Abstract
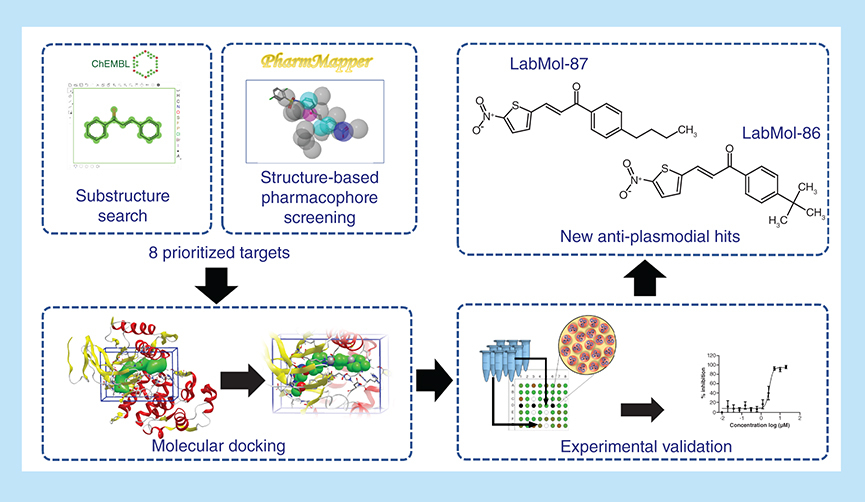
Aim: Computer-aided drug design approaches were applied to identify chalcones with antiplasmodial activity. Methodology: The virtual screening was performed as follows: structural standardization of in-house database of chalcones; identification of potential Plasmodium falciparum protein targets for the chalcones; homology modeling of the predicted P. falciparum targets; molecular docking studies; and in vitro experimental validation. Results: Using these models, we prioritized 16 chalcones with potential antiplasmodial activity, for further experimental evaluation. Among them, LabMol-86 and LabMol-87 showed potent in vitro antiplasmodial activity against P. falciparum, while LabMol-63 and LabMol-73 were potent inhibitors of Plasmodium berghei progression into mosquito stages. Conclusion: Our results encourage the exploration of chalcones in hit-to-lead optimization studies for tackling malaria.
Plain Language Summary
Graphical abstract
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at:www.tandfonline.com/doi/full/10.2217/epi-2016-0184
Financial & competing interests disclosure
The authors thank Brazilian funding agencies, CNPq, CAPES, FAPESP and FAPEG for financial support and fellowships. CH Andrade and FTM Costa are productivity fellows of CNPq. EN Muratov gratefully thank the NIH (grants 1U01CA207160 and R01-GM114015). CH Andrade is supported by CNPq (grant 400760/2014-2) and FAPESP #2017/02353-9. FTM Costa is supported by FAPESP (grants #2012/16525-2, #2017/18611-7 and 2018/0700-4). DY Bargieri is supported by FAPESP (grant #2013/13119-6), Instituto Serrapilheira (grant #G-1709-16618) and CNPq (grant 405996/2016-0). J Calit, GG Cassiano and LT Ferreira were supported by FAPESP (Fellowships #2018/24878-9, #2017/20774-6, and #2017/02031-1, respectively). We are grateful to ChemAxon (https://chemaxon.com/) for providing academic license of their program. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
This research protocol was approved by the Ethics Committee of the Institute of Biomedical Sciences – University of Sao Paulo, protocol number 132/2014-CEUA.
