Figures & data
Table 1 Compositions of the xNa2O-yCs2O-2SiO2 glasses (y = 1.0, 0.8, 0.66, 0.5, 0.33, 0.2; x + y = 1) analyzed by using XRF. The amount of alkali oxides was calculated as SiO2 = 2.
Fig. 1 XRD pattern of Cs2Si2O5 crystal. The XRD pattern of the standard Cs2Si2O5 crystal obtained from the simulation using POWDER CELL is also provided for reference. The hollow pattern is due to the borosilicate glass tube that was used to avoid water absorption.
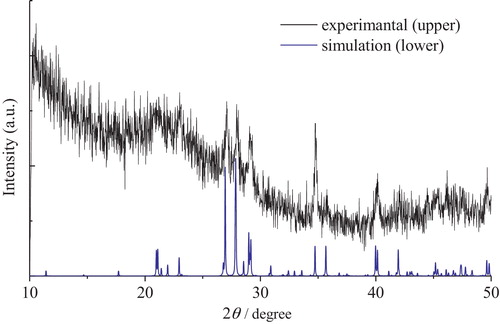
Fig. 2 XRD pattern of Cs6Si10O23 crystal. The XRD pattern of standard Cs6Si10O23 crystal obtained from the simulation using POWDER CELL is also provided for reference.
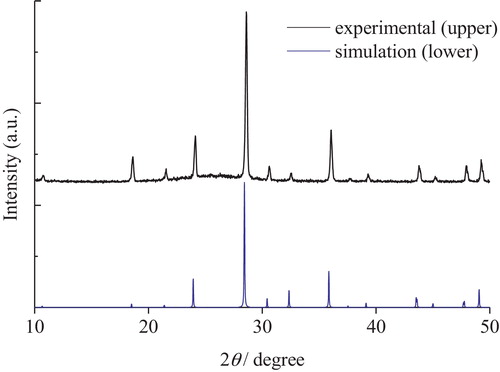
Fig. 4 23Na MAS NMR spectra of xNa2O-yCs2O-2SiO2 glasses (x = 0.2, 0.33, 0.5 0.66, 0.8, 1.0; x + y = 1).
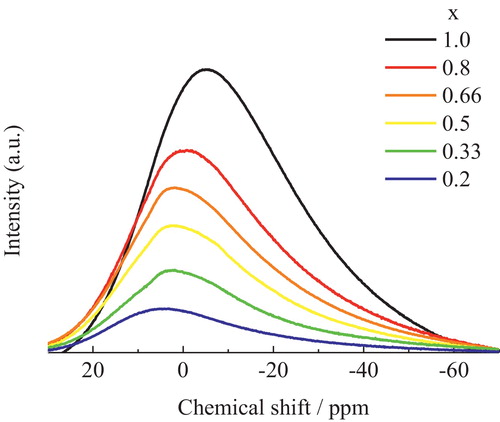
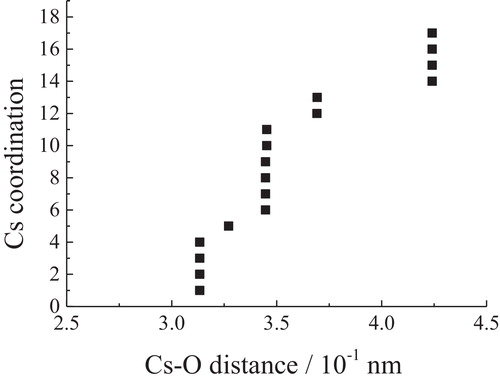
Fig. 6 Relationship between Cs–O distances and coordination numbers in Cs2Si2O5. There exist four kinds of cesium that are crystallographically independent.
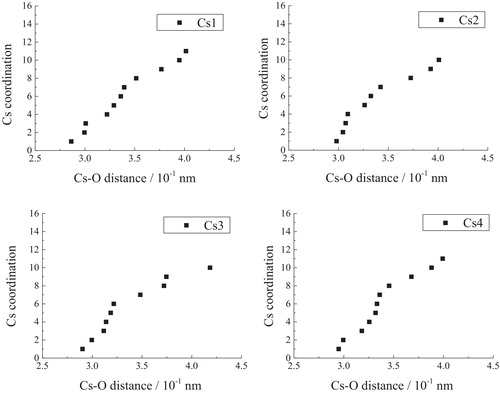
Fig. 5 133Cs MAS NMR spectra of xNa2O-yCs2O-2SiO2 glasses (y = 1.0, 0.8, 0.66, 0.5, 0.33, 0.2; x + y = 1).
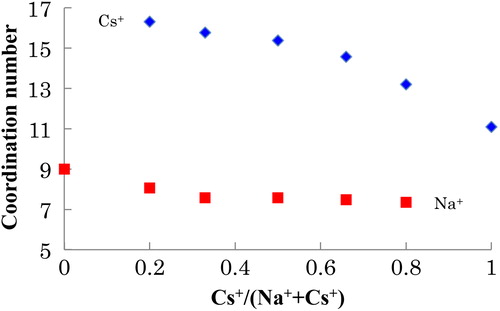
Table 2 Estimated coordination numbers of Na and Cs in xNa2O-yCs2O-2SiO2 glasses (x + y = 1) by using Eq. Equation(1)(1)
(1) . NNa and NCs are the coordination numbers of Na and Cs, respectively.