Figures & data
Table I. Baseline clinical patient characteristics.
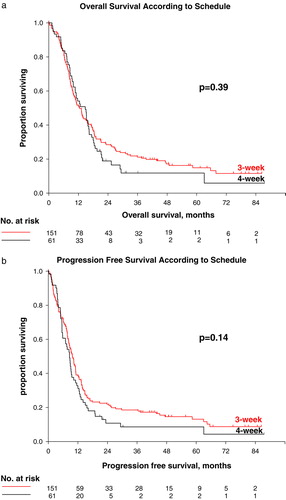
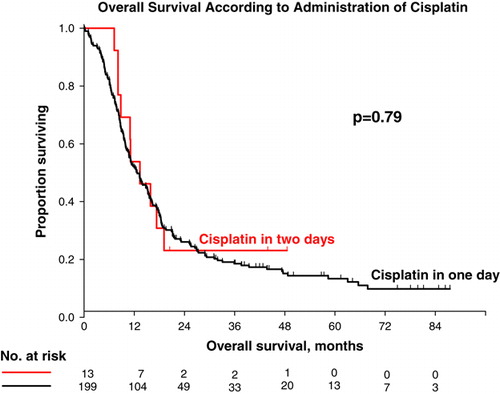
Table II. Survival parameters.
Table III. Summary of efficacy outcomes.
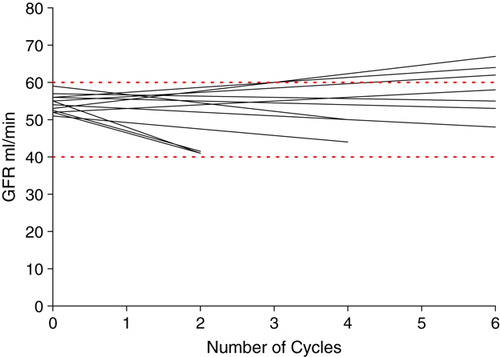
Table IV. Toxicity ratios, n = 212.
Table V. Dose intensity and compliance.
Table VI. Treatment compliance.
Table VII. Presentation of 3- and 4-week GC Schedules.