Figures & data
Figure 1. Overview of collection of materials (biological sampling and questionnaires) in the BioLymph study.
CSF: cerebrospinal fluid; QOL: quality of life; cfDNA: cell-free DNA, PET/CT: positron-emission tomography/computed tomography
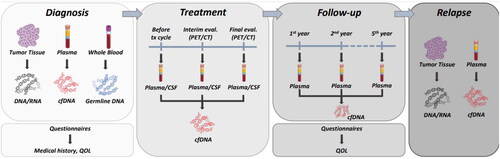
Figure 2. Overview of collection of biological materials in the BioLymph study by lymphoma subtype and treatment protocol.
CSF: cerebrospinal fluid; QOL: quality of life; cfDNA: cell-free DNA; PET/CT: positron-emission tomography/computed tomography; RT: radiotherapy
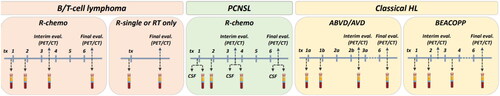
Table 1. Overview of patient characteristics and materials collected at diagnosis, and targeted sequencing performed for the first 100 study participants in the BioLymph study.
Supplemental Material
Download MS Word (20.1 KB)Supplemental Material
Download MS Word (283.5 KB)Data availability statement
Study data are available upon request if in line with ethical and legal permissions.
