Figures & data
Figure 1 Transcellular Ca2+ and Pi transport. Active Ca2+ and Pi transport across renal and intestinal epithelium. A: Ca2+ influx is mediated by TRPV5 in the renal distal convoluted and connecting tubules and TRPV6 in the duodenum. Subsequently, Ca2+ is transported across the cell to the basolateral membrane by Ca2+‐binding proteins calbindin‐D28K (kidney) and calbindin‐D9K (intestine). Extrusion into the blood takes place via the plasma membrane Ca2+‐ATPase (PMCA1b) in the intestine and sodium‐Ca2+ exchanger (NCX1) and PMCA1b in the kidney. B: The ileal brush border, sodium‐dependent Pi cotransporter, NaPi‐IIb, mediates entry of Pi into enterocytes. In the proximal tubules, NaPi‐IIa and NaPi‐IIc are responsible for Pi entry into the tubular epithelial cells. The transport mechanisms involved in basolateral extrusion of Pi into blood are unknown both in the intestine and kidney.
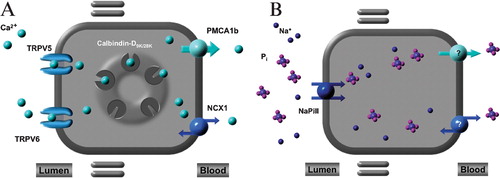
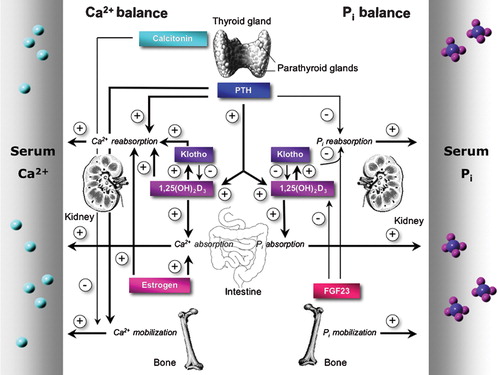
Figure 2 Key players in Ca2+ and Pi homeostasis. The concerted interplay of intestinal uptake, reabsorption in kidney, and bone (de)mineralization establishes the maintenance of a normal Ca2+ and Pi balance. The calcium sensing receptor (CaSR), present in the thyroid gland, senses blood Ca2+ levels and triggers the secretion of the calcitropic hormones parathyroid hormone (PTH) and calcitonin. Ovarian‐produced estrogen stimulates Ca2+ reabsorption also. Similar hormones are involved in the regulation of Pi balance. Blood Pi levels are controlled by PTH, 1,25(OH)2D3, klotho, and fibroblast growth factor member 23 (FGF23). A negative feedback mechanism prevents the accumulation of FGF23 and klotho since 1αOHase‐mediated 1,25(OH)2D3 production is inhibited by FGF23 and klotho.