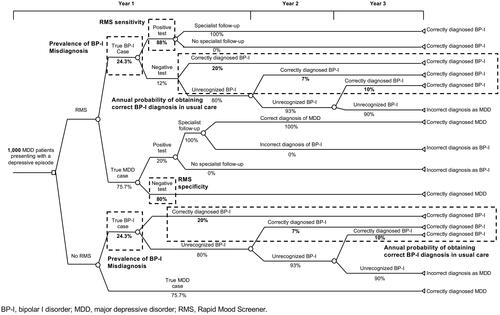
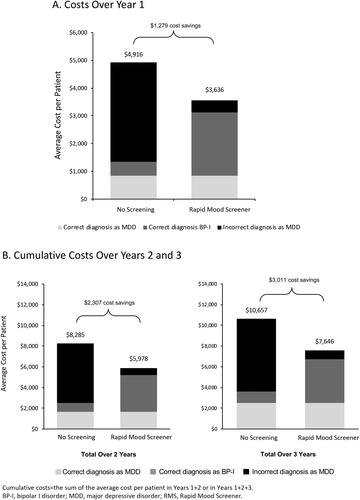
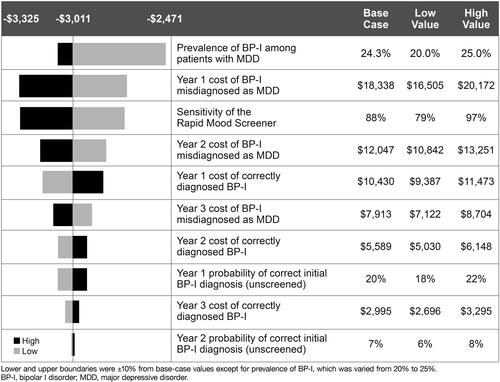
Figures & data
Table 1. Summary of epidemiology, RMS, and cost inputs.
Table 2. RMS screening versus no screening: number of correct diagnoses.
Data availability statement
AbbVie is committed to responsible data sharing. This includes access to anonymized, individual and/or study-level data (analysis data sets), as well as other information (e.g. protocols and Study Reports), as long as the studies are not part of an ongoing or planned regulatory submission and consistent with the informed consent conditions for the study. This includes requests for clinical trial data for unlicensed products and indications. Study data can be requested by any qualified researchers who engage in rigorous, independent scientific research, and will be provided following review and approval of a research proposal and Statistical Analysis Plan (SAP) and execution of a Data Sharing Agreement (DSA). For more information on the process, or to submit a request, contact the authors.