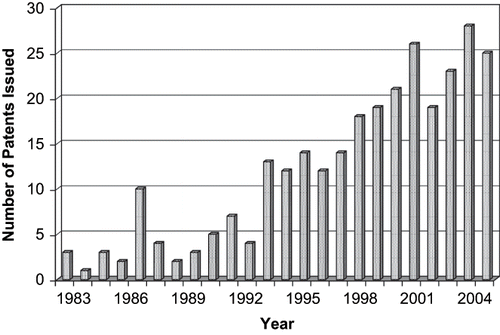
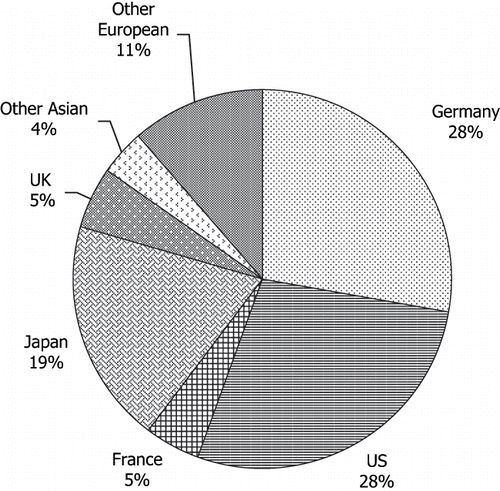
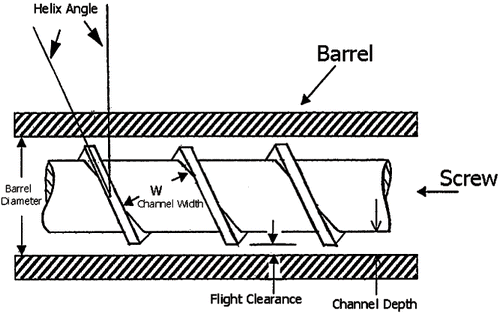
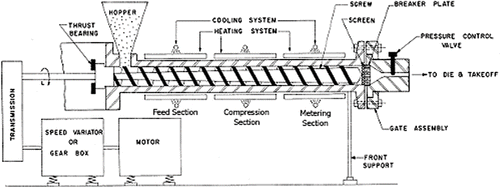
Figures & data
TABLE 1. Carriers Used to Prepare Hot-melt Extruded Dosage Forms
TABLE 2. Common Plasticizers Used in Pharmaceutical Dosage Forms
TABLE 3. Common Processing Aids Used in Hot-melt Extruded Dosage Forms
TABLE 4. Drug Substances Processed by Hot-melt Extrusion Techniques
TABLE 5. Common Methods Used for the Characterization of Hot-melt Extrudates