Figures & data
Table 1. Inclusion and exclusion criteria for HIFU treatment of adenomyosis.
Figure 1. Serial images of epidural catheterisation. The tip of a 17G Tuohy needle was in the epidural space (a), epidural catheter with curved steel wire (white arrows) was advanced cephalad (b), and 3 ml of contrast media was injected (c). There is no evidence of vascular uptake or subdural spreading pattern.
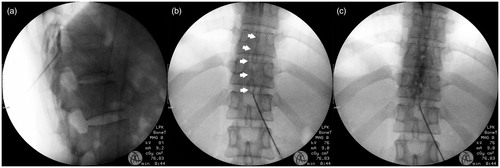
Table 2. Demographic and clinical data of the study population.
Figure 2. The proportion of intraprocedural pain intensity during HIFU treatment in the MAC group (n = 37) and the EA group (n = 31). The pain intensity was reported using a 6-point Likert scale (0, no pain; 1, mild pain; 2, mild-moderate pain; 3, moderate pain; 4, severe pain; 5, very severe pain).
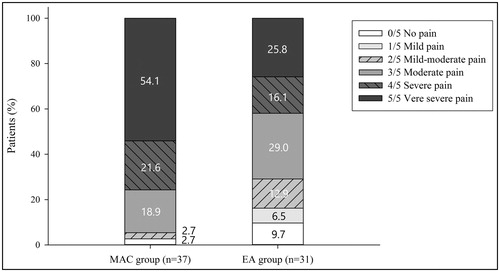
Table 3. Clinical outcomes of epidural analgesia and patient-controlled epidural analgesia for HIFU treatment.
Table 4. Multivariable analysis between the NPV ratio and Clinical Variables.
Table 5. Adverse Events (AEs), Adjuvant Drug Usage, and Complications.
