Abstract
Background. The incidence of prostate cancer (PC) has increased during the last 15 years in Denmark, whereas the mortality has remained largely unchanged. This register study aimed to investigate the trends in PC incidence and mortality in Denmark 1978–2009 with special focus on the recent 15 years. Material and methods. From the nationwide Danish Cancer Registry and Register of Causes of Death, we obtained information on all cases of PC and all deaths in Denmark during 1978–2009. Age-standardised (World Standard Population) incidence and mortality rates were computed for five-year calendar periods (1978–2007) and a two-year calendar period (2008–2009). Trends in incidence rates were estimated for specific age groups, birth cohorts, and clinical stage. Results. The age-standardised incidence rate of PC increased from 29.2 per 100 000 person-years in 1978–1982 to 76.2 per 100 000 person-years in 2008–2009. The incidence increase began primarily in the mid-1990s. The corresponding mortality rates of PC remained largely unchanged during the entire study period; around 19 per 100 000 person-years. The incidence increase was most pronounced among men aged 60 + years. A clear pattern was also seen for selected birth cohorts, with increasing incidence rates among the youngest cohorts, and the highest relative increase in age-specific incidence rates was seen among men born between 1943 and 1947. The distribution of stage changed from 1998, with an increasing proportion of PC patients with localised disease. Conclusion. The observed increase in PC incidence during the period 1993–2009 in Denmark may be attributed primarily to increasing unsystematic use of prostate specific antigen (PSA) testing. The mortality rates remained stable during the same period suggesting that there is not yet any major influence of intensified PSA screening and more frequent use of curatively intended therapy on the overall prognosis of PC.
Background
Prostate cancer (PC) is the second most frequent cancer in men worldwide [Citation1], and the most common non-skin cancer in Denmark amongst men [Citation2]. Internationally, the incidence rates vary more than 25-fold, with the highest rates in USA, New Zealand, Australia, and Western Europe, whereas the lowest rates are recorded in South-Central Asia [Citation1]. The corresponding mortality rates vary less by a factor 10 [Citation1]. The geographic variation in incidence rates may primarily be ascribed to ethnic differences and the use of prostate-specific antigen (PSA) testing [Citation3], but the variation may also be due to differences in risk factors since individuals migrating from countries with low PC incidence to high incidence countries approach the incidence in the high incidence countries within one or two generations [Citation4,Citation5]. Some of the observed international variations may thus be explained by differences in genetic susceptibility to PC, diet and lifestyle factors [Citation3,Citation6].
In Denmark, the incidence of PC has increased dramatically during the last 15 years, whereas the mortality has remained largely unchanged [Citation2,Citation7]. The use of PSA tests as a diagnostic tool in general practice has increased in Denmark since 1995 [Citation8], which is likely to be the main explanation for the increasing PC incidence rates. In Denmark, PSA screening has, however, not been recommended, except amongst men with two or more first or second degree relatives with PC [Citation9,Citation10].
The trends in PC incidence and mortality in Denmark until 2002 were evaluated in a previous study [Citation11]. We decided to update and expand that study investigating the trends in PC incidence and mortality rates in Denmark from 1978 to 2009 with a special focus on the recent 15 years.
Material and methods
Data sources
In this study, we identified all cases of PC registered in the Danish Cancer Registry (DCR) during the period 1978–2009. The DCR has collected data on incident cancers in the Danish population since 1943, including information on civil registry number, cancer type (topography), histology (morphology), date of diagnosis, and stage of disease [Citation12,Citation13]. The staging in the DCR was registered according to a self-derived classification until 2004, dividing neoplasms into “benign”, “unknown whether benign or malignant”, “in situ”, “localised”, “regional”, “distant”, or “unknown” [Citation12], whereas the internationally recognised Tumour, Node, Metastasis (TNM) classification has been employed since 2004 [Citation14]. The civil registry number has been assigned to all Danish residents since 1968 by the Danish Civil Registration System and incorporates information on date of birth and gender [Citation15]. Since 2004, the notification system to DCR has been electronic based on data from the Danish National Patient Register [Citation16] and general practitioners and practicing specialists that are supplemented and validated by information on histology from the Danish Pathology Register [Citation13,Citation17].
Identification of PC cases from the DCR were based on the 10th revision of the International Classification of Disease (ICD-10), code C61 [Citation13]. Information on cause of death and date of death were obtained from the Danish Register of Causes of Death [Citation18] using the civil registry number for linkage.
Statistical analysis
Incidence and mortality rates of PC for five-year calendar periods (1978–2007) and a two-year calendar period (2008–2009) were calculated and age-standardised to the 2000 World Standard Population [Citation1,Citation19]. The incidence and mortality rates were presented as number of incident cases per 100 000 person-years. Trends in incidence rates of PC were computed for specific age groups (50–59 years, 60–69 years, 70–74 years, 75–79 years, 80 + years), selected birth cohorts (1893–1897, 1903–1907, 1913–1917, 1923–1927, 1933–1937, 1943–1947, 1953–1957), and clinical stage. Clinical stage was divided into three categories according to the formal TNM classification, i.e. localised, non-localised, and unknown disease, including PC cases with missing information on stage [the algorithm is described in detail in Supplementary Table I., available online at http//www.informahealthcare.com/10.3109/0284186X.2012.702922]. A sub-analysis including a more specified categorisation of stage was made for 2004–2009, i.e. localised, locally advanced, metastatic, and unknown disease, including PC cases with missing information on stage [the algorithm is described in detail in Supplementary Table II, available online at http//www.informahealthcare.com/10.3109/0284186X.2012.702922]. The more specific categorisation of stage was only computed for 2004 to 2009, because the dichotomous categorisation of localised disease, i.e. localised and locally advanced disease, was not feasible before 2004, because the staging in the former registration system was not based on the TNM classification [Citation12]. For specific age groups (50–59 years, 60–69 years, 70–74 years, 75–79 years, 80 + years), we also computed trends in mortality rates of PC. The categorisation of the age groups was based on the fact that PC rarely occurs before age 50 years [Citation20]. We used SAS statistical software, release 9.1 (SAS Institute, Cary, NC, USA) in all analyses.
Results
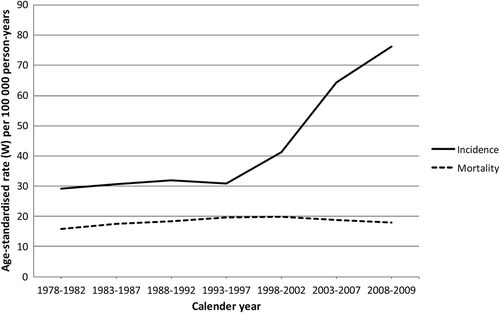
A total of 62 742 PC cases were registered in the DCR during the period 1978–2009; median age was 73.5 years (5–95 percentiles; 58.4–87.1). The age-standardised incidence rates increased from 29.2 per 100 000 person-years in 1978–1982 to 76.2 per 100 000 person-years in 2008–2009 (). The median age at diagnosis decreased from 75.1 years in 1988–1992 to 69.7 years in 2008–2009 (results not shown). The largest change in median age at diagnosis occurred in 2003–2007. The incidence increase was very modest until the five-year period 1993–1997 after which the incidence rate increased steeply from 30.9 to 76.2 per 100 000 person-years, equivalent to a 2.5-fold increase. During the same period, the age-standardised mortality rates remained largely unchanged, around 19 per 100 000 person-years. This pattern resulted in a decrease in the mortality rate/incidence rate ratio from 0.54 in the period 1978–1982 to 0.24 during 2008–2009 (results not shown).
Figure 1. Age-standardised incidence and mortality rates of PC per 100 000 person-years for all PC patients in Denmark 1978–2009.
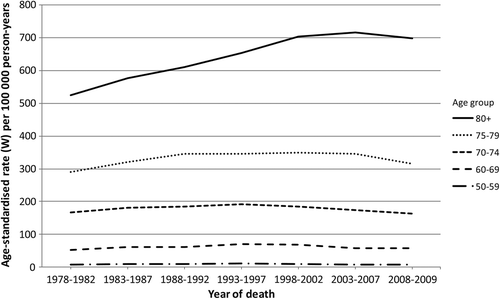
The development of age-specific incidence rates of PC for the predefined age-groups, i.e. 50–59 years, 60–69 years, 70–74 years, 75–79 years, and 80 + years, are shown in . During the study period, the incidence rates were highest among men aged 60 + years. From 1993–1997, the age-specific incidence rates increased mainly among men aged 75 years or below. Among men aged 75 + years, the age-specific incidence rate also increased from 1993–1997, but levelled off during the period 2003–2007 and then subsequently decreased. The age-specific mortality rates of PC were low and almost unchanged among men aged 50–59 years and 60–69 years during the entire study period (). Among men aged 70–74 years and 75–79 years, the mortality rates were approximately one- and two-fold higher compared to those aged 60–69 years, and for the 80 + age-group, the mortality rates increased steadily until 2003–2007 and then decreased slightly during the remaining study period.
Figure 2. Age-specific incidence rates of PC per 100 000 person-years in Denmark 1978 –2009 (age-standardised).
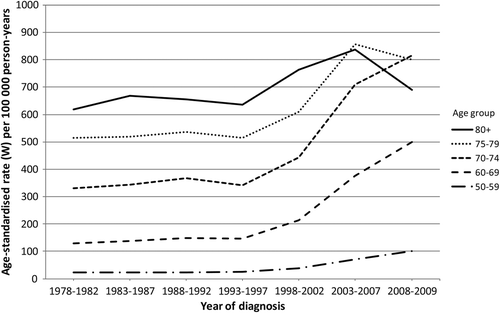
Figure 3. Age-specific mortality rates of PC per 100 000 person-years in Denmark 1978–2009 (age-standardised).
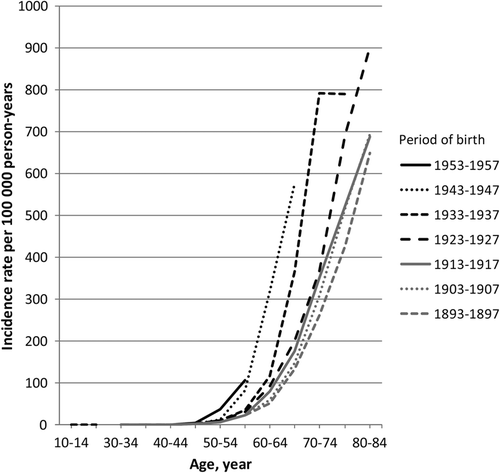
In the analysis of age-specific incidence rates of PC for selected birth cohorts in Denmark between 1893 and 1957, the incidence rates increased from age 50–54 years among all the selected cohorts (). This trend was most apparent among the youngest birth cohorts, and the highest relative increase in age-specific incidence rates were seen among men born between 1943 and 1947.
Figure 4. Age-specific incidence rates of PC per 100 000 person-years for selected birth cohorts in Denmark 1893–1957.
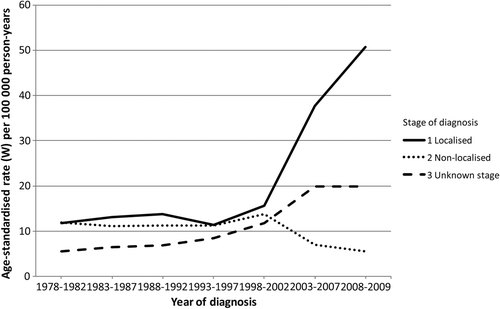
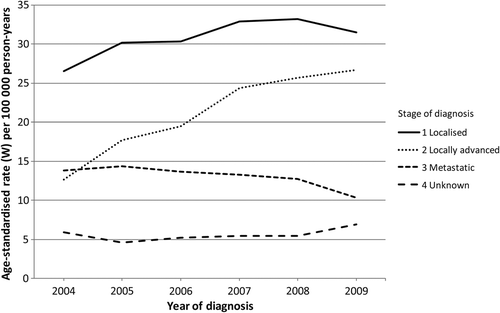
presents the age-standardised incidence rates of PC stratified by stage. The trends in incidence rates for the different stages were parallel until mid-1990s, with lowest rates for unknown stage and about equal rates for localised and non-localised PC. From 1993–1997 to 2008–2009, the incidence rate for localised disease increased from 11.4 to 50.8 per 100 000 person-years. This pattern was comparable to that for overall PC incidence (). During this period, the incidence rate for non-localised stage declined from 11.2 to 5.5 per 100 000 person-years and the rate for unknown stage increased from 8.4 to 19.9 per 100 000 person-years. The additional sub-classification of stage into localised, locally advanced, metastatic and unknown disease from 2004 to 2009 is shown in . During this period, the incidence rates for metastatic disease and unknown stage were almost unchanged; the incidence rate for locally advanced disease increased from 12.6 to 26.7 per 100 000 person-years and the rate for localised disease increased from 26.5 to 31.5 per 100 000 person-years.
Discussion
In this nationwide register study during 1978–2009 in Denmark, we found an increasing PC incidence starting almost exclusively in the mid-1990s. The increasing incidence rates were most pronounced among men aged 60 + years. In birth cohort analyses, increasing incidence rates were primarily seen among the youngest cohorts; notably among men born between 1943 and 1947. The staging of PC changed from 1998, with increasing incidence rates for localised PC and decreasing rates for non-localised disease. By contrast, the mortality rates remained relatively unchanged during the entire study period.
The major strengths of our study are its nationwide coverage including the entire population over a 30-year period, with complete and accurate registration of PC diagnoses. The major limitation is lack of details on Gleason score and PSA levels, thus limiting the ability to classify PC cases according to aggressiveness. Another limitation is the potential of misclassification between regional and metastatic disease. Since curatively intended therapy for PC was introduced later in Denmark than in most other Western countries, an exact staging may not always have been performed during the study period. Consequently, some patients with small asymptomatic metastatic PC may have been misclassified.
Until the late 1990s, the Danish attitude towards diagnosis and treatment of PC was conservative compared to other Western countries, which may explain the marked delay in the increase of PC incidence in Denmark compared to reports on PC trends from other Western countries [Citation3,Citation21]. The observed changes in PC incidence in Denmark are thus parallel to the trends in the other Nordic countries, however, with a delay of approximately 10 years [Citation7]. The introduction of curatively intended treatments in the early 1990s led to an increasing interest in diagnosis of early potentially curable PC and thus to an increase in the use of PSA testing. These diagnostic and therapeutic shifts are reflected by increasing numbers of radical prostatectomy (from 108 to 704) and curatively intended radiotherapy (from 805 to 1547) in the stable male population during the period 2000 to 2007 [Citation22,Citation23]. This pattern has continued and, thus, the attitude in Denmark towards diagnosis and treatment of PC has changed from being conservative to more proactive during a relatively short period. This is compatible with our finding of steeply increasing incidence rates of PC and also intensified PSA testing, although PSA screening is only recommended in Denmark to men with two or more first- or second degree relatives diagnosed with prostate cancer [Citation24]. A similar pattern has been observed in many other, notably Western, countries during the last 15 years in which intensified diagnostic measures, notably PSA testing, has led to a dramatic increase in PC incidence [Citation25].
Apart from the increase in PC incidence rates, this development is reflected by the decrease in median age of PC diagnosis observed during the last decades in many countries, including Denmark. Similarly, the change in stage distribution observed in the present study also demonstrates that the policy toward PC has changed over time, with more patients being diagnosed with clinically localised PC. Still, compared with other Nordic countries, the age-standardised incidence rate of PC in Denmark was lower during the entire study period 1978–2009 [Citation7]. However, during this period the age-standardised mortality rate was similar in all Nordic countries [Citation7]. For instance, in Sweden, the age-standardised incidence rate was 105.3 per 100 000 person years during 2005–2009 [Citation7], which is markedly higher than that observed in Denmark (76.2 during 2008–2009). The mortality rate was, however, almost similar in the two countries during the whole period 1978–2009 [Citation7], which is noteworthy since the relation between incidence and mortality should be evaluated over longer periods, notable for diseases with an often favourable prognosis as PC.
No risk factors have clearly been established for PC, except for age, race, and familiar disposition, and environmental factors are unlikely to explain the world-wide large increase in PC incidence [Citation6,Citation26]. The increasing incidence rate in Denmark can largely be attributed to increasing use of PSA testing carried out primarily in general practice since the mid-1990s [Citation8]. A descriptive study of PSA testing in the second-largest county of Denmark (former County of Aarhus) reported that use of PSA tests increased 43-fold from mid-1990s to 2006 [Citation8]. During this period and onwards, the cut-off limits of PSA for referral to prostate biopsy has been 4.0 ng/ml [Citation9]. A similar increase in PC incidence parallel with an increase in PSA-testing has been observed in many other Western countries [Citation3,Citation26].
Conclusion
The observed increase in PC incidence rates and changes in age- and stage distribution of the disease from 1993–1997 to 2008–2009 can largely be attributed to increasing unsystematic use of PSA tests in Denmark. The mortality rates have been relatively stable during the study period (1978–2009), suggesting that there is not yet any major improvement in PC mortality resulting from intensified use of PSA-testing and more frequent use of curatively intended therapy.
Table II
Download PDF (222.8 KB)Acknowledgements
The study was supported by the Danish Cancer Society and Bjarne Saxhof Foundation. The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010;127:2893–917.
- National Board of Health [The Danish Cancer Registry 2009]. Copenhagen: National Board of Health; 2010. Available from: http://www.sst.dk/publ/Publ2010/DOKU/Registre/Cancerregisteret2009.pdf [accessed 2012 July 5]
- Baade PD, Youlden DR, Krnjacki LJ. International epidemiology of prostate cancer: Geographical distribution and secular trends. Mol Nutr Food Res 2009; 53:171–84.
- Akazaki K, Stemmerman GN. Comparative study of latent carcinoma of the prostate among Japanese in Japan and Hawaii. J Natl Cancer Inst 1973; 50:1137–44.
- Haenszel W, Kurihara M. Studies of Japanese migrants. I. Mortality from cancer and other diseases among Japanese in the United States. J Natl Cancer Inst 1968; 40:43–68.
- Patel AR, Klein EA. Risk factors for prostate cancer. Nat Clin Pract Urol 2009; 6:87–95.
- Engholm G, Ferlay J, Christensen N, Bray F, Gjerstorff ML, Klint A, et al. NORDCAN–a Nordic tool for cancer information, planning, quality control and research. Acta Oncol 2010; 49:725–36.
- Mukai TO, Bro F, Pedersen KV, Vedsted P. [Use of prostate-specific antigen testing]. Ugeskr Laeger 2010; 172:696–700.
- Borre M, Iversen P. [Screening for prostate cancer–what does the evidence show?]. Ugeskr Laeger 2007; 169:1887–8.
- Steinberg GD, Carter BS, Beaty TH, Childs B, Walsh PC. Family history and the risk of prostate cancer. Prostate 1990; 17:337–47.
- Brasso K, Ingimarsdottir IJ, Thomassen L, Friis S, Iversen P. [Prostate cancer in Denmark 1943–2002]. Ugeskr Laeger 2007; 169:129–32.
- Storm HH, Michelsen EV, Clemmensen IH, Pihl J. The Danish Cancer Registry–history, content, quality and use. Dan Med Bull 1997; 44:535–9.
- Gjerstorff ML. The Danish Cancer Registry. Scand J Public Health 2011; 39:42–5.
- Greene FL, Page DL, Fleming ID. AJCC cancer staging manual, 6th. New York: Springer; 2002.
- Pedersen CB. The Danish Civil Registration System. Scand J Public Health 2011; 39:22–5.
- Lynge E, Sandegaard JL, Rebolj M. The Danish National Patient Register. Scand J Public Health 2011; 39:30–3.
- Bjerregaard B, Larsen OB. The Danish Pathology Register. Scand J Public Health 2011; 39:72–4.
- Helweg-Larsen K. The Danish Register of Causes of Death. Scand J Public Health 2011; 39:26–9.
- Curado MP, Edwards B, Shin HR, Storm HH, Ferlay J, Heanue M, et al. Cancer incidence in five continents Vol. IX. IARC Sci Publ No 160. Lyon: IARC; 2007.
- Schottenfeld D, Fraumeni JF. Cancer epidemiology and prevention. 3rd. Oxford: Oxford University Press Inc.; 2006.
- International Agency for Research on Cancer. Cancer incidence in five continents, Vol. I-IX, Lyon: France; 2007. Available from: http://ci5.iarc.fr/ [accessed 2012 July 5].
- National Board of Health [Cancer profile–Prostate cancer 2000–2007]. Copenhagen: National Board of Health; 2009. Available from: http://www.sst.dk/publ/Publ2009/EPT/Kraeftprofiler/Kraeftprofil_Prostatakraeft2000–2007.pdf [accessed 2012 July 5].
- Borre M, Iversen P, Bendixen A, Iversen MG, Kehlet H. [Organisation and early outcome after radical prostatectomy in Denmark 2004–2007]. Ugeskr Laeger 2008; 170:2545–9.
- Danish Prostate Cancer Group. [Clinical Guidelines 2011]. Danish Urologic Cancer Group 2012. http://ducg.dk/prostata/retningslinjer/ [accessed 2012 July 5]
- Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O, et al. International variation in prostate cancer incidence and mortality rates. Eur Urol 2012; 61:1079–92.
- Hsing AW, Tsao L, Devesa SS. International trends and patterns of prostate cancer incidence and mortality. Int J Cancer 2000; 85:60–7.