Figures & data
Table I. Patient characteristics.
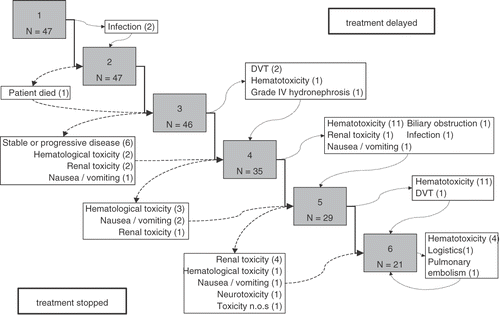
Table II. Most severe toxicity observed per treatment series.
Table III. Responses inside and outside the heated area.
Table IV. Reports in literature on response rate for irradiated recurrent cervical carcinomas (reponses/no. evaluable)*.