Figures & data
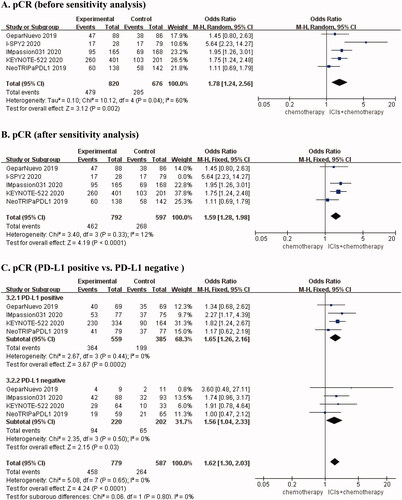
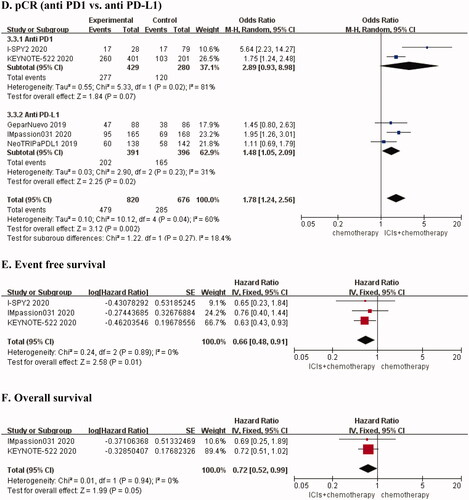
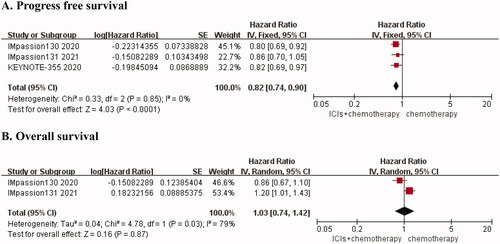
Table 1. Characteristics of included studies.
Table 2. Pooled the endpoints of efficacy and safety.
Table 3. Pooled odds ratio for toxicities in early triple-negative breast cancer.
Table 4. Pooled odds ratio for toxicities in advanced triple-negative breast cancer.