Figures & data
Fig. 1 Early symptoms of wasabi mottle virus development on wasabi leaves from natural infection. (a) Small chlorotic spots. (b, c) Chlorotic streaks developing along leaf veins. (d) Ringspot

Fig. 2 Advanced stages of natural infection by wasabi mottle virus on wasabi leaves. (a, b) Patterns of ringspots. (c) Ringspots and necrotic spots developing on leaves. (d) Extensive chlorotic streaks and vein-clearing. (e) Necrotic ringspots, vein clearing and extensive chlorosis. (f) Healthy leaf

Fig. 3 Symptoms following artificial inoculation of wasabi mottle virus on wasabi leaves. (a) Chlorotic spot. (b) Ringspot. (c) Necrotic ringspot 22 days after mechanical inoculation

Fig. 4 Symptoms following artificial inoculation of wasabi mottle virus on indicator plant hosts within 4–11 days after inoculation. (a) Nicotiana occidentalis. (b) Gomphrena globosa. (c) Tetragonia tetragonioides. (d) Chenopodium quinoa.

Fig. 5 Rod-shaped particles of wasabi mottle virus captured with a Hitachi H-7100 transmission electron microscope (100 kv) and Gatan Digital Micrograph software (v. 2.31.734.0; Gatan Inc., Pleasanton, CA, USA) after staining with uranyl acetate

Fig. 6 Detection of wasabi mottle virus with RT-PCR using primers designed from the full-length sequence. (a) Lanes 1–13 are from symptomatic naturally infected plants; C = control, L = molecular weight ladder. (b) Lanes A to G are from mechanically inoculated plants; C = control, L = molecular weight ladder. (c) Samples originating from 14 asymptomatic ‘Green Thumb’ plants show presence of the virus; L = molecular weight ladder. (d) Samples originating from 13 ‘Daruma’ plants show presence of the virus; C = control, L = molecular weight ladder
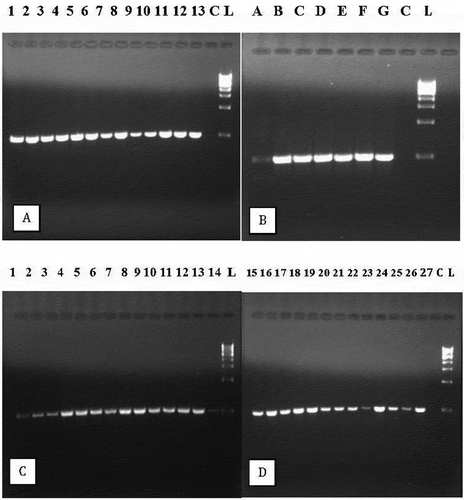
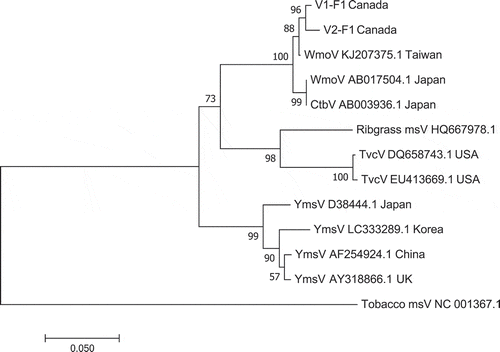
Fig. 7 Phylogenetic tree of wasabi mottle virus sequences from two different leaf samples in BC compared with sequences from GenBank. Number of bootstrap support values ≥50% based on 1000 replicates. Abbreviations are: WmoV = Wasabi mottle virus; Ctbv = Crucifer tobamovirus; Ribgrass msV = Ribgrass mosaic virus; TvcV = Turnip vein-clearing virus; YmsV = Youcai mosaic virus. The outgroup was Tobacco mosaic virus.