Figures & data
Figure 1. Review of major mechanisms for activation of CD-8 T-cells by neoantigens from a tumor. First immature antigen presenting cells (APC) is activated to mature dendritic cells (DC) by binding of neoantigens to major histocompatibility complex type I (MHC I). Co-stimulation for activation of CD8 T-cells is illustrated by the binding of CD28 receptors on T-cells to either B7-1 or B7-2 ligands on the mature DC. The inhibiting receptor CTLA-4 is also illustrated. The main inhibition of peripheral effect of T-cell is exerted by binding of programed death receptor 1 (PD-1) to either programed death ligand 1 or 2 (PD-L1 or PD-L2). Immune checkpoint blockers (ICBs) are represented with antibodies raised against the blocking molecules (marked in red). Interferons (IFN) together with transforming growth factor β (TGFβ) protect tumor cells from immunologic inactivation. Stimulating signals marked (+), inhibition marked (–) (illustration: Kristin Risa).
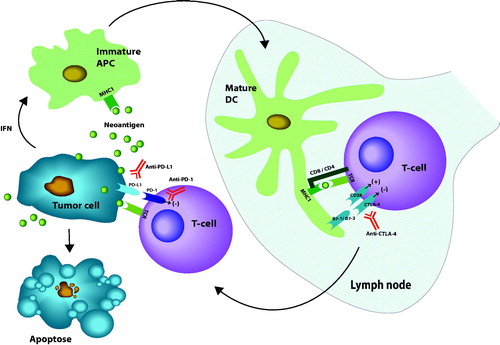
Table 1. Clinical indications approved by U.S. Food and Drug Administration (FDA) and/or European Medicines Agency (EMA) for immune checkpoint blockers. Only the year of first approval for an indication of a drug given in the table.
Table 2. Mechanisms for primary and acquired resistance to immune checkpoint inhibitors.
