Figures & data
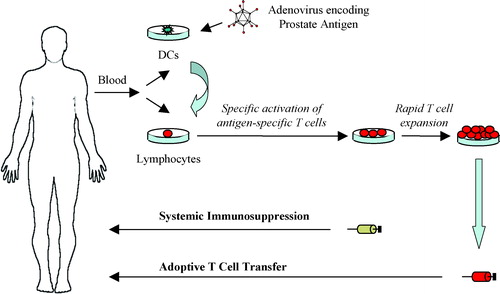
Figure 1. Strategies for adenovirus retargeting. Restricted adenovirus replication can be achieved through tumor- or tissue-specific promoter-controlled E1A gene expression (a), or through genetic modification of E1A and/or E1B to create virus strains with selective replication in tumor cells with deregulated cell cycle pathways (b). Adenovirus infection can be retargeted from its natural binding receptor to a specific cell surface molecule through genetic modification of the virus fiber or capsid proteins which the virus uses for attachment and cell entry (c) or by using bi-specific linkers that bridge virus surface proteins to specific cell surface receptors (d).
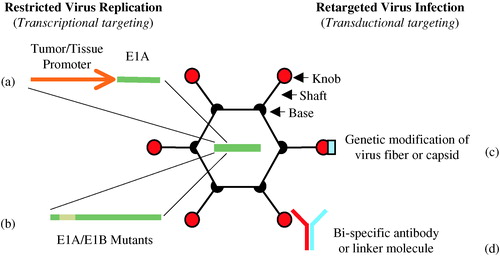
Figure 2. Oncolytic adenovirus therapy. A conditionally replication-competent adenovirus (CRAd) with E1A gene expression controlled by a prostate-specific promoter specifically replicates in and lyses cells of prostate epithelial origin, including prostate adenocarcinoma cells. The oncolytic prostate CRAd virus may be injected intraprostatically in patients with localized prostate cancer or intravascularly in patients with metastatic prostate cancer.
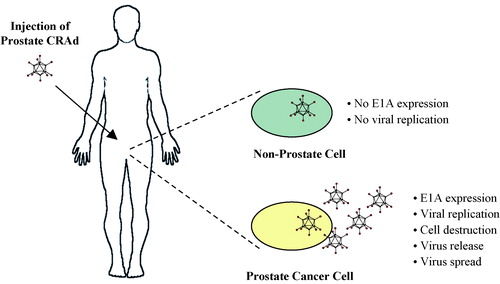
Figure 3. Adoptive transfer of prostate antigen-specific T cells. Monocyte-derived dendritic cells (DCs) are genetically modified with a prostate antigen-encoding adenovirus vector. The modified DCs are mixed with autologous lymphocytes for stimulation of antigen-specific T cells ex vivo. Antigen-specific T cells are selected and rapidly expanded to large numbers for adoptive therapy. The patient receives systemic immunosuppressive chemotherapy prior to the adoptive transfer of T cells.