Figures & data
Figure 1. Sample attrition for Medicaid pediatric growth hormone deficiency cohort. 1Eligible patients were grouped into non-exclusive cohorts based on length of available follow-up, determined by index date and continuous enrollment. For example, all patients in the 36-month continuous enrollment cohort, by definition, are also included in the 3-, 12-, and 24-month continuous enrollment cohorts.
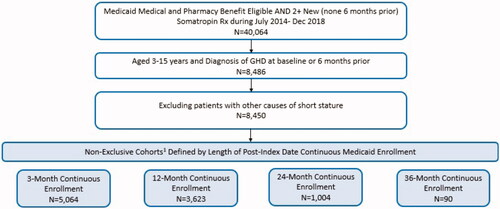
Table 1. Medicaid pGHD patient demographic and baseline clinical characteristics.
Figure 2. Suboptimal somatropin adherence (MPR <0.80) over 3 years among a Medicaid population diagnoses with pGHD. (a) Proportion of patients* with suboptimal somatropin adherence (MPR <0.80) among 3 pGHD cohorts with 1, 2, and 3 years of follow-up. ( b) Sensitivity analysis proportion of patients* with suboptimal somatropin adherence (MPR <0.80) at 1 and 2 years of follow-up among children with pGHD and 2 years of continuous follow-up. (c) Sensitivity analysis proportion of patients* with suboptimal somatropin adherence (MPR <0.80) at 1, 2, and 3 years of follow-up among children with pGHD and 3 years of continuous follow-up. *For categories with n < 11, descriptive statistics were unable to be presented due to Medicaid privacy regulations. Abbreviations. MPR, medication possession ratio; pGHD, pediatric growth hormone deficiency.
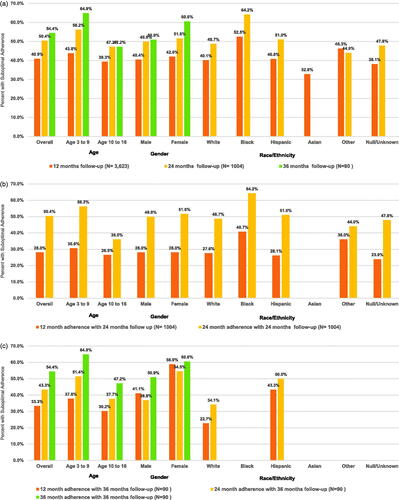
Table 2. Unadjusted logistic regression for baseline characteristics associated with suboptimal adherence to somatropin (MPR <0.80) among Medicaid pGHD cohorts with 1–3 years of follow-up.
Table 3. Unadjusted and adjusted Cox regression model results for baseline characteristics associated with time to discontinuation of somatropin among children with pGHD and ≥3 months of follow-up.
