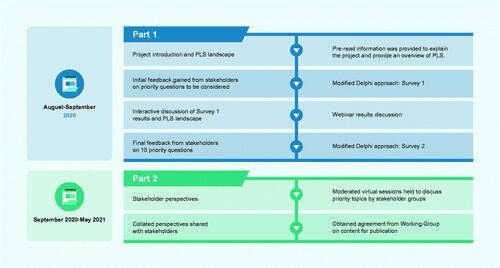
Figures & data
Figure 2. Expert stakeholders who participated in the study. aOne stakeholder accepted the invitation for the project but was not able to complete either survey or participate in the stakeholder discussion sessions. Abbreviations: HCP&CR, healthcare professionals and clinical researchers; M&Soc, media, International Society for Medical Publication Professionals, and the multi-regional clinical trials center; Phar, pharmaceutical industry; P&JE, publishers, journal editors, and National Institutes of Health/National Library of Medicine; PLS, plain language summary; PME, publication/medical education agencies; PP, patient partners.
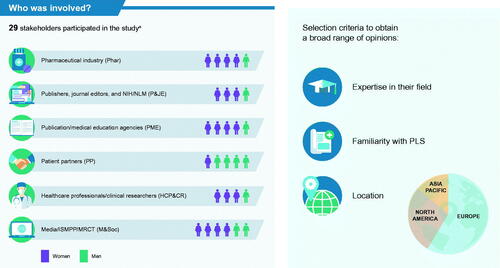
Table 1. Key information that should be included in a PLS.
Figure 3. Who are the key target audience for PLS? Abbreviations: HCP, healthcare professional; PAG, patient advocacy group; PLS, plain language summary.
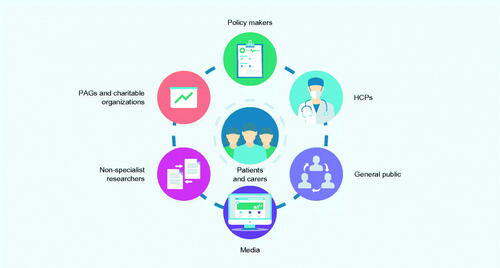
Figure 4. Overview of an example content development, review, and approval process for PLS of journal articles developed with medical writing support. Abbreviation: PLS, plain language summary.
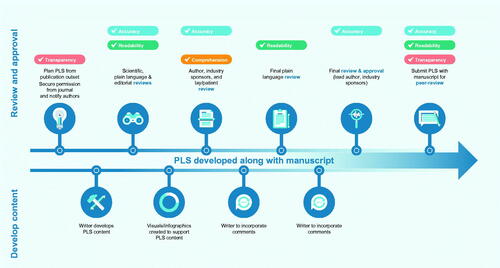
Figure 5. Potential approaches to select which publications should include a PLS. Abbreviation: PLS, plain language summary.
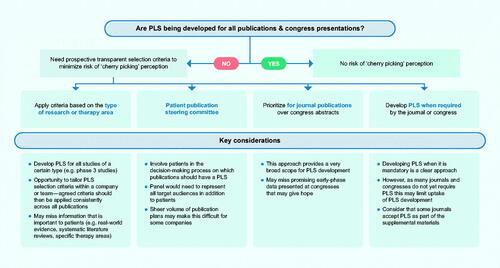
Table 2. Opportunities to overcome real or perceived barriers to the uptake of PLS.
Supplemental Material: PLS Graphic
Download PDF (724.3 KB)Supplemental Material: PLS Perspectives
Download PDF (317.4 KB)Data availability statement
The data that support the findings of this study are available from the corresponding author, Dawn Lobban, upon reasonable request.