Figures & data
Table 1. Squamous cell carcinomas of the base of tongue (n = 545).
Figure 1. (A) Overall survival in all patients with BOTSCC (n = 545) treated with a curative intent 2008–2014, in relation to tumor stage (UICC 7). Log-rank test p = 0.17. OS 5 years: Stage I: 86%, II: 66%, III: 78%, IV: 67%. (B) Overall survival (OS) for all patients with BOTSCC (n = 545) depending on residency, in the different health care regions. OS 5 years: Stockholm, South, South East: 66%, U/Ö, West: 70%, North: 75%. Log-rank test, p = 0.80.
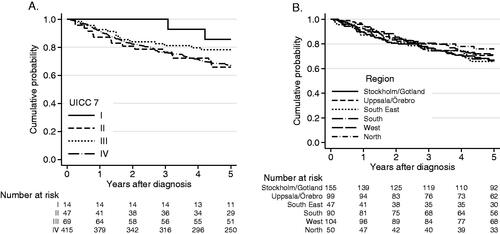
Figure 2. (A) Overall survival according to treatment strategy, for patients with tumor stages I–IV (UICC 7, n = 545). OS 5 years: EBRT: 64%, EBRT + BT: 71%, EBRT + CT: 65%, EBRT + BT + CT: 75%. (B) Overall survival and p16 analysis, p16? = not available for immunohistochemistry. Log-rank test, p < 0.001. OS 5 years: p16+: 77%, p16−: 38%, p16?: 66%.
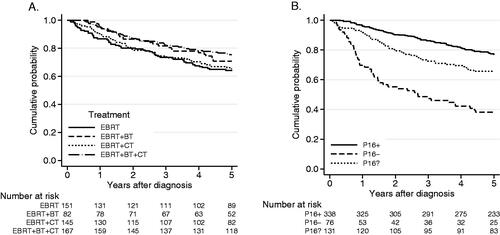
Table 2. Uni- and multivariable Cox regression.
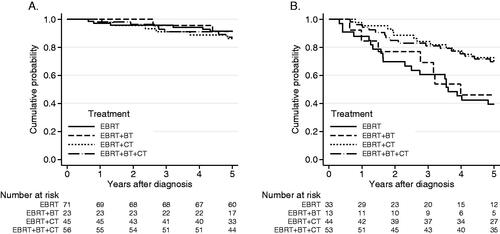
Figure 3. (A) Overall survival for patients with p16+, stage l (UICC 8), according to treatment-strategy (n = 195). Log-rank test, p = 0.81. OS 5 years: EBRT: 92%, EBRT + BT: 91%, EBRT + CT: 86%, EBRT + BT + CT: 87%. (B) Overall survival for patients with p16+ BOTSCC, stages II–III, (UICC 8, n = 143) according to treatment-strategy. Log-rank test, p = 0.004. OS 5 years: EBRT: 39%, EBRT + BT: 46%, EBRT + CT: 70%, EBRT + BT + CT: 69%.
Data availability statement
Data are obtained from the national quality register for head and neck cancer, SweHNCR, for the study after ethical approval and approval from the registration authority.
