Figures & data
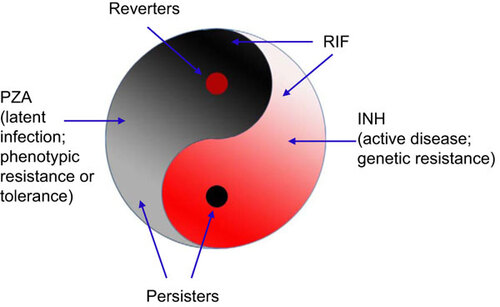
Table 1 Studies and observations that support or are consistent with the Yin–Yang model (see Figure 1)
Table 2 Diseases with known bacterial persistence problems
Table 3 Persister mechanisms in bacteria