Figures & data
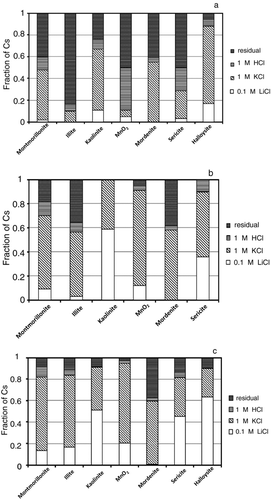
Figure 1 The distribution coefficients (K d) of Cs by minerals and clay minerals at different Cs concentrations. Deviation of duplicate measurement was within the symbols
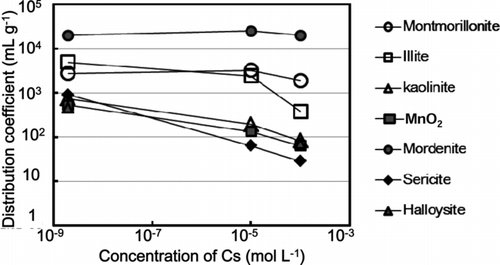
Figure 2 Fractions of Cs at 2 × 10−9 mole L−1 desorbed by different univalent cations at 20°C from montmorillonite, illite, kaolinite, MnO2, mordenite, halloysite, and sericite. Deviation of duplicate measurement was within the symbols
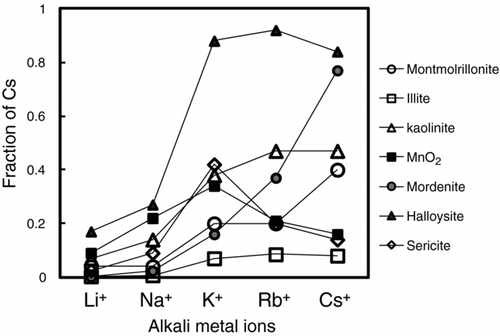
Figure 3 The sequential desorption of the adsorbed Cs at 10−9 mole L−1 (a), 10−5 mole L−1 (b), and 10−4 mole L−1 (c) by the 0.1 M LiCl solution, the 1 M KCl solution, the 1 M HCl solution, and the residual from montmorillonite, illite, kaolinite, MnO2, mordenite, sericite, and halloysite. Desorption data of Cs at 10−5 mole L−1 from halloysite was not measured because of the lack of the sample of halloysite