Figures & data
Table 1. Baseline characteristics before applying IPTW and the absolute standardized difference before and after IPTW.
Figure 1. Patient disposition. Abbreviations. AF, atrial fibrillation; CKD, chronic kidney disease; DVT, deep vein thrombosis; ESKD, end-stage kidney disease; NOAC, non-vitamin K antagonist oral anticoagulant; PE, pulmonary embolism; VKA, vitamin K antagonist; VTE, venous thromboembolism
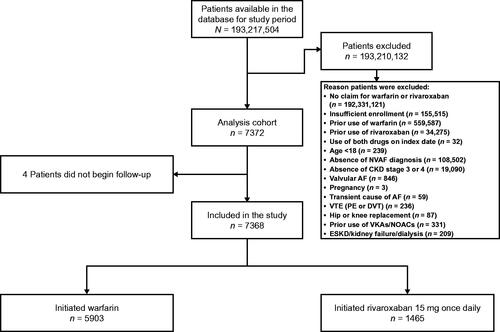
Table 2. Reasons for censoring in the main patient cohort and for each exposure group for the worsening kidney function outcome.
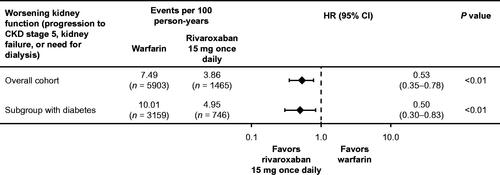
Figure 2. Worsening kidney function (progression to CKD stage 5, kidney failure, or need for dialysis) in the overall cohort and diabetes subgroup using stabilized IPTW methodology to adjust for differences in baseline characteristics. Abbreviations. CI, confidence interval; CKD, chronic kidney disease; HR, hazard ratio; IPTW, inverse probability of treatment weighting
Supplemental Material
Download MS Word (79.6 KB)Data availability statement
This study used data from US IBM Watson MarketScan Commercial Claims and Medicare Supplemental databases.
