?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
The colon is a promising target for drug delivery owing to its long transit time of up to 78 h, which is likely to increase the time available for drug absorption. Progesterone has a short elimination half-life and undergoes extensive first-pass metabolism, which results in very low oral bioavailability (∼25%). To overcome these shortcomings, we developed an oral multiparticulate system for the colonic delivery of progesterone. Zn-pectinate/chitosan microparticles were prepared by ionotropic gelation and characterized for their size, shape, weight, drug entrapment efficiency, mucoadhesion and swelling behavior. The effect of cross-linking pH, cross-linking time and chitosan concentration on progesterone release were also studied. Spherical microparticles having a diameter of 580–720 µm were obtained. Drug entrapment efficiency of ∼75–100% was obtained depending on the microparticle composition. Microparticle mucoadhesive properties were dependent on the pectin concentration, as well as the cross-linking pH. Progesterone release in simulated gastric fluids was minimal (3–9%), followed by burst release at pH 6.8 and a sustained phase at pH 7.4. The in vivo study revealed that the microparticles significantly increased progesterone residence time in the plasma and increased its relative bioavailability to ∼168%, compared to the drug alone. This study confirms the potential of Zn-pectinate/chitosan microparticles as a colon-specific drug delivery system able to enhance the oral bioavailability of progesterone or similar drugs.
Introduction
Colon-specific drug delivery systems have gained increasing interest in the last decades due to the well-recognized importance of this region of the GIT, both for local and systemic administration of conventional and labile drugs (Yang et al., Citation2002; Maestrelli et al., Citation2008a; Bai et al., Citation2015). Drugs which are destroyed by the stomach acid or metabolized by pancreatic enzymes are minimally affected in the colon (Vervoort et al., Citation1998). The colon is also a useful site for delivery of drugs where a delay in drug absorption is required from a therapeutic point of view, e.g. in case of nocturnal asthma, angina, rheumatoid arthritis, etc (Rubinstein, Citation1995; Vervoort et al., Citation1998; Sinha & Kumria, Citation2003; Ramasamy et al., Citation2013). Moreover, colonic delivery is beneficial for systemic absorption of some drugs, e.g. nifedipine, theophylline, isosorbide due to the unique metabolic activity of the colon that makes it an attractive organ for targeting with various drug delivery systems (Bode et al., Citation1996; Ishibashi et al., Citation1999; Semdé et al., Citation2000).
Various approaches have been utilized for achieving colon-specific drug delivery. They can be categorized as: (a) Prodrug approach (Riley & Turnberg, Citation1990; McLeod et al., Citation1994); (b) Pressure-based systems (Muraoka et al., Citation1998; Jeong et al., Citation2001); (c) The temporal control of delivery (Steed et al., Citation1997; Del Curto et al., Citation2009); (d) pH-based systems (Klein et al., Citation2005; Basak & Adhikari, Citation2009; Sareen et al., Citation2014), and (e) Enzyme-based systems (Xi et al., Citation2005; Varshosaz et al., Citation2006; Kshirsagar et al., Citation2011; Ramasamy et al., Citation2013; Rai et al., Citation2014). Polysaccharides which undergo hydrolysis of their glycosidic bonds by the microflora of the colon appear to be the most promising because of their attractive properties, such as wide availability, low cost of production, low toxicity and biodegradability (Chourasia & Jain, Citation2004). Pectin is an anionic polysaccharide consisting of linear chains of α-(1-4) linked d-galacturonic acid residues (El-Gibaly, Citation2002) (). Pectin is non-toxic and not affected by gastric or intestinal enzymes while being completely degraded by pectinolytic enzymes produced by microflora in the colon (Bourgeois et al., Citation2006). The amidated low methoxy (LM) pectin, in which some of the carboxylic acid groups are amidated, is more tolerant to pH variations which makes it an appropriate candidate for the design of colon-targeted drug delivery systems (Oliveira et al., Citation2010). However, pectin high aqueous solubility and swelling properties are considered the major drawbacks that lead to rapid drug release during transit in the upper GIT (Maestrelli et al., Citation2008a; Wei et al., Citation2014). This necessitates the combination of pectin with other polymers or targeting strategies and also highlights the need for cross-linking of pectin chains to maintain the integrity of the dosage form until it reaches the colon (Mura et al., Citation2003).
Figure 1. (A) Chemical structure of pectin showing its functional groups: (a) carboxyl; (b) amide; (c) ester groups. (B) Chemical structure of chitosan. The indices x and y represent the mole fractions of d-glucosamine and N-acetyl-d-glucosamine moieties, respectively. (C) Schematic illustration of the formation of PG-loaded microparticles. * Indicates continuation of the structure.
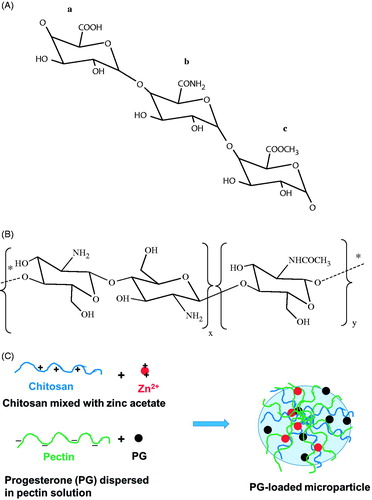
Chitosan is a cationic polysaccharide obtained by alkaline N-deacetylation of chitin, the main component of the protective shells of crustaceans (Sinha et al., Citation2004). It is a co-polymer of d-glucosamine and N-acetyl-d-glucosamine (George & Abraham, Citation2006) (). Chemically, chitosan has one primary amino and two free hydroxyl groups in each d-glucosamine building block. Due to the availability of this amino group, chitosan carries positive charges at pH ≤6.5 which enable it to react with a variety of negatively charged polymers and surfaces (Sinha et al., Citation2004). However, chitosan microspheres have shown limited strength with very fast drug release in the stomach due to the high solubility of chitosan in acidic media (Risbud et al., Citation2000). Despite these results, the use of chitosan for colonic targeting offers a great advantage in virtue of its insolubility at pH values above 6.5 that prevail in the jejunum and ileum parts of the small intestine while it gets soluble again at the colon pH (Wittaya-areekul et al., Citation2006). Thus, chitosan has to be combined with another polymer to prevent premature drug release in the stomach and ideally release it in the colon (Wittaya-areekul et al., Citation2006). Pectin/chitosan microparticles that are formed by the electrostatic interactions between the pectin carboxylate groups and chitosan amino groups could overcome chitosan shortcomings and prevent premature drug release in the stomach due pectin insolubility in acidic media. Further, the insolubility of chitosan at pH ≥6.5 overcomes the pectin high solubility in the small intestine and prevents premature drug release before reaching the colon. These microparticles have many other desirable properties as drug-delivery vehicles, such as biocompatibility, non-toxicity, mucoadhesiveness and biodegradability (Chen & Fan, Citation2007; Mennini et al., Citation2008; Jin et al., Citation2014). Pectin/chitosan microparticles can be further reinforced through ionic cross-linking between pectin and zinc ions with the formation of semi-interpenetrating polymer networks.
Progesterone (PG) is a naturally-occurring female sex hormone, widely used for maintenance of pregnancy in case of habitual or threatened abortion. It is also used as an oral contraceptive, alone or in combination with estrogen in addition to many other uses as hormone replacement therapy (Sweetman, Citation2009). PG is currently administered by I.M. injection or as rectal and vaginal suppositories. Natural PG has a very short elimination half-life (19–95 min) and undergoes extensive pre-systemic elimination in the liver and gut wall which results in very low oral bioavailability (∼25%) (Dollery, Citation1999). Higher doses are then required to achieve effective plasma drug concentrations, which further increase the risk of GI side-effects and also augment the economic burden of treatment. These shortcomings could be overcome by developing a suitable colonic delivery system for PG.
The current work was then undertaken to establish the feasibility of creating Zn-pectinate/chitosan microparticles (MPs) for the colonic delivery of PG. The MPs were prepared at different pectin/PG ratios and at different chitosan concentrations. They were evaluated for their size, shape, mucoadhesion strength and PG entrapment efficiency. PG release kinetics from the microparticles were also investigated. Selected MPs were tested in vivo for their ability to increase PG plasma residence time and enhance its oral bioavailability.
Materials and methods
Materials
Micronized PG was a generous gift from Pharco Pharmaceuticals Inc. (Alexandria, Egypt). Genu pectin (type LM-104 AS-FS) with degree of esterification of 36% and degree of amidation of 14% was obtained from Pkelco, Atlanta, GA. Chitosan (average MW of 850 kDa) with degree of deacetylation of 90% was purchased from Industrial Manufacturing Co. (Kyoto, Japan). Mucin from porcine stomach, type II, with sialic acid content of 1%, acetonitrile and methanol (HPLC grade) were purchased from Sigma Chemical Co. (St. Louis, MO). Zinc acetate dihydrate, monobasic sodium phosphate, disodium hydrogen phosphate, hydrochloric acid, dehydrated ethanol and diethyl ether were obtained from Prolabo Chemicals (Cairo, Egypt). All other chemicals were of analytical grade and were used as received.
Preparation of Zn-pectinate/chitosan MPs
The MPs were prepared following the modified ionotropic gelation technique previously described by El-Gibaly (Citation2002). In a typical experiment, pectin solution (4% w/v) in deionized water was prepared followed by dispersion of PG (1% w/v) under stirring until homogenous dispersion was obtained. Chitosan solution (1% w/v) was prepared by dissolving chitosan in 2% v/v aqueous acetic acid solution followed by mixing with the cross-linking agent solution (4% w/v Zn(CH3COO)2). The pH of this solution was adjusted to 1.2, 3 or 4.8. The homogenous, bubble-free pectin-PG mixture was then added drop-wise at an average rate of 1 ml/min with a disposable syringe (needle size 23 G) into 20 mL of a gently stirred mixture of chitosan/Zn(CH3COO)2. The falling distance was fixed at 5 cm. The produced MPs were allowed to cure in the cross-linking solution for 120 min. The MPs were separated, washed three times with deionized water and dried at room temperature for 48 h and subjected to further evaluation. Empty MPs were prepared following the same procedures and were used as control. Different batches were prepared by varying the formulation parameters, such as cross-linking time, cross-linking solution pH, pectin/PG ratio and chitosan concentration (). All batches were prepared in triplicates.
Table 1. Preparation conditions of progesterone-loaded Zn-pectinate/chitosan microparticles.
Drug-polymer compatibility studies
Fourier-transform infrared spectroscopy studies
Samples (3–4 mg) of PG, pectin, chitosan, empty and PG-loaded MPs were mixed with potassium bromide (IR grade), compressed into disks under vacuum and analyzed using JASCO FT-IR– 4200 type A (JASCO Co., Tokyo, Japan). Spectral scanning was done in the range between 4000 and 500 cm−1.
Differential scanning calorimetry studies
Differential scanning calorimetric (DSC) analyses were performed using a computer-interfaced Shimadzu Calorimeter (Model DSC–50, Kyoto, Japan). Samples (3–4 mg) of PG, pectin, chitosan, empty and PG-loaded MPs were placed in aluminum pans, sealed and continuously heated at a rate of 10 °C/min in the range of 25–350 °C under constant flow of nitrogen gas. Indium was sealed in an aluminum pan and used to calibrate the instrument.
Characterization of microparticles
Size and shape
Twenty MPs were randomly selected from each batch. Morphological analyses were performed on images captured by Leica EC3 camera connected to Leica optical microscope (Leica Microsystems, Heerbrugg Switzerland). The images were digitalized and the perimeter (P) and projected area (A) of the MPs were determined by image analyzing software (Imagej, ver. 1.48 Bethesda, MD). The pixel resolution was ∼8 µm per pixel. The length and breadth were taken as the longest and shortest diameter of each MP, respectively. The MP size was then calculated from equation (Equation1(1)
(1) ) (Das et al., Citation2011):
(1)
(1)
The shape of MPs was shown in terms of elongation ratio (ER) and circularity (C) which were calculated from equations (Equation2)(2)
(2) and Equation(3
(3)
(3) ), respectively:
(2)
(2)
(3)
(3)
where A and P are the projected area and the perimeter of the MP as seen in the image, respectively (Berggren & Alderborn, Citation2001). According to Das et al. (Citation2011), MPs are considered spherical when their corresponding ER values are less than 1.15. Also, the more spherical a particle, the closer its C to 1; while more elongated particles have lower observed circularity (Eun Shim et al., Citation2004).
Morphological examination of MPs
Surface topography of MPs was examined under scanning electron microscope (SEM). Samples of dried MPs were mounted onto stubs, sputter coated with gold in a vacuum evaporator and photographed using a scanning electron microscope (Jeol, JSM-5200, Tokyo, Japan) at 15 keV.
Weight, weight loss and moisture content
Fifty MPs were randomly selected from each batch and dried for 48 h at 25 °C and relative humidity of 60%. Their weight before (WW) and after (WD) drying were accurately measured using an analytical balance (Sartorius TE-2145, Goettingen, Germany). Weight loss of MPs upon drying was calculated from the following equation:
(4)
(4)
In order to completely remove moisture, the selected MPs were placed in oven at 60 °C for 72 h or until no further weight change was observed and the weight of these completely dried MPs was recorded as (WF). The moisture content of MPs was calculated from the following equation (Das & Ng, Citation2010a):
(5)
(5)
Drug entrapment efficiency
MPs (25 mg) were crushed in a mortar, extracted several times with dehydrated ethanol (50 mL), stirred for 2 h and then filtered through filter paper. The filtrate was properly diluted and the absorbance of an aliquot was measured at 242 nm using a Shimadzu, Double-Beam spectrophotometer 150-02, Kyoto, Japan. Empty MPs prepared under the same conditions were used as a control. The percent drug entrapment efficiency was calculated from the following equation:
(6)
(6)
where AQ is the actual quantity of drug in the MPs and TQ is the total quantity of drug used in MP preparation.
In vitro mucoadhesion studies
In vitro evaluation of the mucoadhesive properties of Zn-pectinate/chitosan MPs was carried out using the wash-off method previously described by Wittaya-areekul et al. (Citation2006) with slight modifications. Mucin discs (200 mg) were prepared by direct compression using a single punch hydraulic press (Carver Inc. Wabash, IN) with 12-mm diameter flat-faced die tooling. The discs were obtained by applying a pressure of 2.5 tons for 1 min. Next, each disc was attached on a microscopic slide using cyanoacrylate glue and hydrated with 80 µL of enzyme-free simulated small intestine fluid (SSIF) (10 mM phosphate buffer pH 7.4, 0.02% Tween 80) for 1 min in order to obtain a gel-like adhesive surface. Twenty MPs from each batch were allowed to swell in SSIF for 3 min. The MPs were then attached to the mucin discs by placing a small stainless steel plug weighing 30 g over them for 2 min to force them to adhere to the pre-hydrated mucin disc. The slides were then placed horizontally in the glass cells of the USP XXV dissolution apparatus (Erweka, DT-D6, Heusenstamm, Germany) and MPs were forced to wash-off under the stirring motion of 50 rpm in 200 mL of SSIF (pH 7.4) at 37 °C. The number of MPs remaining attached after 15, 30, 45 and 60 min was counted and mucoadhesion was expressed as the percent of MPs remaining attached at each time point.
Swelling behavior of MPs
Swelling studies of MPs were carried out in enzyme-free simulated gastric fluid (SGF, 0.1 M HCl, pH 1.2) and enzyme-free simulated small intestinal fluid (SSIF, pH 7.4). Constant weight of MPs (100 mg) was placed in the baskets of the USP XXV dissolution apparatus (Erweka, DT-D6, Heusenstamm, Germany) and immersed in 500 mL of these media. MPs were allowed to swell for the required period of time at 37 ± 0.2 °C. At different time intervals, MPs were removed, dried with filter paper and their weight changes, after correction for drug loss were measured. In order to account for the drug loss, MPs were dried at each time point, weighed and a 5-mL sample was withdrawn from the swelling medium and assayed for drug content as described above. The amount of drug released into the medium was then added to the actual MPs weight to compensate for drug loss due to dissolution during the swelling period. The swelling index (SI) of MPs was then calculated from the following equation (Sharma et al., Citation2013):
(7)
(7)
where WT and W0 are the weight of MPs at time t and at the beginning of test, respectively.
In vitro drug release studies
The release of PG from Zn-pectinate/chitosan MPs was investigated using the in vitro USP XXV dissolution apparatus (Erweka, DT-D6, Heusenstamm, Germany) with the dissolution basket assembly at a rotational speed of 50 rpm and a temperature of 37 ± 0.2 °C. In order to simulate pH changes along the GIT, the sequential pH change method reported previously was used with slight modification (Ribeiro et al., Citation2014). Thus, an accurately weighed amount of MPs containing 5 mg drug was immersed in 200 mL of SGF (0.1 M HCl, pH 1.2) for 2 h. Subsequently, the baskets containing the MPs were removed and immediately transferred to 200 mL of fresh SSIF (10 mM phosphate buffer, pH 6.8). After 0.5 h, the baskets were removed and transferred to 200 mL of fresh SSIF (10 mM phosphate buffer, pH 7.4) for the rest of the study. All release media contained 0.02% Tween 80. At pre-determined intervals, 5-mL samples were withdrawn with an equal volume of fresh medium being added in order to maintain a constant volume of the release medium throughout the study. Samples were then assayed for PG content spectrophotometrically at 242 nm. The PG concentration was calculated from a previously constructed calibration curve. Empty MPs were used as a control. All the experiments were done in triplicates and the results were expressed as mean ± SD.
Kinetics of drug release
In order to find the best-fitting drug-release model, the drug-release data was fitted into different models using linear regression analysis. The following models were used:
Zero order: (Sood & Panchagnula, Citation1998)
First order: (Carbinatto et al., Citation2014)
Higuchi diffusion model: (Higuchi, Citation1963)
Baker–Lonsdale model: (Baker & Lonsdale, Citation1974)
Hixson–Crowell cube root law: (Chawla et al., Citation2000)
Korsmeyer–Peppas equation: (Ritger & Peppas, Citation1987)
where R is the fraction of drug released at time t, k is the rate constant corresponding to each model, (UR) represent the unreleased fraction of drug, Mt/M∞ denotes the fraction of drug released at time t and n is the diffusional exponent indicative of the release mechanism.
In vivo evaluation of MPs
The in vivo study was designed as a randomized parallel study using twelve healthy male New Zealand white rabbits weighing 1.8–2 kg. All the experiments were done according to Animal Ethical Guidelines approved by Assiut University, Egypt. Rabbits were randomly divided into two groups, each consisted of six rabbits. They were fasted for 12 h prior to drug administration but had free access to water (Qureshi et al., Citation2014). Food was not allowed for further 12 h after drug administration. A dose of 10 mg/kg body weight of PG powder or equivalent dose of selected microparticle formulations (corresponding to 200 mg human dose) were filled into hard gelatin capsules of size 4 and then administered to rabbits by the aid of stomach tube. Blood samples of about 1–2 mL were withdrawn via an indwelling catheter inserted in the eye vein, into a 5-mL screw-capped heparinized centrifuge tubes at the following time points: pre-dose, 1, 2, 3, 4, 6, 8, 10, 12, 18, 24 and 30 h following drug administration (Andréen et al., Citation2006). Sample preparation and assay method were performed as described previously with slight modifications (Wu et al., Citation2000). Briefly, blood samples were centrifuged at 5000 rpm for 15 min and plasma was separated. Liquid–liquid extraction with diethyl ether was performed to recover PG from plasma before analysis. Samples were then analyzed by high performance liquid chromatography (HPLC, Shimadzu 2010A, Kyoto, Japan) using a RP-C18 column (Phenomenex, 150 × 4.6 mm, 5 µm). The mobile phase consisted of filtered, degassed mixture of acetonitrile and water (70/30 v/v) pumped at a flow rate of 1 ml/min. The column was maintained at room temperature and the effluent was delivered directly into the UV-diode array detector (model K-2500; Knauer, Berlin, Germany). The detection wave length was set at 240 nm. The lower limit of detection for this assay method was found to be around 1 ng/ml. Drug recovery from plasma was found to be >92%. Serum concentrations versus time profiles were fit using one-compartment open model with oral input and first-order output. The peak plasma concentration (Cmax) and the time to attain peak concentration (Tmax) were obtained directly from the plasma concentration-time profile. Absorption rate constant (Ka), elimination half-life (t½), area under the curve (AUC) and other parameters were calculated according to conventional algorithms (Gibaldi, Citation1991).
Statistical analysis of data
All experiments were performed in triplicates and results were expressed as mean ± SD. Statistical analysis of data was performed using the Graph-Pad Prism version 5 software (La Jolla, CA). For statistical evaluation, the data was analyzed by one-way analysis of variance (ANOVA) with Newman–Keuls post-hoc test and differences were considered significant at p < 0.05.
Results and discussion
Preparation of Zn-pectinate/chitosan microparticles
Progesterone-loaded MPs were prepared by adding aqueous pectin solution containing dispersed drug to a mixture of chitosan and zinc acetate aqueous solutions. Gelled spheres were produced immediately due to the formation of intermolecular ionic cross-links between the negatively charged carboxyl groups of pectin and the divalent zinc cations. This bridging of pectin chains forms what is called the egg-box complex. These ionic cross-links were further reinforced by electrostatic interactions between the negatively charged carboxyl groups of pectin and the positively charged amino groups of chitosan () (Marudova et al., Citation2004).
Characterization of the prepared microparticles
FT-IR studies
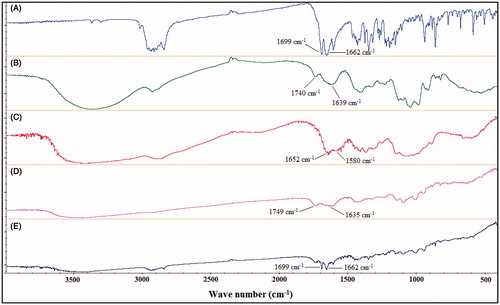
The FT-IR spectra of PG, pectin, chitosan, empty and PG-loaded Zn-pectinate/chitosan MPs are depicted in . The PG spectrum shows two typical intense bands at 1662 and 1699 cm−1 corresponding to stretching vibrations of the C=O groups of PG (Liu et al., Citation2007). The spectrum of pectin () shows a broad band between 3600 and 3000 cm−1 due to stretching of its OH groups in addition to two important bands: low-intensity band at 1740 cm−1 and a stronger one at 1639 cm−1 which are attributed to the esterified and non-esterified carboxyl groups of pectin, respectively (Maestrelli et al., Citation2008a). Chitosan spectrum () shows the characteristic bands of amidic C=O stretching at 1652 cm−1 and of amidic N-H bending at 1580 cm−1 (Bigucci et al., Citation2008). The spectrum of empty MPs evidenced disappearance of pectin band at 1740 cm−1 and strong reduction in the intensity of carboxylate ion band which was also shifted to lower wave number (1635 cm−1), both indicative of Zn-pectinate formation (Maestrelli et al., Citation2008b). Further, the appearance of new band at 1749 cm−1 in this spectrum indicates mutual interaction between the COO− group of pectin and the +NH3 group of chitosan, thus confirming the electrostatic interactions between them (Marudova et al., Citation2005). However, these changes could not be clearly observed in the spectrum of PG-loaded MPs due to overlapping and masking of these changes by the strong absorption bands of PG, which were retained unchanged in all spectra, indicating no significant interactions with the MP components.
Differential scanning calorimetry studies
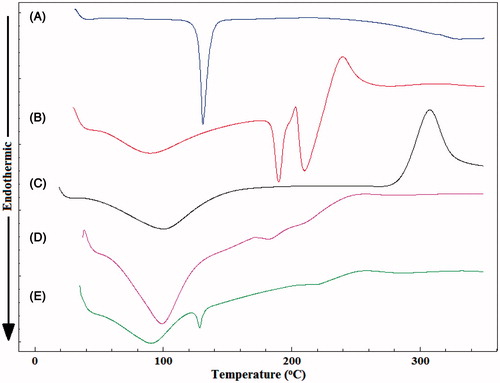
Differential scanning calorimetry (DSC) thermograms of PG, pectin, chitosan, empty and PG-loaded Zn-pectinate/chitosan MPs are presented in . PG exhibited a characteristic sharp endothermic peak at 130 °C corresponding to its melting () (Memişoğlu et al., Citation2003). shows the thermogram of pectin where a forked peak appears at ∼190 °C and 210 °C due to its endothermic transition and melting process, respectively (Auriemma et al., Citation2013). Also, a small exothermic deviation from baseline at around 240 °C was noticeable, indicating the start of pectin chains depolymerization (Maestrelli et al., Citation2008a). Chitosan () shows a broad endothermic peak at ∼95 °C due to the evaporation of absorbed water and a sharp exothermic peak starting at 280 °C and gaining its maximum at 310 °C due to chitosan degradation (Borges et al., Citation2005). The thermograms of both empty () and PG-loaded MPs () show disappearance of the degradation exothermic peaks of chitosan at 310 °C and of pectin at 240 °C, as well as pectin endothermic peaks which could be attributed to the electrostatic interactions between pectin and chitosan. Similar results were previously reported for chitosan-alginate polyelectrolyte complex (Simonoska Crcarevska et al., Citation2008). The thermogram of PG-loaded MPs () shows the characteristic melting peak of PG with no change in the peak shape or position confirming the absence of physical or chemical interactions with the microparticle components. Reduction of PG peak intensity after loading into the microparticles might be attributed to the dilution effect.
Size and shape of microparticles
The size of MPs from different batches ranged between 583.3 ± 30.0 and 718.3 ± 44.4 µm (). Statistical analysis showed no significant difference in size among formulations, except for the formula prepared at pectin/PG ratio of 2:1 (F8) which was very significantly smaller in size (p < 0.001). The smaller size of this formula might be attributed to its lower pectin content compared to other formulations. This produced a less viscous solution and yielded smaller droplets, which in turn resulted in smaller MPs (Jose et al., Citation2011).
Table 2. Physical characteristics and entrapment efficiency of the prepared Zn-pectinate/chitosan microparticles.
The elongation ratio (ER), along with circularity (C) provide an accurate indicator about the MP shape. MPs are considered spherical when their corresponding ER values are less than 1.15 and their C approaches unity (Eun Shim et al., Citation2004; Das et al., Citation2011). All formulations exhibited good sphericity as indicated by their quite acceptable ER and C values (). However, some formulations, namely those made with chitosan concentration <0.5% or pectin/PG ratio of 2:1, possessed slightly higher ER and lower C values, presumably due to depression of one side of their weak surface during drying (Das et al., Citation2011). Maestrelli et al. (Citation2008a) also demonstrated that lower degrees of cross-linking yielded microspheres with irregular shape.
Weight, weight loss and moisture content
The weight of 50 MPs from different batches after drying at room temperature for 48 h ranged from 11.8 to 30.9 mg (). Statistical analysis showed that the MP weight decreased very significantly (p < 0.001) with decreasing the cross-linking pH. For instance, the microparticle weight decreased from 27.2 ± 1.3 (F1) to 22.4 ± 1.6 (F3) when the pH decreased from 4.8 to 1.2, respectively. This could be attributed to the promotion of stronger cross-links at lower pH which resulted in contraction of polymeric chains with consequent squeezing of the entrapped water (Das et al., Citation2011). Moreover, MPs prepared at pectin/PG ratio of 4:1 were very significantly (p < 0.001) heavier than those prepared at pectin/PG of 2:1 (). This might be attributed to the two-fold increase in pectin concentration from 2 to 4% which facilitated the incorporation of more pectin into the MPs and thus increased their weight.
There was no significant difference among formulations in their weight loss (WL) (), except for the formula F8 prepared at pectin/PG ratio of 2:1, which exhibited a very significantly higher WL (p < 0.001). This might be attributed to the lower pectin content of this formula and the lower cross-linking density leading to a more water permeable matrix (Maestrelli et al., Citation2008a). Moisture content (MC) of different batches after drying at 60 °C for 72 h ranged between 5.88 and 9.65% (), which reflects good chemical and microbiological stability of the formulations. MC increased significantly (p < 0.05) by reducing the cross-linking pH from 4.8 to 1.2 (6.09 and 9.65%, respectively). These results were not in accordance with those of Das et al. (Citation2011) who reported a decrease in MC by decreasing the cross-linking pH. This contradiction may be related to differences in the characteristics of pectin used, such as its degree of amidation (DA) which affects its hygroscopicity. Further, the formula having pectin/PG ratio of 2:1 showed a significantly lower MC (p < 0.05) as compared to formula F3 made at a ratio of 4:1 (5.88 and 9.65%, respectively). This might be attributed to the lower polymer content of the former formula, in addition to its higher weight loss during the initial drying step () (Das et al., Citation2011).
Drug entrapment efficiency of microparticles
All the prepared formulations showed high PG entrapment efficiency of ≥88.4%, except for formula F1 in which the entrapment efficiency was 76.7% (). This high drug entrapment efficiency was attributed to the very limited aqueous solubility of PG, which prevented its diffusion from the MPs into the surrounding aqueous medium, in addition to the rapid formation of the cross-linked matrix (Mennini et al., Citation2012). Such high drug entrapment efficiency is advantageous for clinical applications where clinical response could be achieved with minimal amount of MPs.
It was also noticed that the entrapment efficiency increased significantly (p < 0.001) from 76.7 to 101.2% upon decreasing the cross-linking pH from 4.8 to 1.2. Taking into account that the solubility of PG is unaffected by this pH change, one possible explanation is that the high cross-linking density at low pH leads to the formation of a more dense matrix with minimal drug leaching. This was further supported by a slight increase in the entrapment efficiency upon increasing the cross-linking time from 10 to 120 min (97.8 and 101.2%, respectively). Chitosan concentration had a minimal effect on the drug entrapment efficiency, possibly due to its low values in the formulations (0.1–0.5%) ().
In vitro mucoadhesion studies
Mucoadhesion offers the advantage of extending the dosage form residence time along the GIT, which could enhance the oral bioavailability and efficacy of treatment (Bigucci et al., Citation2008). Fortunately, pectin shows maximum mucoadhesion on the large intestinal tissue compared to other regions of the GIT, which represents an added advantage for colon targeting (Thirawong et al., Citation2007). It is noteworthy that the pressure applied to adhere MPs to the hydrated mucin discs is not relevant to the in vivo conditions but this test is helpful in screening the most promising formulation in terms of mucoadheisve strength.
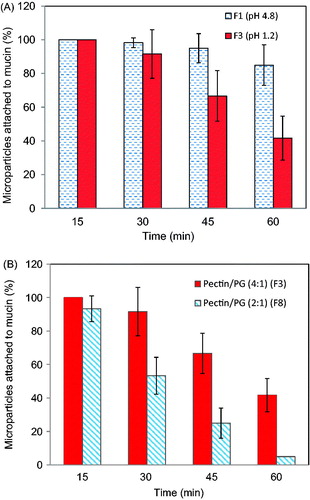
shows the effect of cross-linking pH on the mucoadhesion of Zn-pectinate/chitosan MPs. The MPs were prepared at chitosan concentration of 0.5%, pectin/PG ratio of 4:1 and cross-linking time of 120 min while the pH of cross-linking solution was either 4.8 (F1) or 1.2 (F3). Almost all the MPs fabricated at both pH values remained attached to the mucin discs for 30 min, after which the number of attached particles was dependent on the cross-linking pH. For instance, 85 and 41.7% of MPs remained attached to the mucin discs after 1 h, for the MPs made at pH 4.8 and 1.2, respectively (). The ionization of pectin carboxyl groups (pKa ∼4.2) is higher at pH 4.8 compared to pH 1.2 which in turn, enhances its swelling and physical entanglement with mucin chains (Bigucci et al., Citation2008; Hagesaether et al., Citation2008). shows the effect of pectin/PG ratio on the mucoadhesive properties of MPs prepared at chitosan concentration of 0.5%, cross-linking time 120 min and pH 4.8. Reducing pectin/PG ratio from 4:1 to 2:1 adversely affected the microparticle mucoadhesive properties as indicated by a fall in the percent of adhering particles after 1 h from 41.7 to 5% (). This might be attributed to the lower pectin concentration employed along with the much smaller size of these MPs which make them easily washable from the mucin discs surface (Hagesaether et al., Citation2008).
Figure 4. (A) Effect of cross-linking pH on the mucoadhesive properties of Zn-pectinate/chitosan MPs prepared at pH 4.8 (F1) or pH 1.2 (F3) (Preparation conditions: chitosan concentration, 0.5%; pectin/PG, 4:1; cross-linking time, 120 min) (B) Effect of pectin/PG ratio on the mucoadhesive properties of pectin/chitosan MPs prepared at pectin/PG of 4:1 (F3) or 2:1 (F8) (Preparation conditions: pH, 1.2; chitosan concentration, 0.5%; cross-linking time, 120 min).
Microparticle swelling behavior
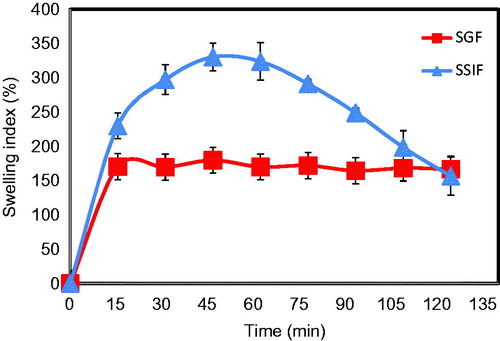
Microparticles based on electrostatic interactions usually show pH dependent swelling behavior due to the pH effect on the ionization of the interacting species (Bigucci et al., Citation2008). shows that the MPs had a moderate swelling in SGF during the first 15 min where the swelling index was ∼150% and remained at that level for the rest of the study period. At the low pH of SGF, the carboxyl groups of pectin were not ionized, which reduced the repulsive forces among the polymer chains and decreased their swelling (Oliveira et al., Citation2010). Although chitosan was fully ionized in SGF and could increase the MP swelling, its lower concentration (0.5%) compared to that of pectin (4%) limited its effect on the MP swelling. Contrary to SGF, the MP swelling in SSIF was much higher. Thus, the microparticles continued to swell for up to 45 min where a swelling index of ∼330% was achieved which was very significantly higher than that observed in SGF (p < 0.001) (). This higher swelling could be attributed to the ionization of pectin carboxylate groups in alkaline media, which increases the repulsion between the polymer chains and hence increases their swelling (Hiorth et al., Citation2006). The MP swelling index started to decrease after 1 h due to microparticle erosion and dissolution. This might be attributed to counter ion exchange between sodium and potassium ions in SSIF and zinc ions in the microparticles leading to the formation of water soluble pectin salts that facilitate microparticle erosion (Munjeri et al., Citation1997; Bajpai & Sharma, Citation2004).
In vitro drug release
In order to get an insight into the drug-release behavior from different formulations, all release studies were carried out under conditions simulating the different pH values encountered along the GIT. Thus, enzyme-free SGF (pH 1.2) was used for the initial 2 h, followed by enzyme-free SSIF (pH 6.8) for 0.5 h and finally enzyme-free SSIF (pH 7.4) until the end of release study. All the studied formulations showed a very significantly slower release in the enzyme-free SGF compared to the drug alone (p < 0.001) confirming the ability of the microparticles to prevent premature drug release in the stomach (). Thus, PG release from Zn-pectinate/chitosan microparticles in SGF was ∼3–9%, compared to 52% for the drug alone under identical conditions. Previous studies have shown that chitosan microparticles cross-linked with tripolyphosphate released ∼60% of a lipophilic drug after 2 h in SGF (Wan et al., Citation2012). Thus, the minimal progesterone release from Zn-pectinate/chitosan microparticles in SGF confirms the ability of these microparticles to overcome the disadvantages of chitosan microparticles.
Figure 6. (A) In vitro release profiles of PG powder and PG-loaded Zn pectinate/chitosan MPs prepared at a cross-linking pH of 4.8 (Formula F1) or pH 1.2 (Formula F3). (B) In vitro release profiles of PG powder and PG-loaded Zn pectinate/chitosan MPs cross-linked for 10 min (Formula F6) or 120 min (Formula F3). (C) In vitro release profiles of PG powder and PG-loaded Zn pectinate/chitosan MPs prepared at a chitosan concentration of 0.1% w/v (Formula F4) or 0.5% w/v (Formula F3). Release medium: enzyme-free SGF (pH 1.2) for the initial 2 h, followed by enzyme-free SSIF (pH 6.8) for 0.5 h and then enzyme-free SSIF (pH 7.4) until the end of release study (30 h).
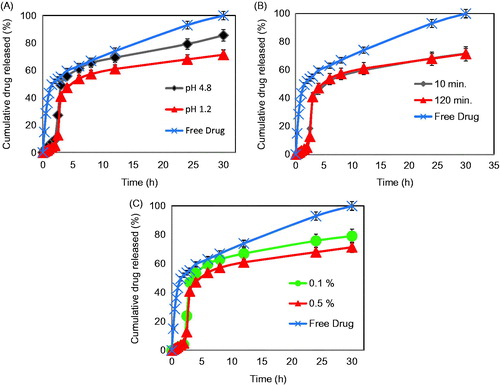
Progesterone release rate rapidly increased upon increasing the pH to 6.8 and 7.4 (). This pH-dependent drug release is presumably attributed to the pH-dependent swelling behavior of the microparticles as illustrated in . High swelling and water uptake of the microparticles at pH 7.4 loosen the microparticle matrix giving a chance for the incorporated drug to be dissolved and released (Yu et al., Citation2009; Araujo et al., Citation2013).
The effect of the cross-linking solution pH on the drug-release pattern is illustrated in . The microparticles prepared at a cross-linking pH of 1.2 pronouncedly released the drug at a slower rate (t1/2 = 11.4 h) as compared to those cross-linked at pH 4.8 (t1/2 = 7 h) (). This might be attributed to the reduced solubility and charge density of pectin at low pH which allowed its conformational transition from the two-fold into the more compact three-fold helical structure (Das & Ng, Citation2010b). In addition, non-ionic interactions between pectin and chitosan, such as hydrophobic interactions and hydrogen bonds are more favored at low pH (Lootens et al., Citation2003; George & Abraham, Citation2006; Bigucci et al., Citation2008). Contrary to the cross-linking pH, the cross-linking time did not affect the drug release rate. Thus, the MPs prepared at a cross-linking time of either 10 or 120 min have almost the same drug release rate (). This is in agreement with previous reports where an increase in the cross-linking time from 5 to 20 min did not appreciably affect the drug release rates (Maestrelli et al., Citation2008b; Mennini et al., Citation2008). This might be attributed to the rapid nature of ionic interactions where a minimum cross-linking time of 5 min was reported to be enough for complete formation of microspheres (Maestrelli et al., Citation2008b; Mennini et al., Citation2008).
The effect of chitosan concentration on the drug release behavior is shown in . Increasing the concentration from 0.1 to 0.5% w/v resulted in slowing the drug release rate in SSIF as evidenced by an increase in the t1/2 value from 8.5 to 11.4 h (). Higher concentration of chitosan might form a more rigid polymeric matrix through contraction of micro-voids which could retard the drug diffusion from MPs (Maestrelli et al., Citation2008a). Several studies have previously shown that a certain dosage form requires at least 2–4 h to reach the colon (Krishnaiah et al., Citation1998; Rubinstein, Citation2005). Taking into account that all the microparticles released a relatively small amount of progesterone during the first 3 h of the study, it could be assumed that most of the incorporated progesterone will be delivered to the colon.
Kinetics of drug release
In order to determine the exact mechanism of drug release from microparticles, the release data was analyzed according to various kinetic models (). A comparative evaluation of the obtained correlation coefficients showed that the Korsmeyer–Peppas model was the most appropriate to describe the kinetics of drug release from most microparticle formulations. Except for the formula prepared at pectin/PG ratio of 2:1 (F8), all formulations exhibited a Korsmeyer–Peppas release exponent (n) >1, indicating that the drug release from microparticles followed the so-called super case-II transport kinetics (). This is a type of non-Fickian diffusion, in which the system has an increased swelling at the relaxing front, usually occurring at long periods of time, i.e. diffusion driven by slow swelling of polymer chains (Langer & Peppas, Citation1981). On the other hand, the results obtained with formula (F8) suggested anomalous (non-Fickian) transport controlled by a combination of fickian diffusion and swelling due to relaxation of polymeric chains (n = 0.88) (Ritger & Peppas, Citation1987).
Table 3. Kinetic assessment of drug release data from different Zn-pectinate/chitosan MPs according to various kinetic models.
Morphological examination of Zn-pectinate/chitosan microparticles
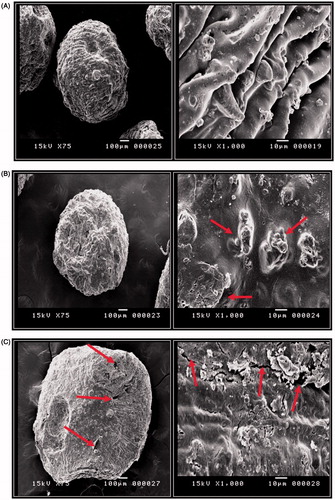
Scanning electron microscopy images show mostly spherical particles with rough and rugged surfaces (). Slight depression of one side of microparticle surface was noticed and was attributed to the drying process (). Drug crystals can be seen embedded in the microparticle matrix (indicated by arrows in ). Microparticles prepared at a cross-linking pH of 4.8 (F1) show larger and deeper surface folds, able generate channels that would promote the diffusion of biological fluids into the matrix and facilitate drug dissolution (, right panel). This might have contributed to the accelerated drug release observed from formula F1 (). In contrast, those prepared at pH 1.2 (F3) show a relatively smooth surface free from cracks and fissures (, right panel) that resisted the penetration of release media and reduced drug release rate as shown in . Microparticles prepared at lower chitosan concentration (0.1%) (F4) were slightly distorted in shape, in agreement with their higher elongation ratio (, ). Rough texture, as well as numerous surface cracks and pores could be clearly observed (indicated by arrows in , right panel). This may account for the higher drug release rate from this formula which was attributed to the lower degree of cross-linking in presence of lower chitosan concentration () (Maestrelli et al., Citation2008b).
In vivo evaluation of PG-loaded microparticles
Based on the higher drug-entrapment efficiency, acceptable mucoadhesive properties and greater ability to sustain drug release, PG-loaded microparticles (formula F3) were selected for further in vivo evaluation. The microparticles were filled into hard gelatin capsules of size 4 and orally administered to healthy rabbits. Equivalent dose of micronized PG powder was similarly treated and used as a control. The pharmacokinetic parameters obtained for the microparticles, in comparison with those of the free drug, are listed in . After oral administration of PG micronized powder, the plasma drug concentration increased progressively and achieved a peak concentration (Cmax) of 1.606 ± 0.037 µg/ml at a Tmax of 6 h. Later, it rapidly decreased and the drug was undetectable in plasma at 30 h post administration (). In contrast, the increase in plasma PG concentration after administration of the drug-loaded microparticles (formula F3) was not as sharp as that observed for the drug alone. One hour post administration, the PG plasma concentration was relatively small (2.4 ± 1 ng/ml). Thereafter, the concentration increased gradually and a Cmax of 1.319 ± 0.008 µg/ml was achieved at a Tmax of 12 h, after which it started to slowly decrease. At 30 h post administration, the PG plasma concentration observed for the microparticles was 0.469 ± 0.013 µg/ml, which was very significantly higher than that achieved for the drug alone (p < 0.001). The drug mean residence time (MRT) in the plasma for the microparticles was more than two-fold higher than that obtained for the drug alone, confirming the ability of the microparticles to achieve a sustained drug effect (). Further, the drug clearance rate for the microparticles was ∼1.5 times less than that of the drug alone (). This significant prolongation of the drug MRT is presumably attributed to the slower drug release from the microparticles, which serve as a drug reservoir. The oral bioavailability of the drug from the microparticles was highly enhanced as indicated by the significantly higher AUC0–∞ value of the microparticles compared to the free drug (p < 0.001). Thus, the relative bioavailability of formula (F3) was found to be 168.18% (). This much better performance of the drug-loaded microparticles might be attributed to their mucoadhesive properties, which increase the residence time in the GIT and hence the time available for drug dissolution and absorption. Further, drug-release studies () have confirmed that most of the microparticle-loaded drug was delivered to the colon, which might allow partial avoidance of pre-systemic drug elimination and enhance its bioavailability (Nappinnai et al., Citation2013).
Figure 8. Mean plasma concentration-time profiles of PG after oral administration of selected Zn-pectinate/chitosan microparticle formula (F3) to healthy rabbits compared to the micronized PG powder.
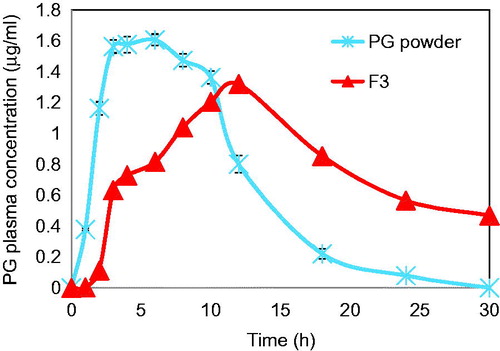
Table 4. Pharmacokinetic parameters of PG after oral administration of micronized PG powder and Zn-pectinate/chitosan microparticles (formula F3) to rabbits.
Conclusion
Colon-targeted Zn-pectinate/chitosan microparticles were successfully prepared and used to enhance the oral bioavailability of progesterone. Drug entrapment efficiency of ≥76.7% was achieved, confirming the efficacy of the preparation method. The mucoadhesion properties of the microparticles were dependent on the cross-linking solution pH and pectin concentration. Drug incorporation into the microparticles prevented its release in the stomach whereas burst release was observed at pH 6.8 followed by a sustained phase at pH 7.4. The cross-linking pH and chitosan concentration exhibited the major influences on drug release rate whereas the cross-linking time had a minimal effect. Drug-loaded microparticles had a relative oral bioavailability of ∼168% and more than twice the plasma residence time of progesterone, compared to the drug alone. Taken together, these results confirm the potential of Zn-pectinate/chitosan microparticles as an effective colon-targeted system able to enhance the oral bioavailability of hydrophobic drugs, such as progesterone or similar drugs.
Declaration of interest
The authors report no conflicts of interest in this work.
References
- Andréen L, Spigset O, Andersson A, et al. (2006). Pharmacokinetics of progesterone and its metabolites allopregnanolone and pregnanolone after oral administration of low-dose progesterone. Maturitas 54:238–44
- Araujo V, Gamboa A, Caro N, et al. (2013). Release of prednisolone and inulin from a new calcium-alginate chitosan-coated matrix system for colonic delivery. J Pharm Sci 102:2748–59
- Auriemma G, Mencherini T, Russo P, et al. (2013). Prilling for the development of multi-particulate colon drug delivery systems: pectin vs. pectin–alginate beads. Carbohydr Polym 92:367–73
- Bai XY, Yan Y, Wang L, et al. (2015). Novel pH-sensitive hydrogels for 5-aminosalicylic acid colon targeting delivery: in vivo study with ulcerative colitis targeting therapy in mice. Drug Deliv. Early Online: 1--7. doi:10.3109/10717544.2014.996924
- Bajpai SK, Sharma S. (2004). Investigation of swelling/degradation behaviour of alginate beads crosslinked with Ca2+ and Ba2+ ions. React Funct Polym 59:129–40
- Baker R, Lonsdale H. (1974). Controlled release: mechanisms and rates. In: Tanquary AC, Lacey RE. eds. Controlled release of biologically active agents. New York: Plenum Press, 15–71
- Basak P, Adhikari B. (2009). Poly (vinyl alcohol) hydrogels for pH dependent colon targeted drug delivery. J Mater Sci Mater Med 20:S137–46
- Basit AW. (2005). Advances in colonic drug delivery. Drugs 65:1991–2007
- Berggren J, Alderborn G. (2001). Effect of drying rate on porosity and tabletting behaviour of cellulose pellets. Int J Pharm 227:81–96
- Bigucci F, Luppi B, Cerchiara T, et al. (2008). Chitosan/pectin polyelectrolyte complexes: selection of suitable preparative conditions for colon-specific delivery of vancomycin. Eur J Pharm Sci 35:435–41
- Bode H, Brendel E, Ahr G, et al. (1996). Investigation of nifedipine absorption in different regions of the human gastrointestinal (GI) tract after simultaneous administration of 13C-and 12C-nifedipine. Eur J Clin Pharmacol 50:195–201
- Borges O, Borchard G, Verhoef JC, et al. (2005). Preparation of coated nanoparticles for a new mucosal vaccine delivery system. Int J Pharm 299:155–66
- Bourgeois S, Gernet M, Pradeau D, et al. (2006). Evaluation of critical formulation parameters influencing the bioactivity of β-lactamases entrapped in pectin beads. Int J Pharm 324:2–9
- Carbinatto FM, de Castro AD, Evangelista RC, Cury BSF. (2014). Insights into the swelling process and drug release mechanisms from cross-linked pectin/high amylose starch matrices. Asian J Pharm Sci 9:27–34
- Chawla V, Tiwary AK, Gupta S. (2000). Characterization of polyvinyl alcohol microspheres of diclofenac sodium: application of statistical design. Drug Dev Ind Pharm 26:675–80
- Chen H, Fan M. (2007). Chitosan/carboxymethyl cellulose polyelectrolyte complex scaffolds for pulp cells regeneration. J Bioact Compat Polym 22:475–91
- Chourasia MK, Jain SK. (2004). Polysaccharides for colon targeted drug delivery. Drug Deliv 11:129–48
- Das S, Chaudhury A, Ng K-Y. (2011). Preparation and evaluation of zinc–pectin–chitosan composite particles for drug delivery to the colon: role of chitosan in modifying in vitro and in vivo drug release. Int J Pharm 406:11–20
- Das S, Ng KY. (2010a). Impact of glutaraldehyde on in vivo colon-specific release of resveratrol from biodegradable pectin-based formulation. J Pharm Sci 99:4903–16
- Das S, Ng KY. (2010b). Resveratrol-loaded calcium-pectinate beads: effects of formulation parameters on drug release and bead characteristics. J Pharm Sci 99:840–60
- Del Curto MD, Maroni A, Foppoli A, et al. (2009). Preparation and evaluation of an oral delivery system for time-dependent colon release of insulin and selected protease inhibitor and absorption enhancer compounds. J Pharm Sci 98:4661–9
- Dollery C. (1999). Therapeutic drugs, 2nd ed. London, UK: Churchill Livingstone
- El-Gibaly I. (2002). Oral delayed-release system based on Zn-pectinate gel (ZPG) microparticles as an alternative carrier to calcium pectinate beads for colonic drug delivery. Int J Pharm 232:199–211
- Eun SS, Yang S, Choe S. (2004). Mechanism of the formation of stable microspheres by precipitation copolymerization of styrene and divinylbenzene. J Polym Sci Part A: Polym Chem 42:3967–74
- George M, Abraham TE. (2006). Polyionic hydrocolloids for the intestinal delivery of protein drugs: Alginate and chitosan—a review. J Control Release 114:1–14
- Gibaldi M. (1991). Biopharmaceutics and clinical pharmaceutics, 4th ed. Philadelphia (PA): Lea & Febiger
- Hagesaether E, Bye R, Sande SA. (2008). Ex vivo mucoadhesion of different zinc-pectinate hydrogel beads. Int J Pharm 347:9–15
- Higuchi T. (1963). Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci 52:1145–9
- Hiorth M, Versland T, Heikkilä J, et al. (2006). Immersion coating of pellets with calcium pectinate and chitosan. Int J Pharm 308:25–32
- Ishibashi T, Ikegami K, Kubo H, et al. (1999). Evaluation of colonic absorbability of drugs in dogs using a novel colon-targeted delivery capsule (CTDC). J Control Release 59:361–76
- Jeong Y-I, Ohno T, Hu Z, et al. (2001). Evaluation of an intestinal pressure-controlled colon delivery capsules prepared by a dipping method. J Control Release 71:175–82
- Jin L, Ding Y, Feng M, Cao Q. (2014). Preparation oral levofloxacin colon-specific microspheres delivery: in vitro and in vivo studies. Drug Deliv 17:1–7
- Jose S, Prema MT, Chacko AJ, et al. (2011). Colon specific chitosan microspheres for chronotherapy of chronic stable angina. Colloids Surf B Biointerfaces 83:277–83
- Klein S, Stein J, Dressman J. (2005). Site-specific delivery of anti-inflammatory drugs in the gastrointestinal tract: an in-vitro release model. J Pharm Pharmacol 57:709–19
- Krishnaiah YSR, Satyanarayana S, Prasad RYV, Rao NS. (1998). Gamma scintigraphic studies on guar gum matrix tablets for colonic drug delivery in healthy human volunteers. J Control Release 55:245–52
- Kshirsagar SJ, Bhalekar MR, Shewale NS, et al. (2011). Development of enzyme-controlled colonic drug delivery using amylose and hydroxypropyl methylcellulose: optimization by factorial design. Drug Deliv 18:385–93
- Langer R, Peppas N. (1981). Present and future applications of biomaterials in controlled drug delivery systems. Biomaterials 2:201–14
- Liu Q, Wang X-Y, Zhang H. (2007). Solvent effects on infrared spectra of progesterone in CHCl3/cyclo-C6H12 binary solvent systems. Spectrochim Acta A Mol Biomol Spectrosc 66:202–7
- Lootens D, Capel F, Durand D, et al. (2003). Influence of pH, Ca concentration, temperature and amidation on the gelation of low methoxyl pectin. Food Hydrocoll 17:237–44
- Maestrelli F, Cirri M, Corti G, et al. (2008a). Development of enteric-coated calcium pectinate microspheres intended for colonic drug delivery. Eur J Pharm Biopharm 69:508–18
- Maestrelli F, Zerrouk N, Cirri M, et al. (2008b). Microspheres for colonic delivery of ketoprofen-hydroxypropyl-β-cyclodextrin complex. Eur J Pharm Sci 34:1–11
- Marudova M, Lang S, Brownsey GJ, Ring SG. (2005). Pectin–chitosan multilayer formation. Carbohydr Res 340:2144–9
- Marudova M, MacDougall AJ, Ring SG. (2004). Pectin–chitosan interactions and gel formation. Carbohydr Res 339:1933–9
- McLeod AD, Friend DR, Tozer TN. (1994). Glucocorticoid-dextran conjugates as potential prodrugs for colon-specific delivery: hydrolysis in rat gastrointestinal tract contents. J Pharm Sci 83:1284–8
- Memişoğlu E, Bochot A, Şen M, et al. (2003). Non-surfactant nanospheres of progesterone inclusion complexes with amphiphilic β-cyclodextrins. Int J Pharm 251:143–53
- Mennini N, Furlanetto S, Cirri M, Mura P. (2012). Quality by design approach for developing chitosan-Ca-alginate microspheres for colon delivery of celecoxib-hydroxypropyl-β-cyclodextrin-PVP complex. Eur J Pharm Biopharm 80:67–75
- Mennini N, Furlanetto S, Maestrelli F, et al. (2008). Response surface methodology in the optimization of chitosan–Ca pectinate bead formulations. Eur J Pharm Sci 35:318–25
- Munjeri O, Collett JH, Fell JT. (1997). Hydrogel beads based on amidated pectins for colon-specific drug delivery: the role of chitosan in modifying drug release. J Control Release 46:273–8
- Mura P, Maestrelli F, Cirri M, et al. (2003). Development of enteric-coated pectin-based matrix tablets for colonic delivery of theophylline. J Drug Target 11:365–71
- Muraoka M, Hu Z, Shimokawa T, et al. (1998). Evaluation of intestinal pressure-controlled colon delivery capsule containing caffeine as a model drug in human volunteers. J Control Release 52:119–29
- Nappinnai M, Sivaneswari S, Swain S, et al. (2013). Formulation optimization and characterization of gastroretentive cefpodoxime proxetil mucoadhesive microspheres using 32 factorial design. J Pharm Res 7:304–9
- Oliveira GF, Ferrari PC, Carvalho LQ, Evangelista RC. (2010). Chitosan–pectin multiparticulate systems associated with enteric polymers for colonic drug delivery. Carbohydr Polym 82:1004–9
- Qureshi MJ, Ali J, Baboota S, et al. (2014). Pharmacokinetic study of a capsule-based chronomodulated drug delivery system of salbutamol sulphate in rabbits. Trop J Pharm Res 13:17–22
- Rai G, Yadav AK, Jain NK, Agrawal GP. (2014). Eudragit-coated dextran microspheres of 5-fluorouracil for site-specific delivery to colon. Drug Deliv 20:1–10
- Ramasamy T, Ruttala HB, Shanmugam S, Umadevi SK. (2013). Eudragit-coated aceclofenac-loaded pectin microspheres in chronopharmacological treatment of rheumatoid arthritis. Drug Deliv 20:65–77
- Ribeiro LNM, Alcântara ACS, Darder M, et al. (2014). Pectin-coated chitosan–LDH bionanocomposite beads as potential systems for colon-targeted drug delivery. Int J Pharm 463:1–9
- Riley S, Turnberg L. (1990). Sulphasalazine and the aminosalicylates in the treatment of inflammatory bowel disease. QJM 75:551–62
- Risbud MV, Hardikar AA, Bhat SV, Bhonde RR. (2000). pH-sensitive freeze-dried chitosan–polyvinyl pyrrolidone hydrogels as controlled release system for antibiotic delivery. J Control Release 68:23–30
- Ritger PL, Peppas NA. (1987). A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J Control Release 5:37–42
- Rubinstein A. (1995). Approaches and opportunities in colon-specific drug delivery. Crit Rev Ther Drug Carrier Syst 12:101–49
- Rubinstein A. (2005). Colonic drug delivery. Drug Discov Today Technol 2:33–7
- Sareen R, Jain N, Rajkumari A, Dhar KL. (2014). pH triggered delivery of curcumin from Eudragit-coated chitosan microspheres for inflammatory bowel disease: characterization and pharmacodynamic evaluation. Drug Deliv. Early Online: 1--8. doi:10.3109/10717544.2014.903534
- Semdé R, Amighi K, Devleeschouwer MJ, Moës AJ. (2000). Effect of pectinolytic enzymes on the theophylline release from pellets coated with water insoluble polymers containing pectin HM or calcium pectinate. Int J Pharm 197:169–79
- Sharma S, Kaur J, Sharma G, et al. (2013). Preparation and characterization of pH-responsive guar gum microspheres. Int J Biol Macromol 62:636–41
- Simonoska Crcarevska M, Glavas Dodov M, Goracinova K. (2008). Chitosan coated Ca–alginate microparticles loaded with budesonide for delivery to the inflamed colonic mucosa. Eur J Pharm Biopharm 68:565–78
- Sinha VR, Kumria R. (2003). Microbially triggered drug delivery to the colon. Eur J Pharm Sci 18:3–18
- Sinha VR, Singla AK, Wadhawan S, et al. (2004). Chitosan microspheres as a potential carrier for drugs. Int J Pharm 274:1–33
- Sood A, Panchagnula R. (1998). Drug release evaluation of diltiazem CR preparations. Int J Pharm 175:95–107
- Steed K, Hooper G, Monti N, et al. (1997). The use of pharmacoscintigraphy to focus the development strategy for a novel 5-ASA colon targeting system (“TIME CLOCK®” system). J Control Release 49:115–22
- Sweetman SC. (2009). Martindale: The complete drug reference, 36th ed. London: Pharmaceutical press
- Thirawong N, Nunthanid J, Puttipipatkhachorn S, Sriamornsak P. (2007). Mucoadhesive properties of various pectins on gastrointestinal mucosa: an in vitro evaluation using texture analyzer. Eur J Pharm Biopharm 67:132–40
- Varshosaz J, Dehkordi JA, Golafshan S. (2006). Colon-specific delivery of mesalazine chitosan microspheres. J Microencapsul 23:329–39
- Vervoort L, Rombaut P, Van den Mooter G, et al. (1998). Inulin hydrogels. II. In vitro degradation study. Int J Pharm 172:137–45
- Wan S, Sun Y, Sun L, Tan F. (2012). Chitosan microparticles for oral bioavailability improvement of the hydrophobic drug curcumin. Pharmazie 67:525–8
- Wei X, Lu Y, Qi J, et al. (2014). An in situ crosslinked compression coat comprised of pectin and calcium chloride for colon-specific delivery of indomethacin. Drug Deliv. Early Online: 1--8. doi:10.3109/10717544.2013.879965
- Wittaya-areekul S, Kruenate J, Prahsarn C. (2006). Preparation and in vitro evaluation of mucoadhesive properties of alginate/chitosan microparticles containing prednisolone. Int J Pharm 312:113–18
- Wu Z, Zhang C, Yang C, et al. (2000). Simultaneous quantitative determination of norgestrel and progesterone in human serum by high-performance liquid chromatography-tandem mass spectrometry with atmospheric pressure chemical ionization. Analyst 125:2201–5
- Xi MM, Zhang SQ, Wang XY, et al. (2005). Study on the characteristics of pectin–ketoprofen for colon targeting in rats. Int J Pharm 298:91–7
- Yang L, Chu JS, Fix JA. (2002). Colon-specific drug delivery: new approaches and in vitro/in vivo evaluation. Int J Pharm 235:1–15
- Yu C-Y, Yin B-C, Zhang W, et al. (2009). Composite microparticle drug delivery systems based on chitosan, alginate and pectin with improved pH-sensitive drug release property. Colloids Surf B Biointerfaces 68:245–9