Abstract
Background: A new series of 3,5-disubstituted thiazolidin-2,4-dione molecules were derived and characterized using various spectral techniques (1H NMR, IR, carbon, hydrogen, nitrogen, etc.) and physicochemical parameters. Materials & methods: The molecules were derived using Knoevenagel condensation followed by Mannich reaction and further synthesized analogues were screened for their antioxidant and antimicrobial potential using 2,2-diphenyl-1-picrylhydrazyl free radical scavenging method and serial tube dilution method, respectively, along with in silico studies (docking and absorption, distribution, metabolism and excretion parameters) to explore the drug–receptor interaction and druglikeness. Results & conclusion: In antimicrobial screening, the analogs MP2, MM6, MM7 and MM8 displayed promising activity while molecule MM4 exhibited better antioxidant potential in the series. In molecular docking analysis, the best-fitted analogs, namely, MM6 and MM7, showed good interactions.
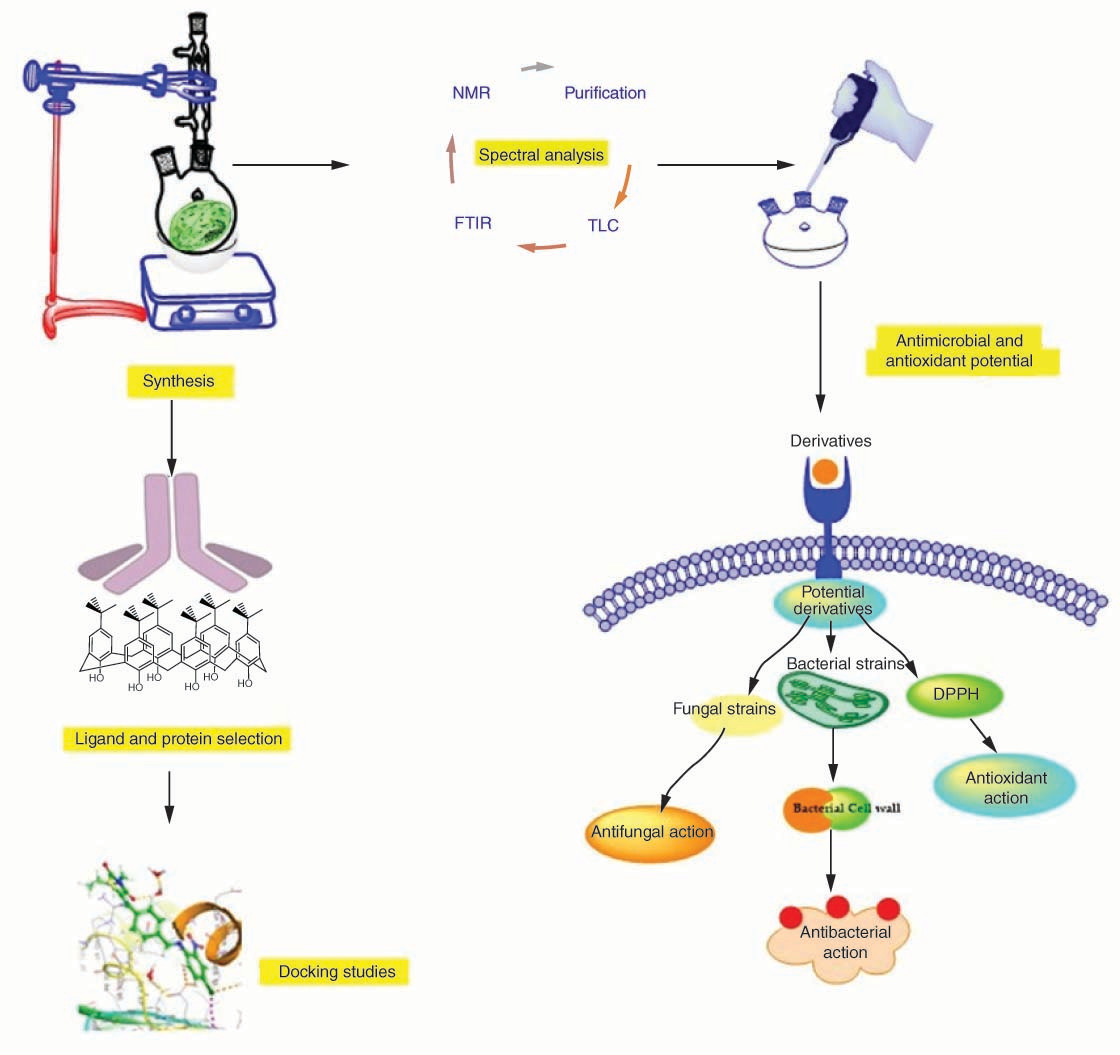
Graphical abstract
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at: www.tandfonline.com/doi/suppl/10.4155/fmc-2023-0237
Author contributions
H Kumar, A Sharma, MG Marwaha: writing (original draft) and formal analysis; D Kumar, RK Marwaha, M Dhanawat, N Aggarwal: editing; RK Marwaha: conceptualisation, editing and supervision. All authors approved the final version of the manuscript.
Acknowledgments
The authors are thankful to the Head, Department of Pharmaceutical Sciences, Maharshi Dayanand University and College of Pharmacy for providing necessary facilities.
Financial disclosure
The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
Conflicts of interest
The authors declare no conflict of interest.
