Abstract
This manuscript proposes an innovative approach to mitigate the gastrointestinal adversities linked with nonsteroidal anti-inflammatory drugs (NSAIDs) by exploiting amylose as a novel drug delivery carrier. The intrinsic attributes of V-amylose, such as its structural uniqueness, biocompatibility and biodegradability, as well as its capacity to form inclusion complexes with diverse drug molecules, are meticulously explored. Through a comprehensive physicochemical analysis of V-amylose and ulcerogenic NSAIDs, the plausibility of amylose as a protective carrier for ulcerogenic NSAIDs to gastrointestinal regions is elucidated. This review further discusses the potential therapeutic advantages of amylose-based drug delivery systems in the management of gastric ulcers. By providing controlled release kinetics and enhanced bioavailability, these systems offer promising prospects for the development of more effective ulcer therapies.
Challenges associated with nonsteroidal anti-inflammatory drugs (NSAIDs)
The primary cause of NSAID toxicity stems from its nonselective inhibition of cyclooxygenase enzymes, leading to disruptions in the gastric mucosa's defense mechanism and the formation of ulcers.
Polysaccharides show promise as advanced drug delivery systems due to their biodegradability, biocompatibility and ability to provide controlled release kinetics, thereby enhancing pharmacokinetics and maintaining optimal drug concentrations across physiological barriers.
Structural uniqueness of V-amylose
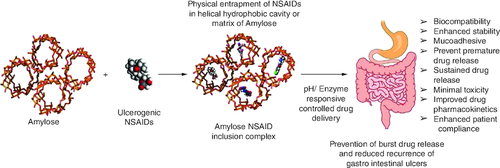
The tightly packed helical structure, hydrophobic interior and hydrophilic exterior with the 6-glucose unit per turn and a pitch height of 7.91–8.17 Å renders V-amylose a privileged delivery system for ulcerogenic NSAIDs.
Potential therapeutic advantages of V-amylose & NSAIDs conjugates
Emperical findings of V-amylose NSAID complexes demonstrated potential attributes of innovative drug delivery systems such as biodegradability, biocompatibility and controlled release kinetics for enhancing pharmacokinetics and maintaining optimal NSAID concentration in patients.
Amylose-based delivery platforms such as hydrogels, solid dosage formulation, enteric coated tablets and conjugates have proven effective in precisely targeting and delivering fenamic acid NSAIDs to the mucosal regions and gastrointestinal tract, minimizing ulcer development and maximizing therapeutic efficacy.
Incorporating V-amylose into drug delivery not only addresses ulcerogenicity concerns but also enhances the pharmacokinetics and bioavailability of NSAIDs.
Further research and advancement are imperative to translate these findings into practical applications.
Financial disclosure
The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.