Figures & data
Table 1. In vitro activity of bexagliflozin and its principal human metabolites.
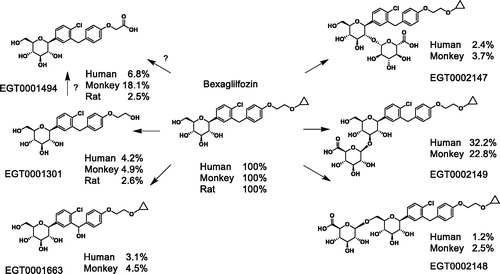
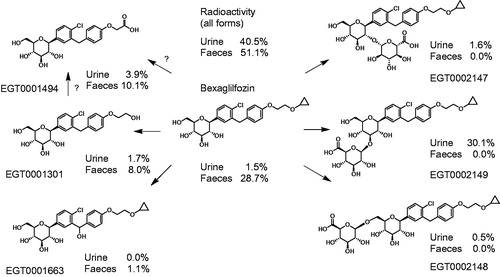
Table 2. Distribution of radiolabel among parent compound and metabolites in rats, monkeys and humans.
Table 3. Relative proportions of metabolites in male and female cynomolgus monkeys.
Figure 1. Plasma concentration of total radioactivity in plasma, blood and as bexagliflozin in plasma as a function of time from dose administration in humans.
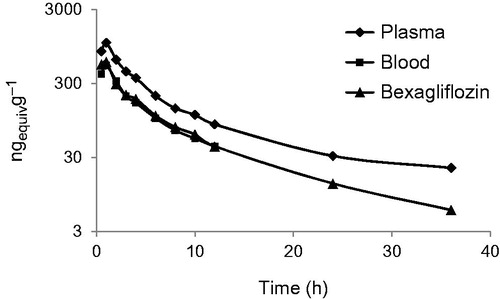
Table 4. Pharmacokinetic parameters for bexagliflozin and its principal metabolites in humans, as inferred by distribution of 14C-labelled material.
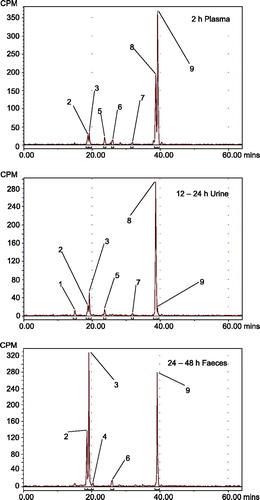
Figure 2. Representative radiochromatograms of human samples. Radiochromatograms depicting the nine discrete human specimen peaks are shown. Peak identifications: 1, metabolite HM1; 2, EGT0001301; 3, EGT0001494; 4, metabolite HM2; 5, EGT0002147; 6, EGT0001663; 7, EGT0002148; 8, EGT0002149; 9, bexagliflozin.