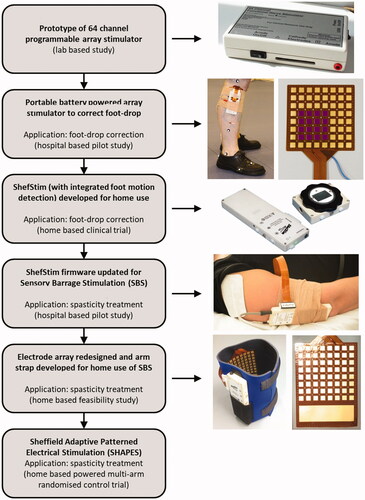
Figures & data
Table 1. Relevant standards, regulations and recommendations related to usability engineering of medical devices.
Figure 1. NIHR Devices for Dignity MIC innovation process [Citation22].
![Figure 1. NIHR Devices for Dignity MIC innovation process [Citation22].](/cms/asset/e7f27b54-9eb8-4279-980d-1f1222136281/ijmt_a_2089257_f0001_c.jpg)
Figure 3. Illustration of the relative size and positioning of SHAPES system in order for the treatment to be administered (stimulator in yellow, electrode array in green, arm sleeve in blue).
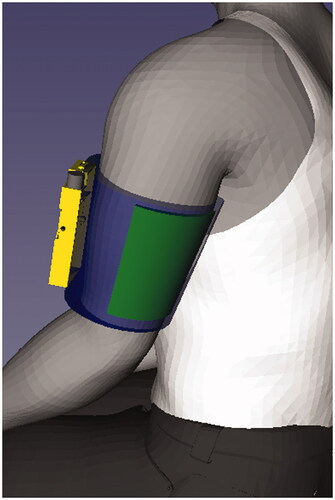
Figure 4. Models created for pre-pandemic usability studies of showing different shapes and forms for the stimulator (A). Models incorporating different control options were later produced (B) [Citation40].
![Figure 4. Models created for pre-pandemic usability studies of showing different shapes and forms for the stimulator (A). Models incorporating different control options were later produced (B) [Citation40].](/cms/asset/9ce67c0e-5f00-4c2c-8ff1-0d9caf472d80/ijmt_a_2089257_f0004_c.jpg)
Table 2. A summary of challenges faced and strategies employed in response.