Figures & data
Table 1. Demographic and clinical sample characteristics.
Figure 1. Successfully treated patients (STPs) at week 8 by treatment group.
*p < .05. Treated patients; last observation carried forward. STP Mood and Tolerability: vortioxetine n = 61/211; venlafaxine, n = 45/226; STP Mood and Functioning: vortioxetine n = 59/211; venlafaxine, n = 53/226.
Abbreviations. MADRS, Montgomery–Åsberg Depression Rating Scale; SDS, Sheehan Disability Scale; STP, Successfully treated patient; TEAE, Treatment-emergent adverse event; XR, Extended release.
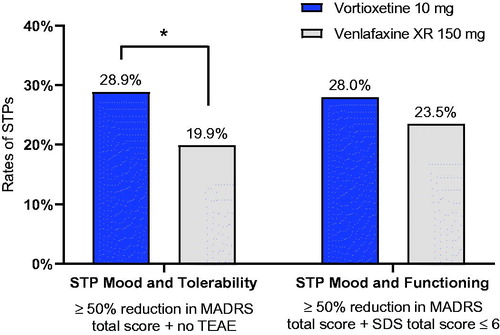
Table 2. Summary of drug costs, treatment outcomes and base case results.
Table 3. Clinical sensitivity results.
Table 4. Region sensitivity results, brand and generic prices.
Supplemental Material
Download PDF (5.6 KB)Data availability
The authors confirm that the data supporting the findings of this study are available within the article. The authors may be contacted for further data sharing.
