Figures & data
Figure 1. Comparison of the spectra of the iron standards (0–2 μg Fe+3) in working solution. Ferene-s (A), ferrozine (B), and the corresponding standard curves (0.1–4 μg Fe+3) measured at the peak absorbance for each compound (560 nm for ferrozine and 595 nm for ferene-s), (C).
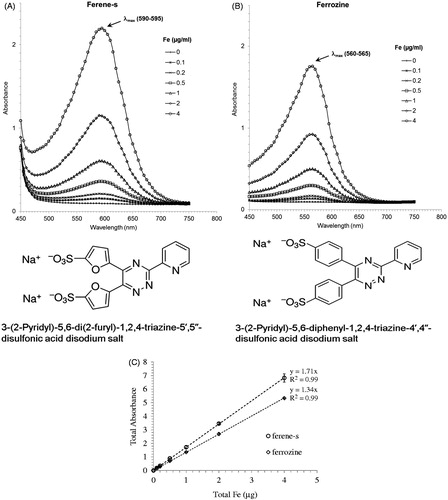
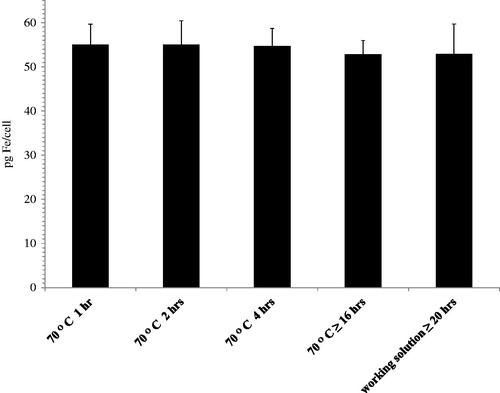
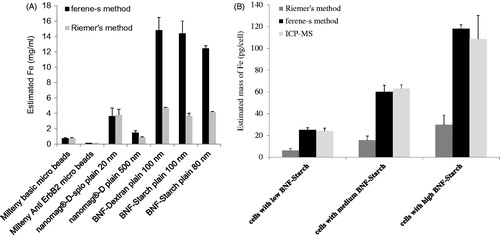
Table 1. Iron concentrations obtained from various iron oxide nanoparticles.
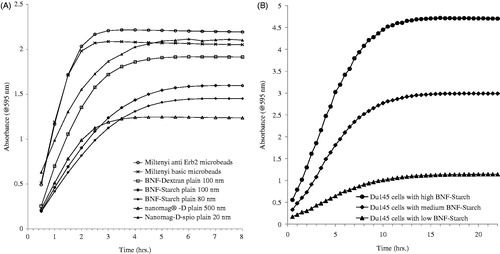
Figure 2. Time course of absorbance at 595 nm for ferene-s assay in working solution. Iron oxide nanoparticles with various formulations (A) and intracellular BNF-Starch iron oxide nanoparticles (B), DU145 cells with low, medium and high Fe content). Absorbance readings are adjusted to reflect equivalent number of cells.