Figures & data
Table 1. Background data of the patients with pediatric-onset inflammatory bowel disease and treated with anti-TNF-alpha antagonist adalimumab between 2012 and 2018 at two tertiary care hospitals in Finland.
Table 2. Therapy with anti-TNF-alpha antagonist adalimumab (ADA) of the patients with pediatric-onset inflammatory bowel disease.
Figure 1. Trough levels during the first 3 months of adalimumab therapy are related to the probability of ongoing therapy during the first year.
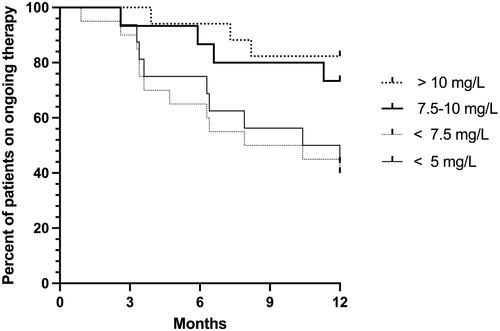
Table 3. Disease activity at induction and last follow-up of ongoing adalimumab therapy.
