Figures & data
Figure 1. Microfluidic channel masks (left) and SEM images of the corresponding PDMS channels (right) of a T-junction (top) and co-flow with flow focusing (bottom) geometries. In the mask images (left), arrows denoting continuous, dispersed, sheath, and emulsion flows show direction of flow and approximate relative velocities (proportional to arrow length) of the flow in each channel.

Figure 2. Relevant flow regimes in microfluidic flows (use mL hr−1) and typical aerosol particle sampling (use liters per minute, LPM), for a given volumetric flow rate at a fixed hydraulic diameter. Microfluidic flow is considered in channels 100 µm tall ranging from 10 µm wide with ethanol () to 1 mm wide with heavy mineral oil (
). Typical microfluidic flows have Re < 1. Typical aerosol particle sampling is considered for sample tubing from about 1/8 in. to 1 in. diameter for sampling particles in air (
).
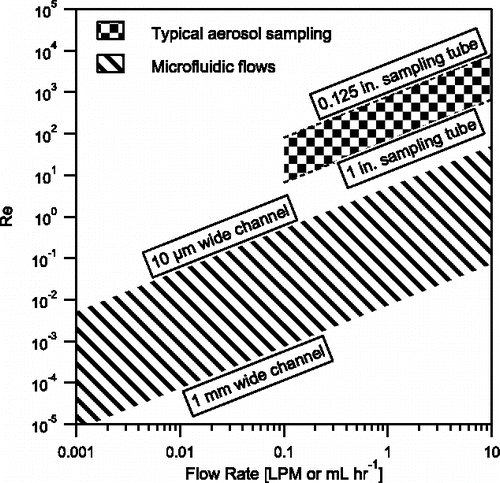
Table 1. Selected aerosol generation techniques with typical size distribution parameters. CV is coefficient of variance, , (Equation Equation(7)
[7] ).
Table 2. Comparison of typical particle diameters for various aerosol experiment setups, sampling instruments, and atmospheric conditions. Although biphasic microfluidics is typically operated at droplet sizes at the large end of coarse mode aerosol and into the cloud droplet regime, more refinement of microfluidic droplet generation techniques will lower the smallest diameters accessible to microfluidic experiments.
Figure 3. Selected microfluidic methods for producing temperature gradients, chemical gradients, and electric fields. (a) Reproduced from Stan, C. A., Schneider, G. F., Shevkoplyas, S. S., Hashimoto, M., Ibanescu, M., Wiley, B. J. and Whitesides, G. M. (2009). A Microfluidic Apparatus for the Study of Ice Nucleation in Supercooled Water Drops. Lab Chip, 9(16):2293–2305 with permission of The Royal Society of Chemistry. (b) Reproduced from Morel, M., Galas, J.-C., Dahan, M. and Studer, V. (2012). Concentration Landscape Generators for Shear Free Dynamic Chemical Stimulation. Lab Chip, 12(7):1340–1346 with permission of The Royal Society of Chemistry. (c) Reproduced from Fair, R. B., Khlystov, A., Srinivasan, V., Pamula, V. K. and Weaver, K. N. (Citation2004). Integrated Chemical/Biochemical Sample Collection, Pre-Concentration, and Analysis on a Digital Microfluidic Lab-on-a-Chip Platform. Edited by Linda A Smith and Daniel Sobek. Proc. SPIE, 5591:113–124 with permission of SPIE.
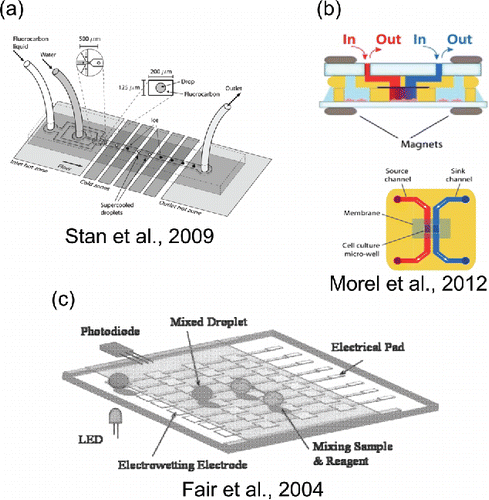
Figure 4. Selected microfluidic chemical and physical measurement techniques. (a) Reprinted with permission from Metcalf, A. R., Boyer, H. C. and Dutcher, C. S. (2016). Interfacial Tensions of Aged Organic Aerosol Particle Mimics Using a Biphasic Microfluidic Platform. Environ. Sci. Technol., 50(3):1251–1259. Copyright 2016 American Chemical Society. (b) Reprinted with permission from Song, H., Tice, J. D. and Ismagilov, R. F. (2003). A Microfluidic System for Controlling Reaction Networks in Time. Angew. Chem., 115(7):792–796. Copyright 2003 WILEY-VCH Verlag GmbH & Co. (c) Reprinted from Damit, B. (2017). Droplet-Based Microfluidics Detector for Bioaerosol Detection. Aerosol Sci. Technol., 51(4):488–500 with permission of the publisher (Taylor & Francis Ltd, http://www.tandfonline.com) and The American Association for Aerosol Research, www.aaar.org.
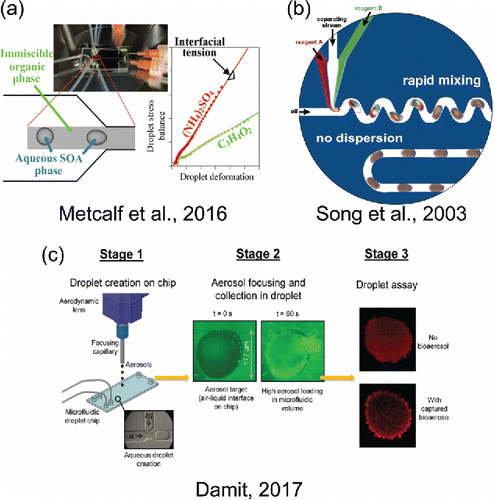
Figure 5. Selected microfluidic droplet and particle sorting techniques. (a) Reprinted with permission from Maenaka, H., Yamada, M., Yasuda, M. and Seki, M. (Citation2008). Continuous and Size-Dependent Sorting of Emulsion Droplets Using Hydrodynamics in Pinched Microchannels. Langmuir, 24(8):4405–4410. Copyright 2008 American Chemical Society. (b) Reprinted with permission from Squires, T. M. and Quake, S. R. (Citation2005). Microfluidics: Fluid Physics at the Nanoliter Scale. Rev. Mod. Phys., 77(3):977–1026. Copyright 2005 by the American Physical Society. (c) Adapted by permission from Macmillan Publishers Ltd: Nature Protocols, Mazutis, L., Gilbert, J., Ung, W. L., Weitz, D. A., Griffiths, A. D., and Heyman, J. A. (2013). Single-Cell Analysis and Sorting Using Droplet-Based Microfluidics. Nat. Protoc., 8 (5):870 -891, copyright 2013.
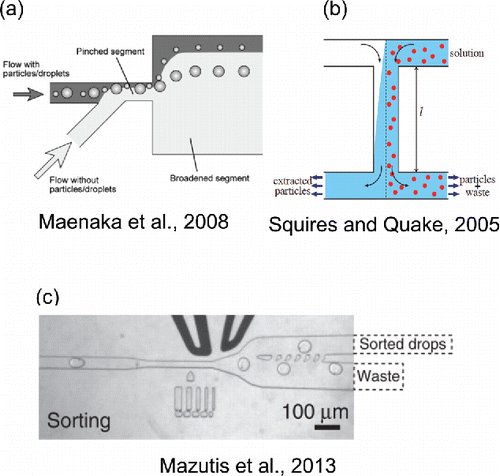
Figure 6. Selected microfluidic trapping and manipulation techniques. (a) Reproduced from Dylla-Spears, R., Townsend, J. E., Jen-Jacobson, L., Sohn, L. L. and Muller, S. J. (2010). Single-Molecule Sequence Detection via Microfluidic Planar Extensional Flow at a Stagnation Point. Lab Chip, 10(12):1543–1549 with permission of The Royal Society of Chemistry. (b) Reprinted with permission from Tanyeri, M. and Schroeder, C. M. (2013). Manipulation and Confinement of Single Particles Using Fluid Flow. Nano Lett., 13(6):2357–2364. Copyright 2013 American Chemical Society. (c) Reprinted with permission from Shenoy, A., Rao, C. V. and Schroeder, C. M. (Citation2016). Stokes Trap for Multiplexed Particle Manipulation and Assembly Using Fluidics. Proc. Natl. Acad. Sci. U. S. A., 113(15):3976–3981.
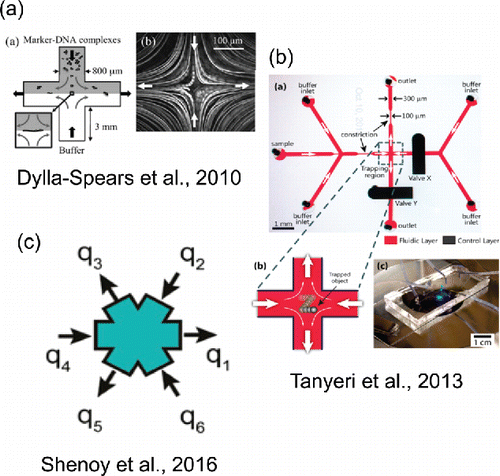
Table 3. Selected microfluidic techniques and potential aerosol science applications.
