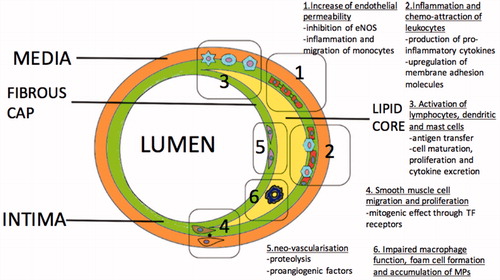
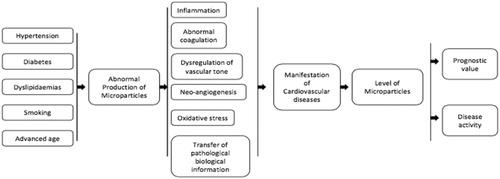
Figures & data
Table 2. Main antigen markers used for MP cell origin determination.
Table 3. Bioactive molecules of microparticles.
Table 4. Inflammation and microparticles (MPs).
Table 5. Angiogenesis and microparticles (MPs).
Table 6. Studies with microparticles (MPs) in diabetic populations or high glucose concentration conditions.
Table 7. List of medications induced reduction of microparticle (MP) levels in diabetic populations.
Table 8. Studies with microparticles (MPs) in acute coronary syndrome populations (ACS).
Table 9. Studies with microparticles (MPs) in heart failure.
Table 10. Studies where microparticle levels are associated with outcomes.