Abstract
The purpose of this study was to investigate the potential of cubic liquid crystalline nanoparticles for ocular delivery of tropicamide. Ultrasound-assisted fragmentation of cubic liquid crystalline bulk phases resulted in cubic liquid crystalline nanoparticles employing Pluronic F127 as dispersant. The effects of process variables such as sonication time, sonication amplitude, sonication depth, and pre-mixing time on particle size and polydispersity index was investigated using central composite design. The morphology of tropicamide-loaded nanoparticles was found to be nearly cubical in shape by transmission electron microscopy observation. Further, small angle X-ray scattering experiment confirmed the presence of D and P phase cubic structures in coexistence. The optimized tropicamide-loaded cubic nanoparticles showed in vitro corneal permeation of tropicamide across isolated porcine cornea comparable to its commercial preparation, Tropicacyl®. Ocular tolerance was evaluated by Hen’s egg–chorioallantoic membrane test and histological studies. The results of in vivo mydriatic response study demonstrated a remarkably higher area under mydriatic response curve (AUC0→1440 min) values of cubic nanoparticles over Tropicacyl® indicating better therapeutic value of cubic nanoparticles. Furthermore, tropicamide-loaded cubic nanoparticles exhibited prolonged mydriatic effect on rabbits as compared to commercial conventional aqueous ophthalmic solution.
Introduction
Ocular drug delivery is one of the most challenging problem facing pharmaceutical researchers due to the unique anatomy, physiology, and biochemistry of eye. It is reported that effectively 5% or even less of the instilled dose reaches intraocular tissues (Ahmed Citation2003; Zhang et al., Citation2004; Ali et al., Citation2014). Most of the topically instilled drugs drain away quickly from the surfaces of the eyes by various mechanisms, such as blinking, lacrimation, tear dilution, and tear turnover. In addition, different layers of cornea, conjunctiva, and sclera constitute a compact barrier for ocular drug delivery (Gaudana et al., Citation2010). Several novel formulation-based approaches such as emulsions, nanoparticles, and liposomes are being explored for their potential as ocular drug delivery vehicle to overcome the ocular barriers by improving the transcorneal permeation (Law et al., Citation2000; Lallemand et al., Citation2012; Dilbaghi et al., Citation2013; De Sa et al., Citation2015). Cubic liquid crystalline nanoparticles have gained significant scientific interest recently. These are self-assembled bicontinuous liquid crystalline phases comprising lipid bilayer enclosing water channels providing both hydrophilic and hydrophobic region for encapsulation of drugs with varying solubility (Siekmann et al., Citation2002). Cubic liquid crystalline nanoparticles can be easily administered in a liquid form and seemed to have high diffusivity across the corneal epithelium (Gan et al., Citation2010; Li et al., Citation2013). In addition, prolonged drug release, bio-adhesion, biocompatibility, and biodegradability makes these drug delivery systems interesting as new therapeutic tools in ocular delivery (Engstrom et al., Citation1995; Spicer et al., Citation2001; Dian et al., Citation2013). Despite the astounding properties of cubic nanoparticles as innovative drug carriers, little research has so far been conducted on evaluating the potential of cubic nanoparticles for ophthalmic drug delivery (Gan et al., Citation2010; Li et al., Citation2013). The aim of this work was to study the performance of cubic nanoparticles as innovative ocular delivery systems for tropicamide, a mydriatic agent, chosen as a model drug. Tropicamide is a nonselective antimuscarinic agent which is used for eye examinations before and after eye surgery, cycloplegic retinoscopy, dilated funduscopic exam, cycloplegia, etc. Tropicamide blocks the receptor in the muscles of the eye that control the size of pupil and lens shape, thereby, producing dilation of pupil, i.e. mydriasis.
Several methods have been reported for production of cubic nanoparticles (Boyd Citation2003; Barauskas et al., Citation2005; Bei et al., Citation2009a). However, top down techniques is being most frequently employed as it can produce nanoparticles by consuming less time and energy and more economic (Bei et al., Citation2009b). Recently it has been reported that ultrasonic condition such as duration of treatment and amplitude influences the particle size and poly dispersity index of nanoemulsion (Tang et al., Citation2012). Furthermore, it has also been reported that position of ultrasound source strongly affects the particle size of lipid-based vesicles (Silva et al., Citation2010). Ultrasound-assisted production of cubic nanoparticles has been extensively reported in the literature (Ferreira et al., Citation2006; Chong et al., Citation2012; Driever et al., Citation2013; Verma & Ahuja Citation2015). However, ultrasonic set up is poorly described resulting in non-reproducibility of this methodology. Therefore, this study was undertaken to optimize operating parameters of probe-type sonicator to engineer the characteristics of cubic liquid crystalline nanoparticles. The influence of ultrasonic processing parameters, namely pre-mixing time, ultrasonic amplitude, sonication depth, and sonication time on particle size and polydispersity index (PdI) of cubic liquid crystalline nanoparticles was studied using response surface methodology. The optimized batch of cubic nanoparticles was evaluated for ocular drug delivery by loading with tropicamide. The crystalline structure and morphology of tropicamide-loaded cubic nanoparticles was investigated by small angle X-ray scattering and transmission electron microscopy (TEM), respectively. Further, the tropicamide-loaded cubic nanoparticles were evaluated comparatively with commercial conventional formulation of tropicamide for in vitro corneal permeation characteristics using isolated porcine cornea and for in vivo mydriatic response using rabbits.
Material and methods
Chemicals and reagents
Monoolein (RYLO MG 19, monoglycerides content > 95%) was a generous gift from Danisco Cultor (Grinsted, Denmark). Tropicamide was obtained as gift sample from Optica Pharmaceuticals (Yamunanagar, India). Pluronic F127 (PEO98-polyPPO67-PEO98) was obtained from Ranbaxy Research Laboratory (Gurgaon, India). Sephadex® G-50 was procured from Sigma-Aldrich (St. Louis, MO). Commercial formulation of eye drop (Tropicacyl®, Sunways Pvt. Ltd. Mumbai, India) was purchased from local pharmacy (Hisar, India). All other chemicals used were of analytical grade.
Experimental setup
The model of probe sonicator that had been used for the present study is: Q55 (Qsonica sonicators, Newtown, CT). It can deliver a power output maximum of 55 W. The amplitude can be varied from 10% to 100%. The probe tip was made up of titanium. The sample holder was an open glass test tube having height and inner diameter 120 mm and 14 mm, respectively, which contained 10 ml of the sample. The test tube was held straight in a water bath maintained at 25 °C using water bath. The probe tip was set at the center of test tube. Time of sonication was controlled by stop watch.
Preparation of cubic liquid crystalline nanoparticles
The cubic liquid crystalline nanoparticles were prepared by top down technique (Guo et al., Citation2010). Briefly, water was added to molten monoolein (4.5%w/v) in the sample holder and kept at room temperature until transparent cubic phase gel was formed. Thereafter, aqueous solution of Pluronics F127 (0.5%w/v) was added and pre-mixing was carried out as given in design protocol using a vortex mixer (Cyclo Mixer VM102, Remi Equipments, Mumbai, India) to form coarse dispersion. The resultant coarse dispersion was subjected to probe sonication as per the parameters specified in design protocol using ultrasonic processor to give cubic liquid crystalline nanoparticles.
Experimental design
The central composite design (CCD) (with α = 1) was employed to find out optimum process parameters. The independent variables selected were: sonication time (X1), amplitude (X2), sonication depth (X3), and premixing time (X4). Each factor was studied at 3 levels (i.e. −1, 0, +1). The dependent variables chosen were: particle size (Y1) and PdI (Y2). The central point (0, 0, 0) was studied in sextet. The total design matrix showed 30 runs (), to be carried out randomly to eliminate the effects of extraneous or nuisance variables. Statistical analysis was performed using Design Expert software (Version 7.0.0, Stat-ease Inc., Minneapolis, MN). Analysis of variance (ANOVA) was carried out to predict whether there are significant differences between independent variables or not.
Table 1. Central composite design used to study effect of process variables on particle size and PdI and results of zeta potential.
Particle size, zeta potential, and polydispersity index measurement
The average particle size (Z-average) and PdI was measured using Zetasizer nano ZS (Malvern Instruments, Malvern, UK). All the measurements were performed in triplicate.
Preparation of tropicamide-loaded ophthalmic cubic nanoformulation
The optimized batch of cubic nanoparticles prepared by ultrasonic fragmentation was evaluated for ocular drug delivery using tropicamide as a model drug. Required quantity of tropicamide was added to water and pH was adjusted to 4 for complete dissolution of tropicamide (Kumar & Ahuja Citation2014). The aqueous solution of tropicamide was then sterilized by autoclaving at 121 °C, for 15 min, at 15 lbs/in2. This sterilized solution was added to molten monoolein under aseptic conditions to form bulk phase gel, followed by pre-mixing and probesonication to get tropicamide-loaded ophthalmic cubic nanoformulation.
Characterization of tropicamide-loaded ophthalmic cubic nanoformulation
Particle size, zeta potential, and polydispersity index measurement
The Z-average and PdI was measured using Zetasizer nano ZS (Malvern Instruments, Malvern, UK). All the measurements were performed in triplicate.
Encapsulation efficiency
Gel filtration chromatographic technique was employed to separate unentrapped tropicamide from the cubic nanoparticles (Verma & Ahuja Citation2015). Sephadex G-50 powder was filled into the column and water was added into the column in order to form sephadex gel avoiding bubbles and cracks. An aliquot of 1 ml of the tropicamide-loaded cubic nanoformulation was passed through a column of sephadex with specific length and inner diameter of 6.5 cm and 2.3 cm, respectively. The column contained 27.01 ml of gel. The eluate was analyzed for the contents of free tropicamide by UV spectrophotometric analysis at λmax of 257 nm. The percent encapsulation of tropicamide into nanoparticles was calculated with the formula:
(1)
where, Trt is the total tropicamide in formulation and Tre is the amount of tropicamide found in eluant aqueous phase.
Transmission electron microscopy
The morphology of tropicamide-loaded cubic nanoformulation was observed by TEM on a Philips CM 10 electron microscope operating at an accelerated voltage of 100 kV.
Small angle X-ray scattering analysis
SAXS measurements were used to identify the inner structure of tropicamide-loaded cubic nanoformulation. SAXS diffractograms were acquired on a SAXSess mc2 (Anton Paar, Austria) equipped with a line collimation set-up. The experiments were conducted using Ni-filtered Cu Kα radiations (1.542 Å) from a copper rotating anode operating at 45 kV and 50 mA. The samples were filled into a 1 mm quartz capillary in a steel sample holder and the sample to detector distance was 261 mm. Measurements were carried out at 25 ± 0.5 °C with exposure time of 3 h for nanoparticles. In order to reduce the noise and to cancel out the effect of LC monodomains contribution in the final SAXS diffractograms, the capillaries were slightly rotated every 10 min. All scattering signals were treated with SAXSquant software (Anton Paar).
Evaluation for ocular drug delivery
In vitro corneal permeation study
Corneal permeation characteristics of tropicamide-loaded cubic ophthalmic nanoformulation were comparatively evaluated with the commercially available conventional ophthalmic preparation (Tropicacyl 1%, (w/v)) using isolated porcine eyes cornea as model (Gratieri et al., Citation2011). Fresh whole eyeballs were obtained from the local butcher shop immediately after slaughtering and transported to the laboratory in cold normal saline within an hour. Cornea was carefully excised along with 2–4 mm of scleral tissue and finely cleaned and washed till free from proteins with cold normal saline. Isolated cornea was mounted by clamping between the donor and receptor compartments of modified Franz diffusion cell, with endothelial side facing the receptor and epithelial side facing the donor. The receptor compartment contained 11.5 ml of freshly prepared phosphate-buffered saline (PBS) pH 7.4 maintained at 35 ± 0.5 °C under magnetic stirring. Area available for corneal permeation was 0.785 cm2. The test formulation (1 ml) was placed in the donor compartment over the cornea. An aliquot of 1 ml of the sample was withdrawn from receptor compartment at fixed time intervals and analyzed for the contents of tropicamide. The study was conducted using paired corneas, i.e. one cornea of the animal was used for the permeation study of cubic nanoparticles and the contra lateral cornea was used for conventional commercial formulation of aqueous drug solution. Corneal hydration level was determined by removing the scleral tissue from the cornea at the end of experiment and weighing followed by dehydration by overnight soaking in methanol and drying in an oven at 90 °C and weighing again.
Determination of tropicamide flux and permeability
The cumulative amount of tropicamide permeating across the porcine cornea was plotted against time and slope of the linear portion of the graph was calculated. The steady state flux (Js, μg/h/cm2) and apparent permeability coefficient (Kp, cm/h) were calculated as follows (Peltola et al., Citation2003):
(2)
(3)
where dQ/dt is the linear portion of the slope (µg/h), A is the corneal surface area (in this study, 0.785 cm2), and C0 is the initial drug concentration (µg/ml).
Ocular tolerance
Ocular irritancy potential of tropicamide-loaded cubic ocular nanoformulation was assessed employing Hen’s Egg Test Chorioallantoic Membrane (HET-CAM). HET-CAM study is established alternative technique to the Draize rabbit eye test to check potential irritation effects in the eye (Luepke Citation1985). Ten-day-old fertilized hen’s eggs were procured from a poultry farm. The eggs were placed in a stand with the equatorial side up where a small window was opened to expose the CAM. Only eggs with an air sac and live embryo were used for further testing. An aliquot of 0.5 ml of the test samples were placed directly onto the CAM’s surface and CAM was observed for 5 min for appearance of any of the following phenomena: hemorrhage, vasoconstriction, and coagulation for which a score was calculated (Dehelean et al., Citation2011; Kaur et al., Citation2012). Saline solution and a 0.1 N sodium hydroxide solution were used as the negative and positive controls, respectively.
In vivo mydriatic activity
Tropicamide produces a rapid mydriatic response and therefore bioavailability was assessed by measuring pupil diameter (Kumar & Ahuja Citation2014). The protocol for in vivo mydriatic study in rabbit was designed and an approval of institutional animal ethics committee was obtained. All tests were carried out on non-anesthetized Albino rabbits procured from disease free small animal house of Lala Lajpat Rai University of Veterinary and Animal Sciences (Hisar, India). Three albino rabbits with equivalent pupil light response were used in the study. Each rabbit was acclimatized to the laboratory testing conditions for 1 h prior to initiating the study. The animals were positioned in restraining boxes in the normal upright position in a room with constant light intensity and devoid of distractions. Baseline pupil diameter measurements were taken every minute for 5 min prior to dosing. One drop of the cubic nanoformulation was carefully instilled into the lower cul-de-sac region of the right eye whereas one drop of Tropicacyl® was instilled into the left eye of each rabbit. After 15 min the second dose was instilled followed by third dose after 30 min. At appropriate time intervals pupil diameter measurements. The % increment in pupil diameter was calculated as follows:
(4)
where, Io is average baseline diameter and It the pupil diameter at time t.
Effect on corneal integrity
Freshly excised cornea was incubated at 37 °C for 1 h with the formulation employing PBS pH 7.4 and sodium dodecyl sulfate (SDS) aqueous solution (0.1% w/v) as negative and positive control, respectively. After incubation corneas were washed with PBS and immediately fixed with a formaline solution 8% (w/w). The material was dehydrated with an alcohol gradient, put in melted paraffin, and solidified in block form. Cross sections (<1 µm) were cut, stained with hematoxylin and eosine (H&E), blinded and microscopically observed (Baydoun et al., Citation2004). Fresh cornea was also treated by same staining procedure and observed under light microscope for comparison purpose.
Results and discussion
Preparation and optimization of cubic nanoparticles
Monoolein forms stiff and highly viscous bulk cubic phase gel when added to water. In order to produce nanoparticles, it is desIDRDle to fragment the highly viscous cubic phase gel using numerous dispersive techniques. It has been reported by Bei and coworkers (Bei et al., Citation2009a) that characteristics of cubic nanoparticles prepared employing high speed homogenization, greatly vary with operating parameters such as homogenization speed and time.
Ultrasonic sound-assisted production of nanoparticles has been widely employed in recent years. Ultrasonic is defined as sound intensity (above 18 Hz) exceeding the threshold of human hearing. The production of ultrasound entails elastic deformation of ferroelectric materials within a high frequency electrical field (Kuttruff Citation1988; Raichel Citation2000). It has been reported that ultrasound processing parameters strongly affect the size of nanoparticles (Floris et al., Citation2013). The present work is intended to provide optimum sonication conditions in order to formulate optimal monoolein-based cubic liquid crystalline nanoparticles. The effect of process variables such as, sonication time (X1) and sonication amplitude (X2), sonication depth (X3), and pre-mixing time (X4) on particle size (Y1) and PdI (Y2) of cubic liquid crystalline nanoparticles was studied employing central composite experimental design. The preliminary studies were conducted in order to determine the levels of independent variables.
Response surface methodology is a collection of mathematical models such as central composite design (CCD), Box–Behnken design and three-level factorial design having different properties and characteristics. Amongst these three methodologies, CCD is a most popular technique that allow optimization, estimation, and evaluation of the main and interaction of variables with least number of experiments (Hao et al., Citation2012). Here, in accordance with design matrix of CCD, a total of 30 runs were required for predicting the variation of the Z-average (Y1) and PdI (Y2) as a function of the process variables which are: sonication time (X1), sonication amplitude (X2), sonication depth (X3) and pre-mixing time (X4). The combination of process parameters in random order and the experimental obtained values of particle size and zeta potential are presented in .
The obtained values of responses was fitted into various polynomial models and subjected to multiple regression analysis. The response Y2 fitted best into response surface reduced quadratic model for (p value is 0.0304) whereas response Y1 fitted best into reduced quadratic model (p value is 0.0012) with inverse square root transformation. The regression equations (Equations Equation5(5) and Equation6
(6) ) in terms of coded values of factors are as follows:
(5)
(6)
The polynomial equations showed above presents the quantitative effects of process variables and their interaction effects on the responses Y1 and Y2. The polynomial equations comprise coefficients for intercept, first order main effect (i.e. X1), interaction terms (i.e. X1X4) and higher order effects (i.e. ). The sign of coefficients indicates positive or negative effects on the response. Positive sign shows synergistic effect indicating the increase in response at higher level of independent variable. The negative coefficient presents antagonistic effect showing increase in response at low levels of independent variable. The magnitude of main effects indicates the relative impact of each factor on the response.
The magnitude of actual factor coefficient and p value for both responses is shown in . Generally, model terms is considered significant when p value <0.05 while p value greater than 0.1000 indicates the insignificant model terms (Korbahti & Tanyolac Citation2008; Bashir et al., Citation2010). As it can be seen from table that response Y1 is affected significantly (p value < 0.05) by linear and quadratic contribution of X1 and X2 whereas, response Y2 was influenced by linear contribution of X1, X2, and X3 and quadratic contribution of X4.
Table 2. Summary of each factor effect and p values, for responses Y1 and Y2.
The model F value was found to be 59.97 and 26.44 respectively, for Y1 and Y2, which implies that the model is significant. There is only a 0.01% chance that a model F value could occur due to noise. The magnitude of lack of fit F value for Y1 and Y2 was 1.54 and 1.76, respectively, indicating that the “Lack of Fit” is not statistically significant relative to the pure error which further ensures the reliability of model. As it can be seen that there is a 33.37% and 27.66% chance for Y1 and Y2, respectively that this F value could occur due to noise. The magnitude of R2, correlation coefficient was 0.9259 (Y1) and 0.8938 (Y2) shows a good correlation between experimental and predicted values. Further, predicted R2 (0.8795 and 0.7502 for Y1 and Y2, respectively) was in reasonable agreement with the adjusted R2 (9.105 and 0.8600 for Y1 and Y2, respectively). Adequate precision is given by signal to noise ratio. The values of adequate precision were higher than 4 (25.096 and 18.923 for Y1 and Y2, respectively) indicating an adequate signal. Therefore, the model could be used to navigate the design space.
and portray the 3-D response surface plots prepared using model generated by response surface methodology. displays the combined effect of sonication depth and sonication amplitude on particle size. The plot shows the curvilinear relationship between the independent and dependent variables. Notably, when sonication amplitude was increased, significant decrease in particle size was observed. Furthermore, it can be inferred from the plot that at low levels of sonication amplitude, decreasing the sonication depth led to slight decrease in the particle size. However, effect of decrease in size by decreasing depth was diminished at higher amplitude levels. shows the combined effect of pre-mixing time and sonication time on particle size. It is evident from the plot that the effect of sonication time is more prominent than the pre-mixing time. Furthermore, increasing sonication time resulted in significant decrease in the particle size of nanoparticles whereas pre-mixing time did not result in any change in particle size. This might be explained by the fact that mechanical energy of vortex shaking is insufficient to aid dispersion of nanoparticles during pre-mixing. It is well known that ultrasonic waves generate gas bubbles in liquids called cavitation. The formation of the cavitation due to application of high intensity ultrasound leads to intense mechanical forces in liquids (Karaman et al., Citation2012). High irradiation power exerts greater shear forces within the dispersion thereby, increasing number of events and the cavity collapse intensity of cavitation. The decrease in particle size could be ascribed to increase in cavitation effect with increase in sonication amplitude (Silva et al., Citation2010; Jadhav et al., Citation2015). Similarly, the decrease in size on increasing sonication time can be attributed to higher shearing with increase in exposure time. These results are contradictory to the previous investigation by Tang and coworkers (Bei et al., Citation2009b) which reported the increase in droplet size and PdI of Cremophore EL-based nanoemulsion with increase in sonication amplitude and irradiation time.
Figure 1. 3-D response surface plot showing the interaction effect for particle size as a function of (a) sonication amplitude and depth (b) sonication time and pre-mixing time.
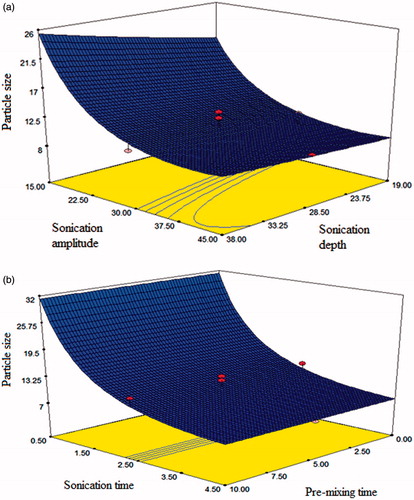
Figure 2. 3-D response surface plot showing the interaction effect for PdI as a function of (a) sonication amplitude and depth (b) sonication time and pre-mixing time.
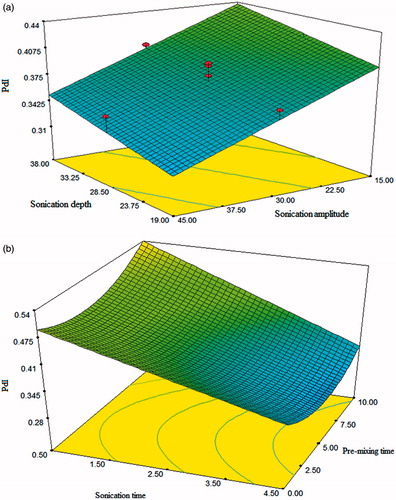
displays the combined effect of sonication depth and sonication amplitude on PdI. The plot depicts the linear relationship between the independent and dependent variables. It can be elicited that increases in sonication amplitude led to significant decrease in PdI values. Decreasing the sonication depth was found to decrease the PdI value which was more pronounced at lower amplitude values. The effect of pre-mixing time and sonication time on PdI is presented in . Pre-mixing time has shown curvilinear relationship PdI while sonication time has shown a linear functionality. As can be seen from plot, in the middle value pre-mixing time, magnitude of PdI is least. However, reason could not be understood. Furthermore, PdI value was decreased with increase in sonication time. Cubic liquid crystalline nanoparticles comprising uniform size distribution could be prepared at higher level of amplitude and time whereas at low level of sonication depth.
To develop liquid crystalline nanoparticles with desired parameters, numerical optimization tool along with desIDRDility approach was employed. The optimization was performed setting constraints for particle size and PdI to be minimum as the goal to locate the optimum values of independent variables. The Design Expert software provided us with 21 solutions; out of this one with highest desIDRDility was selected for preparing optimized formulation. The optimal calculated parameters obtained were: sonication time (4.5 min), sonication amplitude (40%), sonication depth (19 mm), and pre-mixing time (4.5 min).
To check the reliability of developed mathematical models, the response of the optimal sonication conditions and three additional checkpoint formulations covering the entire range of experimental domain was recorded. For each of the test runs, comparative analysis was performed for experimentally observed response and the response predicted by the mathematical model. displays the composition of optimum and random check points, their observed and predicted response variables and calculated percentage prediction error. The correlation between observed and predicted values was observed to be linear with correlation coefficient, R2 of 0.988 and 0.983 for particle size and PdI, respectively, confirming the model validity. The lower magnitude of percentage prediction error (−0.415 to −0.040 for Y1, −0.097 to +0.054 for Y2) is indicative of robustness of applied mathematical model and high prognostic ability of response surface methodology.
Table 3. The observed predicted values and % prediction error at check points for the responses Y1 and Y2.
Characterization of cubic nanoparticles
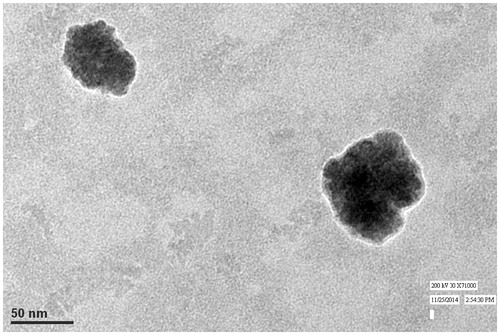
The zeta potential of the blank liquid crystalline nanoparticles was found to be in the range of −16.8 to −2.64 mV as shown in . Nonionic monoolein releases subtle amount of oleic acid which is anionic resulting in anionic behavior of dispersion. Blank liquid crystalline nanoparticles were loaded with tropicamide and studied for ocular drug delivery applications. The % drug loading was found to be 96.57 ± 0.23 (%) which could be attributed to high internal area of cubic phase nanoparticles. The particle size, PdI, and zeta potential of tropicamide-loaded cubic nanoformulation was found to be 54.52 ± 1.12 nm, 0.248 ± 0.05, and − 8.76 ± 0.82 mV, respectively. It can be seen from the results that drug loading led to increase in average size of the nanoparticles while PdI remained unaffected. Although, the absolute value of zeta potential was very low but no aggregation and visual sign of instability was observed in nanoparticles. It is presumed that the hydrophobic polypropylene oxide chain adsorbs to the surface of cubic nanoparticles leading to adhesion of polymer to nanoparticles while hydrophilic polyethylene oxide chain extend out into aqueous environment in order to provide steric shielding, thereby, preventing aggregation (Lancelot et al., Citation2014). The transmission electron micrograph of tropicamide-loaded cubic nanoparticles is shown in . It can be observed from the figure that the particles are cubical in shape.
SAXS is considered most excellent tool for structural determination of cubic liquid crystalline phases. The overlay of SAXS diffraction profile of blank cubic nanoparticles and tropicamide-loaded cubic nanoparticles are shown in . The scattering curve of blank cubic nanoparticles prepared at optimal process conditions has shown two set of medium intensity peaks in the characteristics ratio of; (
was scarcely observed) and
, confirming the presence of dispersed cubic phase particles of Im3m (P-type phase) and Pn3m (D-type phase) symmetry (). Phase diagram of monoolein/water system exhibits mainly cubic phase of Pn3m symmetry (D-type phase) in excess of water (concentration above 40%w/w) (Hyde et al., Citation1984; Qiu & Caffrey Citation2000). It is reported that Pluronic F127 causes swelling in cubic lattice thereby leading to the transition from D phase to P phase (Landh Citation1994). Possibly, transition in the phases is due to the embedment of hydrophobic polypropylene oxide chain of Pluronic F127 into bilayer of monoolein-based cubic phases (Dong et al., Citation2006). The non-uniform distribution of Pluronic F127 into cubic nanoparticles gave rise to formation of two distinct structures: one without Pluronic F127, D phase and another with Pluronic F127, P phase. Phase coexistence in dispersion carrying monoolein and Pluronic F127 in excess water has already been reported (Nakano et al., Citation2002). The scattering curve for tropicamide-loaded cubic nanoparticles was without any remarkable difference when compared to the SAXS curve of blank nanoparticles. The presence of Braggs peaks at the spacing ratios (
and
) consistent with the formation of P- and D-phases in tropicamide-loaded cubic nanoparticles indicating that tropicamide did not produce any phase transition in cubic nanoparticles. illustrate the fit of “s” values to the corresponding Miller indices (h2 + k2 + l2)1/2 (where h, k, and l are the miller indices) identified for both phases in the blank cubic nanoparticles and tropicamide-loaded cubic nanoparticles. The linearity of the plots and the (0, 0) intercept are the indications of valid space group assignment. The value of cubic cell lattice parameter (a) was calculated from the slope of the plots (Rodriguez & Kunieda Citation2000). Tropicamide incorporation did not alter the unit cell dimension because no difference in lattice parameter was observed in the presence and absence of tropicamide in cubic phases. Bonnet ratio (Br) was also determined, given by the ratio of lattice parameter of two different cubic phases (as shown in Equation (Equation7
(7) ) with periodic minimal surface structures.
(7)
Figure 4. SAXS results for tropicamide-loaded cubic nanoformulation. (a) Overlay showing scattering curve of cubic nanoparticles and tropicamide-loaded cubic nanoparticles. (b) Data fitting graphs of blank cubic nanoparticles. (c) Data fitting graphs of tropicamide-loaded cubic nanoparticles.
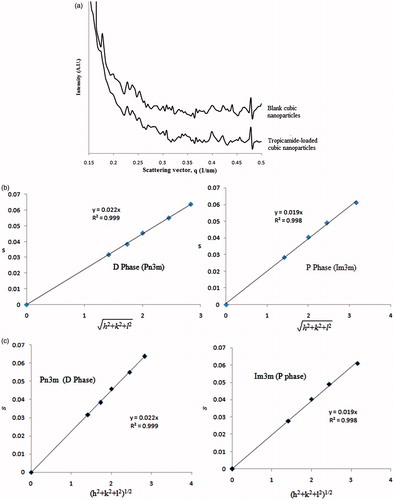
Table 4. SAXS data for blank cubic nanoparticles and tropicamide-loaded cubic nanoparticles.
The Bonnet ratio was also remained unaffected and calculated to be 1.16 which is close to the theoretically predicted value of 1.279 for both blank cubic nanoparticles and tropicamide-loaded cubic nanoparticles (Muller et al., Citation2010).
Evaluation as ocular drug delivery vehicle
presents the results of in vitro corneal permeation profile of tropicamide from the cubic nanoparticles and commercial ophthalmic solution (Tropicacyl®) across the isolated porcine cornea. It can be seen that permeation characteristics of tropicamide-loaded cubic nanoparticles are comparable to that of conventional commercial preparation (Tropicacyl®). Furthermore, no remarkable difference in values of flux (Jss) and apparent permeability (Kp) were seen as listed in . The commercial formulation, which was used for comparative evaluation contained chlorbutol as a preservative. It is reported that chlorbutol enhances the corneal permeability (Dilbaghi et al., Citation2013). Even though, no preservative was added in the cubic nanoparticles, it provided % permeation of tropicamide comparable to the commercial formulation. This could be attributed to the nanometric size of the cubic nanoparticles. Earlier it was reported that nanoparticles provide higher corneal permeation due to endocytic uptake (Calvo et al., Citation1996; Gupta et al., Citation2000). In addition, lipid monoolein have also been reported to provide corneal penetration-enhancing effect (Li et al., Citation2013). The corneal hydration levels are indicative of corneal integrity and its normal levels lies in range of 75–80% (Maurice et al., Citation1970). Since, the corneal hydration levels in the present study are within limits (), the integrity of corneal epithelium and endothelium was not compromised by formulation tested.
Figure 5. In vitro corneal permeation profile tropicamide-loaded cubic nanoformulation and commercially available conventional aqueous preparation of tropicamide (Tropicacyl®).
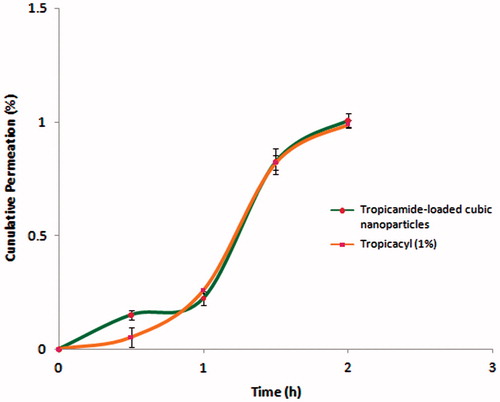
Table 5. Steady state flux (Jss) and permeability (Kp) of tropicamide through porcine cornea and corneal hydration level.
The toxicological and ocular irritation of the cubic nanoparticles was evaluated employing the HET-CAM study. The study depends on individual interpretations of tissue reactions. Severe coagulation was observed in positive control, i.e. 0.1 N NaOH (positive control) and no reaction in case of 0.9% NaCl (negative control). The values of potential irritation scores were calculated to be 0.85 and 11.12 for tropicamide-loaded cubic nanoparticles and 0.1 N NaOH, respectively. The tropicamide containing cubic nanoparticles were found to be nonirritant as the potential irritation score was less than 1 (Dehelean et al., Citation2011).
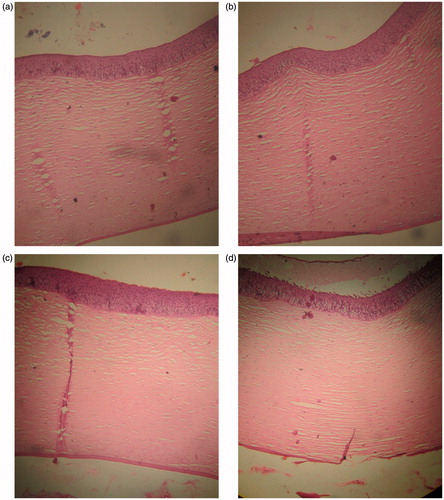
Further, the effect of formulation on corneal cell structure and tissue integrity was determined by histochemical studies. The cross section of fresh cornea was compared with cornea incubated with cubic nanoparticles, PBS (pH 7.4) and SDS (). The corneal cells and tissues appeared to be deformed in cornea exposed to SDS (positive control) when compared to the cross section of fresh cornea. Intercellular space widening and separation of superficial epithelium layer was also observed. On the other hand, epithelium and stroma structure were found to be well maintained in corneal tissues exposed to PBS pH 7.4 (negative control) and cubic nanoparticles as observed in non-treated eye. The results showed a good corneal biocompatibility corroborating the results of HET-CAM study and therefore, demonstrated a preferable ocular tolerance.
Figure 6. Histological section of (a) non-treated, (b) tropicamide-loaded cubic nanoformulation-treated, (c) phosphate-buffered saline pH 7.4 (negative control)-treated, and (d) SDS (positive control)-treated corneas.
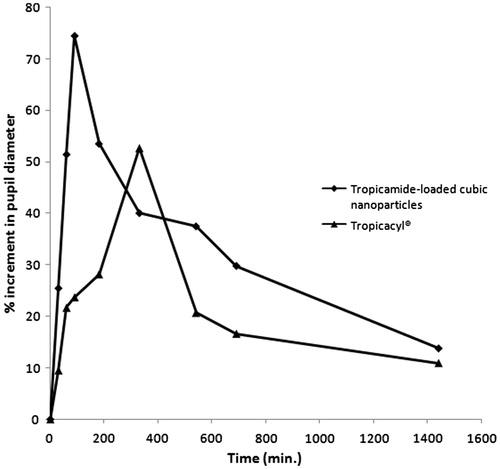
Optimized tropicamide-loaded nanoparticles were evaluated for their therapeutic performance by measuring in vivo mydriatic response in rabbits. The plot between % increments in pupillary diameter of eye as the function of time is exemplified in . Both formulations showed increase in pupillary response up to a maxima then decrease in response was observed. It can be observed from the results that it took 90 min to achieve maximum pupillary response in case of tropicamide-loaded cubic nanoparticles while Tropicacyl® elicited maximum response in 330 min. The reason for which could be attributed to rapid absorption of nanoparticles through ocular tissues. Further, the values of maximum increment (%) in pupillary diameter were observed to be 74.52 ± 2.11% and 52.64 ± 1.18% for tropicamide cubic nanoparticles and commercial preparation respectively, revealing higher therapeutic efficiency of nanoparticles in vivo as compared to Tropicacyl®. Area under mydriatic response curve (AUC0→1440 min) was remarkably higher for tropicamide-loaded cubic nanoparticles than Tropicacyl® (45 752.52%.min and 30 514.09%.min for tropicamide-loaded cubic nanoparticles and Tropicacyl®, respectively). Further results obtained were suggestive of the prolonged mydriatic response of cubic nanoparticles as compared to Tropicacyl®. It is presumed that lipid bilayer in cubic nanoparticles may serve to prolong contact time with ocular tissues. In addition, nonspecific interactions (hydrophobic and van der Waals) of the cubic nanoparticles with the superficial oily layer of the tear film could be another possible explanation for this finding (Alany et al., Citation2006). Cubic liquid crystalline nanoparticles are therefore a promising effective system for delivery and prolonged release of tropicamide with reduced ocular toxicity, to pursue a better patient compliance.
Conclusions
Ultrasound-assisted preparation of cubic nanoparticles was optimized employing central composite experimental design. The optimized parameters were used for preparing tropicamide-loaded ophthalmic cubic nanoformulation. Tropicamide-loaded cubic nanoparticles were observed to exist in both P and D phase form. The results of comparative evaluation of tropicamide-loaded cubic nanoparticles with commercial conventional ophthalmic solution revealed no significant difference in in vitro corneal permeation characteristics but significantly faster onset and higher intensity of mydriatic action was observed by in vivo study for the cubic nanoparticles formulation. According to HET-CAM’s and histochemical studies, the developed cubic ophthalmic nanoformulation should not bring discomfort to the patient. In conclusion, cubic liquid crystalline nanoparticles can be a promising vehicle for effective ocular delivery of tropicamide.
Acknowledgements
The authors express gratitude to SAIF, AIIMS, New Delhi and SMITA Research labs, IIT, Delhi for TEM and SAXS analysis. The authors are extremely grateful to the Coordinator, DST FIST, Dept. of Pharmaceutical Sciences (GJUS&T, Hisar) for particle size analysis.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
References
- Ahmed I. (2003). The noncorneal route in ocular drug delivery. In: Mitra AK, ed. Ophthalmic drug delivery systems. New York: Marcel Dekker, 335–63
- Alany RG, Rades T, Nicoll J, et al. (2006). W/O microemulsions for ocular delivery: evaluation of ocular irritation and precorneal retention. J Control Release 111:145–52
- Ali J, Fazil M, Qumbar M, et al. (2014). Colloidal drug delivery system: amplify the ocular delivery. Drug Deliv 21:1–17
- Barauskas J, Johnsson M, Joabsson F, Tiberg F. (2005). Cubic phase nanoparticles (cubosome): principles for controlling size, structure, and stability. Langmuir 21:2569–77
- Bashir MJK, Aziz HA, Yusoff MS, Adlan MN. (2010). Application of response surface methodology (RSM) for optimization of ammoniacal nitrogen removal from semi-aerobic landfill leachate using ion exchange resin. Desalination 254:154–61
- Baydoun L, Düvel A, Daniels R, et al. (2004). Comparison of different ibuprofen-amino acid compounds with respect to emulsifying and cytotoxic properties. Int J Pharm 274:157–65
- Bei D, Marszalek J, Youan BBC. (2009a). Formulation of dacarbazine-loaded cubosomes – part I: influence of formulation variables. AAPS PharmSciTech 10:1032–9
- Bei D, Marszalek J, Youan BC. (2009b). Formulation of dacarbazine-loaded cubosomes-part II: influence of process parameters. AAPS PharmSciTech 10:1040–7
- Boyd BJ. (2003). Characterisation of drug release from cubosomes using the pressure ultrafiltration method. Int J Pharm 260:239–47
- Calvo P, Vila-Jato JL, Alanson MJ. (1996). Comparative in vitro evaluation of several colloidal system, nanoparticles, nanocapsules and nanoemulsions as ocular drug carriers. J Pharm Sci 85:30–6
- Chong JYT, Mulet X, Waddington LJ, et al. (2012). Highthroughput discovery of novel steric stabilizer for cubic lyotropic liquid drystalline nanoparticles dispersions. Langmuir 28:9223–32
- De Sa FAP, Taveira SF, Gelfuso GM, et al. (2015). Liposomal voriconazole (VOR) formulation for improved ocular delivery. Colloids Surf B 133:331–8
- Dehelean C, Alexa E, Feflea S, et al. (2011). Ochratoxin A: a toxicologic evaluation using in vitro and in vivo bioassays. Ann Oradea Univ Biol Fasc 28:99–103
- Dian L, Yang Z, Li F, et al. (2013). Cubic phase nanoparticles for sustained release of ibuprofen: formulation, characterization, and enhanced bioavailability study. Int J Nanomed 8:845–54
- Dilbaghi N, Kaur H, Ahuja M, Kumar S. (2013). Evaluation of tropicamide-loaded tamarind seed xyloglucan nanoaggregates for ophthalmic delivery. Carbohydr Polym 94:286–91
- Dong YD, Larson I, Hanley T, Boyd BJ. (2006). Bulk and dispersed aqueous phase behavior of phytantriol: effect of vitamin E acetate and F127 polymer on liquid crystal nanostructure. Langmuir 22:9512–18
- Driever CD, Mulet X, Waddington LJ, et al. (2013). Layer by layer polymer coating on discrete particles of cubic liquid crystalline dispersions. Langmuir 29:12891–900
- Engstrom S, Ljusberg-Wahren H, Gustafsson A. (1995). Bioadhesive properties of the monoolein-water system. Pharm Tech Eur 7:14–17
- Ferreira DA, Bentley M, Karlsson G, Edwards K. (2006). Cryo-TEM investigation of phase behaviour and aggregate structure in dilute dispersions of monoolein and oleic acid. Int J Pharm 310:203–12
- Floris A, Meloni MC, Lai F, et al. (2013). Cavitation effect on chitosan nanoparticle size: a possible approach to protect drugs from ultrasonic stress. Carbohydr Polym 94:619–25
- Gan L, Han S, Shen J, et al. (2010). Self-assembled liquid crystalline nanoparticles as a novel ophthalmic delivery system for dexamethasone: improving preocular retention and ocular bioavailability. Int J Pharm 396:179–87
- Gaudana R, Ananthula HK, Parenky A, Mitra AK. (2010). Ocular drug delivery. AAPS J 12:348–60
- Gratieri T, Gelfuso GM, de Freitas O, et al. (2011). Enhancing and sustaining the topical ocular delivery of fluconazole using chitosan solution and poloxamer/chitosan in situ forming gel. Eur J Pharm Biopharm 79:320–7
- Guo C, Wang J, Cao F, et al. (2010). Lyotropic liquid crystal systems in drug delivery. Drug Discov Today 15:1032–40
- Gupta AK, Madan S, Majumdar DK, Maitra A. (2000). Ketorolac entrapped in polymeric micelles: preparation, characterisation and ocular anti-inflammatory studies. Int J Pharm 209:1–14
- Hao J, Wang F, Wang X, et al. (2012). Development and optimization of baicalin-loaded solid lipid nanoparticles prepared by coacervation method using central composite design. Eur J Pharm Sci 47:497–505
- Hyde ST, Andersson S, Ericsson B, Larsson K. (1984). A cubic structure consisting of a lipid bilayer forming an infinite periodic minimum surface of the gyroid type in the glycerolmonooleat-water system. Z Kristallogr 168:213–19
- Jadhav AJ, Holkar CR, Karekar SE, et al. (2015). Ultrasound assisted manufacturing of paraffin wax nanoemulsions: process optimization. Ultrason Sonochem 23:201–7
- Karaman S, Yilmaz MT, Ertugay MF, et al. (2012). Effect of ultrasound treatment on steady and dynamic shear properties of glucomannan based salep dispersions: optimization of amplitude level, sonication time and temperature using response surface methodology. Ultrason Sonochem 19:928–38
- Kaur H, Ahuja M, Kumar S, Dilbaghi N. (2012). Carboxymethyl tamarind kernel polysaccharide nanoparticles for ophthalmic drug delivery. Int J Biol Macromol 50:833–9
- Korbahti BK, Tanyolac A. (2008). Electrochemical treatment of simulated textile wastewater with industrial components and Levafix Blue CA reactive dye: optimization through response surface methodology. J Hazard Mater 151:422–31
- Kumar A, Ahuja M. (2014). Aqueous carboxymethyl gum kondagogu as vehicle for ocular delivery. J Pharm Investig 44:237–42
- Kuttruff H. (1988). Physik und Technik des Ultraschalls. Stuttgart: S. Hirzel Verlag
- Lallemand F, Daull P, Benita S, et al. (2012). Successfully improving ocular drug delivery using the cationic nanoemulsion, novasorb. J Drug Deliv 2012:604204
- Lancelot A, Sierra T, Serrano JL. (2014). Nanostructured liquid crystalline particles for drug delivery. Expert Opin Drug Deliv 11:547–64
- Landh T. (1994). Phase behavior in the system pine oil monoglycerides-poloxamer 407-water at 20°C. J Phys Chem 98:8453–67
- Law SL, Huang KJ, Chiang CH. (2000). Acyclovir-containing liposomes for potential ocular delivery. Corneal penetration and absorption. J Control Release 63:135–40
- Li J, Wu L, Wu W, et al. (2013). A potential carrier based on liquid crystal nanoparticles for ophthalmic delivery of pilocarpine nitrate. Int J Pharm 455:75–84
- Luepke NP. (1985). Hen’s egg chorioallantoic membrane test for irritation potential. Food Chem Toxicol 23:287–91
- Maurice DM, Riley MV, Graymore CN. (1970). Biochemistry of the eye. London. Academic Press, 6–16
- Muller F, Salonen A, Glatter O. (2010). Monoglyceride-based cubosomes stabilized by laponite: separating the effects of stabilizer, pH and temperature. Colloids Surf A Physicochem Eng Asp 358:50–6
- Nakano M, Teshigawara T, Sugita A, et al. (2002). Dispersions of liquid crystalline phases of the monoolein/oleic acid/Pluronic F127 system. Langmuir 18:9283–8
- Peltola S, Saarinen-Savolainen P, Kiesvaara J, et al. (2003). Microemulsions for topical delivery of estradiol. Int J Pharm 254:99–107
- Qiu H, Caffrey M. (2000). The phase diagram of the monoolein/water system: metastability and equilibrium aspects. Biomaterials 21:223–34
- Raichel DR. (2000). The science and applications of acoustics. New York: Springer
- Rodriguez C, Kunieda H. (2000). Effect of electrolytes on discontinuous cubic phases. Langmuir 16:8263–9
- Siekmann B, Bunjes H, Koch MH, Westesen K. (2002). Preparation and structural investigations of colloidal dispersions prepared from cubic monoglyceride-water phases. Int J Pharm 244:33–43
- Silva R, Ferreira H, Little C, Cavaco-Paulo A. (2010). Effect of ultrasound parameters for unilamellar liposome preparation. Ultrason Sonochem 17:628–32
- Spicer PT, Hayden KL, Lynch ML, et al. (2001). Novel process for producing cubic liquid crystalline nanoparticles (cubosomes). Langmuir 17:5748–56
- Tang SY, Manickam S, Wei TK, Nashiru B. (2012). Formulation development and optimization of a novel Cremophore EL-based nanoemulsion using ultrasound cavitation. Ultrason Sonochem 19:330–45
- Verma P, Ahuja M. (2015). Optimization, characterization and evaluation of chitosan-tailored cubic nanoparticles of clotrimazole. Int J Biol Macromol 73:138–45
- Zhang W, Prausnitz MR, Edwards A. (2004). Model of transient drug diffusion across cornea. J Control Release 99:241–58