Figures & data
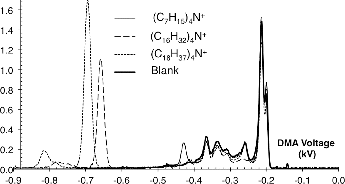
FIG. 1. Mobility spectrum for a spray of tetrahexadecylammonium bromide, showing on its right boundary the sharp peak associated to (C16H33)4N+.
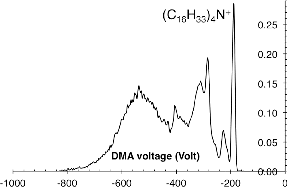
FIG. 2. Mobility spectra from sprays of (C18H37)4N+Br− (open circles) and (C7H15)4N+Br− ions (filled circles) taken under identical conditions of the DMA. The dominant and most mobile peak in the spray of (C18H37)4N+Br− is (C18H37)4N+.
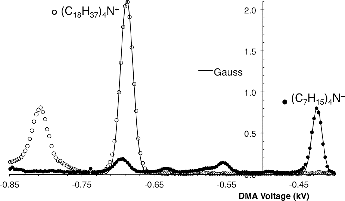
Table 1 Measured electrical mobilities Z in laboratory air based on the (C7H15)4N+ ion standard
FIG. 3. Mobility spectra after passing the electrospray ions through a charge-reduction chamber containing radioactive Ni-63.