Figures & data
Figure 1. Reasons for discontinuation of biologic therapy. *Data for patients who provided a reason for discontinuation of therapy and physicians who reported a reason for their patient discontinuing therapy; †lack of control of shortness of breath/exacerbations; ‡includes all other respiratory symptoms not classifiable. OCS, oral corticosteroid.
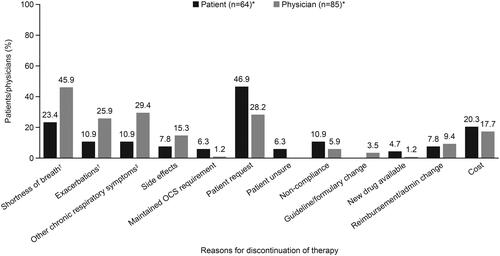
Figure 2. Asthma medication use at the time of the survey. ICS inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist; LRA, leukotriene receptor antagonist; OCS, oral corticosteroid; SABA, short-acting β2-agonist; SAMA, short-acting muscarinic antagonist.
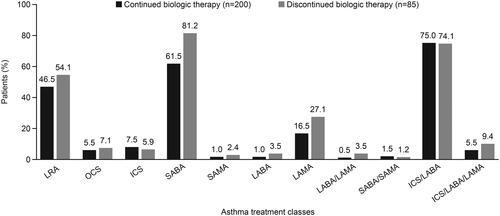
Table 1. Patient demographics and baseline characteristics.
Table 2. Patient comorbidities.
Figure 5. Patient (A) and physician (B)-reported reasons for missing a biologic administration appointment.
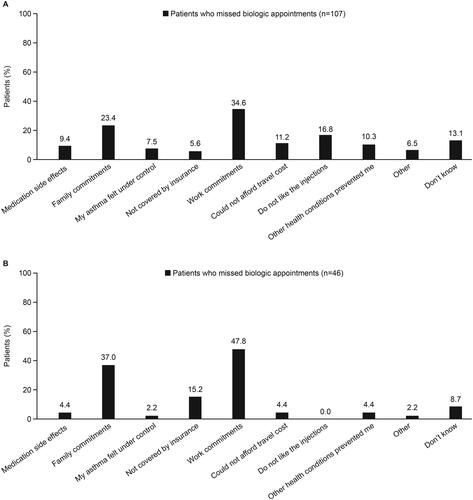
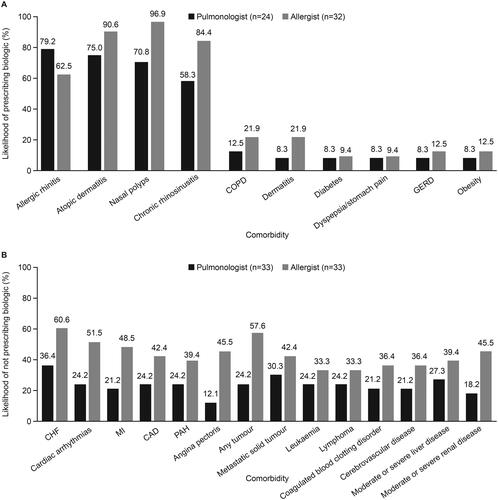
Figure 6. Comorbidities (A) increasing and (B) decreasing the likelihood of physician prescribing of biologics. CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; MI, myocardial infarction; PAH, pulmonary arterial hypertension.
Supplemental Material
Download PDF (809.1 KB)Supplementary_Material_Response_to_reviewers_final.docx
Download MS Word (143.2 KB)Data availability statement
GSK makes available anonymized individual participant data and associated documents from interventional clinical studies that evaluate medicines, upon approval of proposals submitted to www.clinicalstudydatarequest.com. To access data for other types of GSK sponsored research, for study documents without patient-level data and for clinical studies not listed, please submit an enquiry via the website. The data that support the findings of this study are available from Adelphi Real World, where restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of Adelphi Real World.