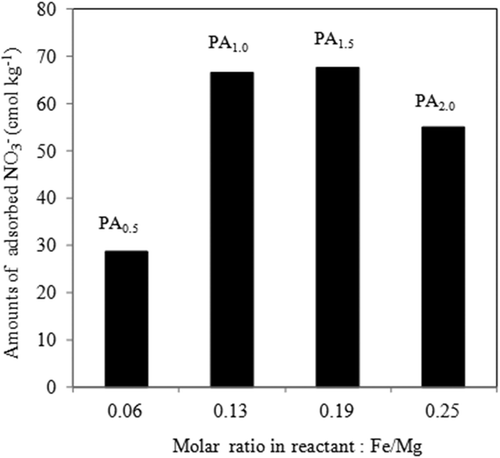
Figures & data
Table 1. Chemical composition of PA1.5.
Table 2. X-ray powder diffraction data.
Table 3. The pH value at equilibrium and AEC of PA1.5.
Figure 3. NO3– exchange isotherm of the PA1.5. The approximated curve was drawn by the formula of Langmuir.
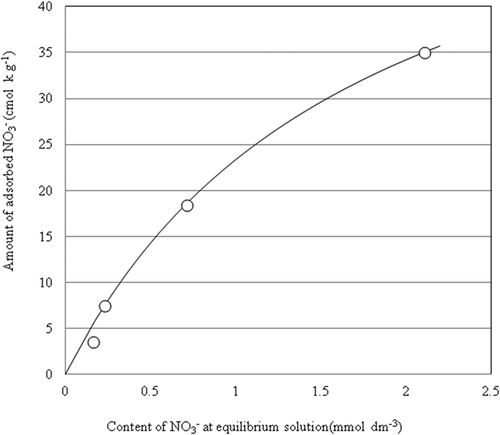
Figure 4. Relationship between NO3– adsorption and the Cl–desorption. Simple regression analysis was done by Excel software.
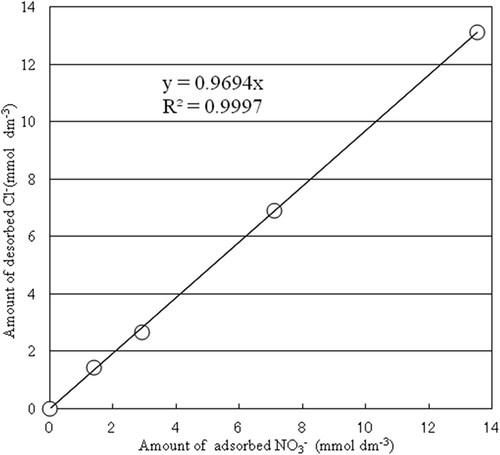
Table 4. The anion adsorption, selectivity coefficient (KGT), and the distribution coefficient (Kd) for each anion on the equilibrium solution containing one kind of anion.
Table 5. The anion adsorption and distribution coefficient (Kd) for each anion on the equilibrium solution containing three kinds of anion.
Table 6. The competitive adsorption for each anion in the imitative soil solution.