Abstract
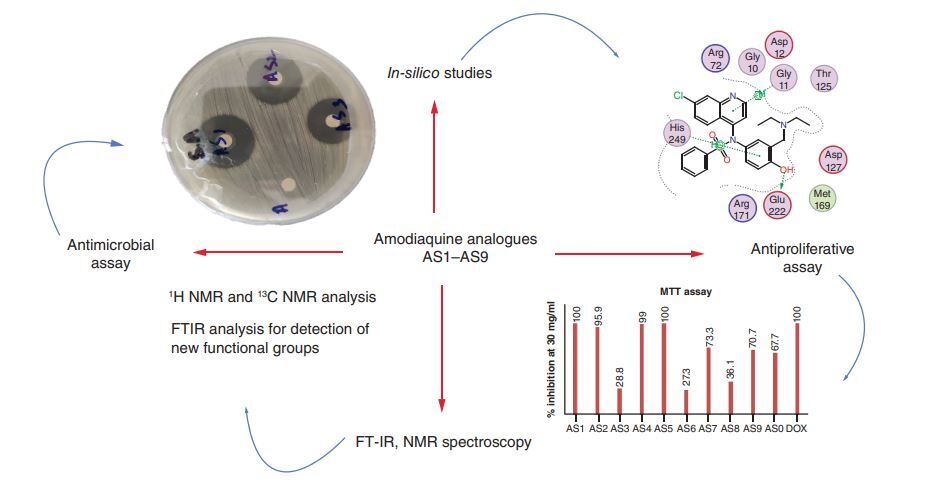
Aim: To synthesize and explore the therapeutic potential of amodiaquine analogues. Methodology: New promising analogues were synthesized by nucleophilic substitution at the 4-amino position and were characterized using 1H NMR, 13C NMR and FT-IR spectroscopic techniques. Results: Antibacterial and cytotoxic screening revealed the high potency of these compounds; analogue AS1 had an 34.3 ± 0.18 mm zone of inhibition against Pseudomonas aeruginosa. Excellent activity against fungal strains, that is, Candida albicans (39.6 ± 0.23 mm) was shown by analogue AS2. Analogue AS1 had an IC50 = 4.2 μg/ml against the HeLa cell line (cervical cancer) and binding energy against 5GWK (-8.32688 kcal/mol), 1PFK (-6.4780 kcal/mol) and 1TUP (-6.5279 kcal/mol) in the docking study. Conclusion: The obtained results reveal that these analogues exhibit potent antimicrobial and cytotoxic potential.
Graphical abstract
Financial disclosure
The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
