in phosphorus, were significant (P < 0.0001 and P = 0.0036, respectively). Dashed lines are the upper (9.3 nM BCE/mM Cr) and lower (54.3 nM BCE/mM Cr) limits of urinary NTX levels. The normal range of serum ALP was 135–310 IU/L.
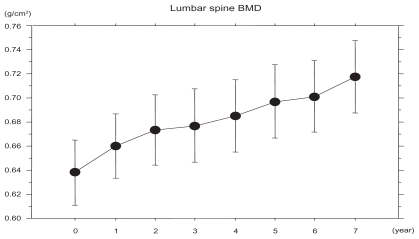
shows that the lumbar spine BMD continued to increase for seven years. Increase in lumbar BMD after three, five, and seven years of treatment was +6.1%, +9.4%, and +12.8%, respectively.
Adverse events
No serious adverse events, including ONJ, atypical femoral diaphysis fractures, or atrial fibrillation were observed.
Incident fractures
Osteoporotic fractures including vertebral, hip, wrist, and proximal humeral fractures were assessed. During the seven-year treatment period, two patients experienced vertebral fractures and two patients experienced wrist fractures. The incidence of osteoporotic fractures was 8.6% and that of vertebral fractures was 4.3%.
Discussion
To our knowledge, this is the first report showing the outcome of ALN treatment for seven years in Japanese patients with osteoporosis. ALN successfully suppressed bone turnover and increased lumbar spine BMD compared with baseline values over the seven-year treatment period without causing any severe adverse events in postmenopausal Japanese women with osteoporosis. Of importance is the effect of ALN treatment for seven years on lumbar spine BMD and bone turnover, the necessity of long-term ALN treatment, and the incidence of adverse events including ONJ, atypical femoral diaphysis fractures, and atrial fibrillation.
Studies have shown that ALN (5 mg daily or 35 mg weekly) decreases urinary NTX (−45% at three months) and serum ALP (about −20% at three years), and increases in lumbar BMD (+9.2% at three years) in postmenopausal Japanese women with osteoporosis.Citation6,Citation8 Our previous study showed that ALN treatment for five years sustained metacarpal BMD following reductions in urinary levels of NTX (−43.6% at three months) and serum levels of ALP (−18.0% at five years). In the present study, ALN treatment increased lumbar spine BMD (6.1% at three years) subsequent to reductions in urinary levels of NTX (−48.2% at three months) and serum levels of ALP (−24.1% and −19.3% at three and five years, respectively). The results of the present study are comparable with those of previous studies.
Urinary NTX levels were reduced to within the normal range for Japanese women after three months of treatment, while serum ALP levels remained within the normal range throughout the seven-year treatment period. Measurement of urinary NTX levels was permitted only twice (just before and within six months after the start of medication) in Japan for medical insurance reason. Thus, we evaluated urinary NTX only at three months after the start of treatment, because a urinary NTX measurement performed at this time provides important information and is sufficient to monitor the effects of treatment for osteoporosis.Citation15 We failed to show urinary NTX data after seven years of ALN treatment. However, we evaluated serum bone-specific ALP levels (normal range for Japanese women 7.9–29.0 U/L)Citation16 after seven years of treatment. Serum levels of bone-specific ALP (mean ± SD) at seven years was 10.0 ± 2.0 U/L, suggesting a sustained effect of ALN treatment on bone turnover for seven years.
How long postmenopausal women with osteoporosis can continue ALN treatment is debatable. A survey of the effects of treatment discontinuation would provide valuable information on this issue. Several reports have demonstrated that discontinuation of ALN affects BMD and/or levels of bone resorption markers at 1–2 years of discontinuation after 1–2 years of treatment, two years of discontinuation after two years of treatment, and 3–7 years of discontinuation after 2–6 years of treatment all decreased the BMD toward the baseline level.Citation17–Citation19 Discontinuation of ALN treatment for five years after five years of treatment resulted in a gradual loss of its effects, as measured using BMD and biochemical markers of bone turnover,Citation4 and increased the risk of clinical vertebral fractures, compared with continuation of ALN treatment.Citation20 A recent study also showed that among women without any vertebral fractures after five years of ALN treatment, continuation of ALN treatment for another five years reduced the risk of nonvertebral fractures in women with a femoral neck BMD T-score ≤ − 2.5 at five years, but not in women with a T-score > − 2, compared with the discontinuation of ALN treatment for five years after five years of treatment.Citation21 These results suggest that continuous ALN treatment for 10 years is optimal for selected patients.
If patients are not considered to have a high risk of fractures, the discontinuation of ALN treatment could probably be considered after treatment for five years, with subsequent monitoring of BMD and bone turnover markers. The most important risk factor for fractures is considered to be BMD.Citation21 Other risk factors are low-trauma fractures after the age of 40 years, a maternal history of osteoporotic fractures after the age of 65 years, a thin body build, prolonged amenorrhea, early menopause, chronic corticosteroid use (>six months), and diseases predisposing an individual to osteoporosis.Citation22 In the present study, treatment was continued in all patients because they had BMD below 70% of the YAM or at least one of the aforementioned risk factors, as evaluated at the lumbar spine after five years of ALN treatment.
ONJ, atypical femoral diaphysis fractures, and atrial fibrillation have been reported as severe adverse events,Citation10–Citation12 although whether these events are significantly related to ALN treatment remains uncertain. The efficacy and safety of ALN for 10 years has been established in postmenopausal Western women with osteoporosis.Citation4 However, adherence (persistence and compliance) of patients to treatment with oral bisphosphonates is very poor.Citation23 Because patients who withdrew from treatment were not followed up, the long-term safety of ALN remains uncertain in this patient group.
Once-yearly intravenous zoledronate has been reported to prevent morphometric vertebral, nonvertebral, and hip fractures in postmenopausal women with osteoporosis effectively (relative risk 0.30, 0.75, and 0.59, respectively) Citation24 and reduce mortality in patients who suffer a low-trauma hip fracture.Citation25 Because adherence to treatment with once-yearly intravenous zoledronate may be better than that with oral alendronate in clinical practice, zoledronate could be more useful in the prevention of osteoporotic fractures and reduce medical expenses related to treatment of hip fractures.
The incidence of vertebral fractures was 4.3% in the present study. A RCT of the two-year minodronate study showed that the incidence of vertebral and nonvertebral fractures in postmenopausal Japanese controls with established osteoporosis was 21.3% and 3.5%, respectively. Citation26 However, a three-year RCT of ALN showed that the incidence of vertebral fractures was 2% for the ALN group and 8% for the active control (alfacalcidol) group in postmenopausal Japanese women with osteoporosis.Citation6 One possible explanation for the higher incidence of vertebral fractures in the present study might be that there was a higher proportion of patients who had prevalent vertebral fractures in terms of higher risk of incident fractures at baseline as well as a comparatively long duration of observation. Another possibility might be the existence of subclinical osteomalacia on a background of significantly decreased serum calcium levels because of lack of calcium and vitamin D supplementation.
The present study confirmed the effect of ALN on lumbar spine BMD and bone turnover in postmenopausal Japanese women with osteoporosis who continued treatment for seven years. However, this study has notable limitations. First, it was a retrospective cohort study with a small sample size. Second, the subjects did not receive either elementary calcium or natural vitamin D supplementation. Natural vitamin D supplementation is not routinely given in Japan. This makes it difficult to compare our study with those of others, as most other studies have included postmenopausal women with osteoporosis taking calcium and vitamin D supplements. Moreover, performing a seven-year RCT with a sufficient number of subjects is difficult, but prospective studies with a large number of subjects are needed to establish the long-term efficacy and safety of ALN treatment with calcium and vitamin D supplementation.
In conclusion, the present retrospective study showed that ALN successfully suppressed bone turnover and increased lumbar spine BMD, compared with baseline values, over the course of a seven-year treatment period without causing any severe adverse events in postmenopausal Japanese women with osteoporosis.
Disclosures
The authors report no funding sources or conflict of interest in this work.
References
- BlackDMCummingsSRKarpfDBRandomised trial of effect of alendronate on risk of fracture in women with existing vertebral fracturesLancet1996348153515418950879
- CummingsSRBlackDMThompsonDEEffect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: Results from the Fracture Intervention TrialJAMA1998280207720829875874
- WellsGACranneyAPetersonJAlendronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal womenCochrane Database Syst Rev2008231CD00115518253985
- BoneHGHoskingDDevogelaerJPAlendronate Phase III Osteoporosis Treatment Study GroupTen years’ experience with alendronate for osteoporosis in postmenopausal womenN Engl J Med20043501189119915028823
- KushidaKShirakiMNakamuraTThe efficacy of alendronate in reducing the risk for vertebral fracture in Japanese patients with osteoporosis: A randomized, double-blind, active-controlled, double-dummy trialCurr Ther Res2002639606620
- KushidaKShirakiMNakamuraTAlendronate reduced vertebral fracture risk in postmenopausal Japanese women with osteoporosis: A 3-year follow-up studyJ Bone Miner Metab20042246246815316867
- ShirakiMKushidaKFukunagaMA double-masked multicenter comparative study between alendronate and alfacalcidol in Japanese patients with osteoporosisOsteoporos Int19991018319210525709
- UchidaSTaniguchiTShimizuTTherapeutic effects of alendronate 35 mg once weekly and 5 mg once daily in Japanese patients with osteoporosis: A double-blind, randomized studyJ Bone Miner Metab20052338238816133688
- IwamotoJMiyataASatoYFive-year alendronate treatment outcome in older postmenopausal Japanese women with osteoporosis or osteopenia and clinical risk factors for fracturesTher Clin Risk Manag2009577377919851524
- RuggieroSLDodsonTBAssaelLAAmerican Association of Oral and Maxillofacial Surgeons position paper on bisphosphonate-related osteonecrosis of the jaws – 2009. UpdateJ Oral Maxillofac Surg20096721219371809
- LenartBALorichDGLaneJMAtypical fractures of the femoral diaphysis in postmenopausal women taking alendronateN Engl J Med20083581304130618354114
- HeckbertSRLiGCummingsSRUse of alendronate and risk of incident atrial fibrillation in womenArch Intern Med200816882683118443257
- OrimoHSugiokaYFukunagaMDiagnostic criteria of primary osteoporosisJ Bone Miner Metab199816139150
- OrimoHHayashiYFukunagaMDiagnostic criteria for primary osteoporosis: Year 2000 revisionJ Bone Miner Metab20011933133711685647
- IwamotoJTakedaTSatoYEarly changes in urinary cross-linked N-terminal telopeptides of type I collagen level correlate with one-year response of lumbar bone mineral density to alendronate in Japanese postmenopausal women with osteoporosisJ Bone Miner Metab20052323824215838627
- NishizawaYNakamuraTOhtaHGuidelines for the use of biochemical markers of bone turnover in osteoporosis (2004)J Bone Miner Metab2005239710415750686
- RavnPBidstrupMWasnichRDAlendronate and estrogen-progestin in the long-term prevention of bone loss: Four-year results from the early postmenopausal intervention cohort study. A randomized, controlled trialAnn Intern Med199913193594210610644
- StockJLBellNHChesnutCHIIIIncrements in bone mineral density of the lumbar spine and hip and suppression of bone turnover are maintained after discontinuation of alendronate in postmenopausal womenAm J Med19971032912979382121
- BaggerYZTankoLBAlexandersenPAlendronate has a residual effect on bone mass in postmenopausal Danish women up to seven years after treatment withdrawalBone20033330130713678770
- BlackDMSchwartzAVEnsrudKEEffects of continuing or stopping alendronate after 5 years of treatment: The Fracture Intervention Trial Long-term Extension (FLEX): A randomized trialJAMA20062962927293817190893
- SchwartzAVBauerDCCauleyAEfficacy of continued alendronate for fractures in women without prevalent vertebral fracture: The FLEX trial (Abstr 1057)Presented at the 29th Annual Meeting of the American Society for Bone and Mineral ResearchHonolulu, HISeptember 16–20, 2007
- MeunierPJDelmasPDEastellRDiagnosis and management of osteoporosis in postmenopausal women: Clinical guidelines. International Committee for Osteoporosis Clinical GuidelinesClin Ther1999211025104410440625
- RabendaVHiligsmannMReginsterJYPoor adherence to oral bisphosphonate treatment and its consequences: A review of the evidenceExpert Opin Pharmacother20091011319236178
- BlackDMDelmasPDEastellROnce-yearly zoledronic acid for treatment of postmenopausal osteoporosisN Engl J Med20073561809182217476007
- EriksenEFLylesKWColón-EmericCSAntifracture efficacy and reduction of mortality in relation to timing of the first dose of zoledronic acid after hip fractureJ Bone Miner Res2009241308131319257818
- MatsumotoTHaginoHShirakiMEffect of daily oral minodronate on vertebral fractures in Japanese postmenopausal women with established osteoporosis: A randomized placebo-controlled double-blind studyOsteoporos Int2009201429143719101754